Introduction
Materials and Methods
Results
Taxonomic Description
Habitat and Ecology
Key for identification of the genus Melilotus in Korea
Discussion
Introduction
Fabaceae Lindl. is the third largest family of flowering plants, containing 730 genera and more than 19,400 species distributed worldwide (Ahmad et al., 2016; Pedrol et al., 2018). Several species are economically important; for instance, legumes are used as a food source, and they fix atmospheric nitrogen via a symbiotic relationship with bacteria (Aboel-Atta, 2009; Conn et al., 2008; POWO, 2019; Seo et al., 2022; Yan et al., 2017). However, Fabaceae is also a family that has many species spread around the world because they coexist with bacteria and are highly adaptable even in barren areas (Fort et al., 2015; Zhao et al., 2021).
Melilotus (L.) Mill. (Fabaceae: Trifolieae) comprises 20 taxa, which are distributed in central and eastern Europe, Mediterranean countries, Asia, and North America (Bobrov, 1945; Isely, 1954; Langran et al., 2010). Many species are considered weeds in arable land, and they primarily grow in disturbed areas (Lee et al., 2022); in few areas, they are treated as invasive alien plants that cannot be controlled owing to their extensive spread (Conn et al., 2008; Conn and Seefeldt, 2009; GBIF Secretariat, 2021).
Morphologically, Melilotus is clearly distinguished from related genera based on characteristics such as the leaflet margin, stipules attached to the petiole base, long raceme, and legume character (Choi, 2018; Isely, 1954). In Korea, a total of four species of alien plants have been reported, including Melilotus suaveolens, Melilotus albus, Melilotus indicus, and Melilotus officinalis, which are native to Europe and Asia (Kim et al., 2019; Korea National Arboretum, 2021).
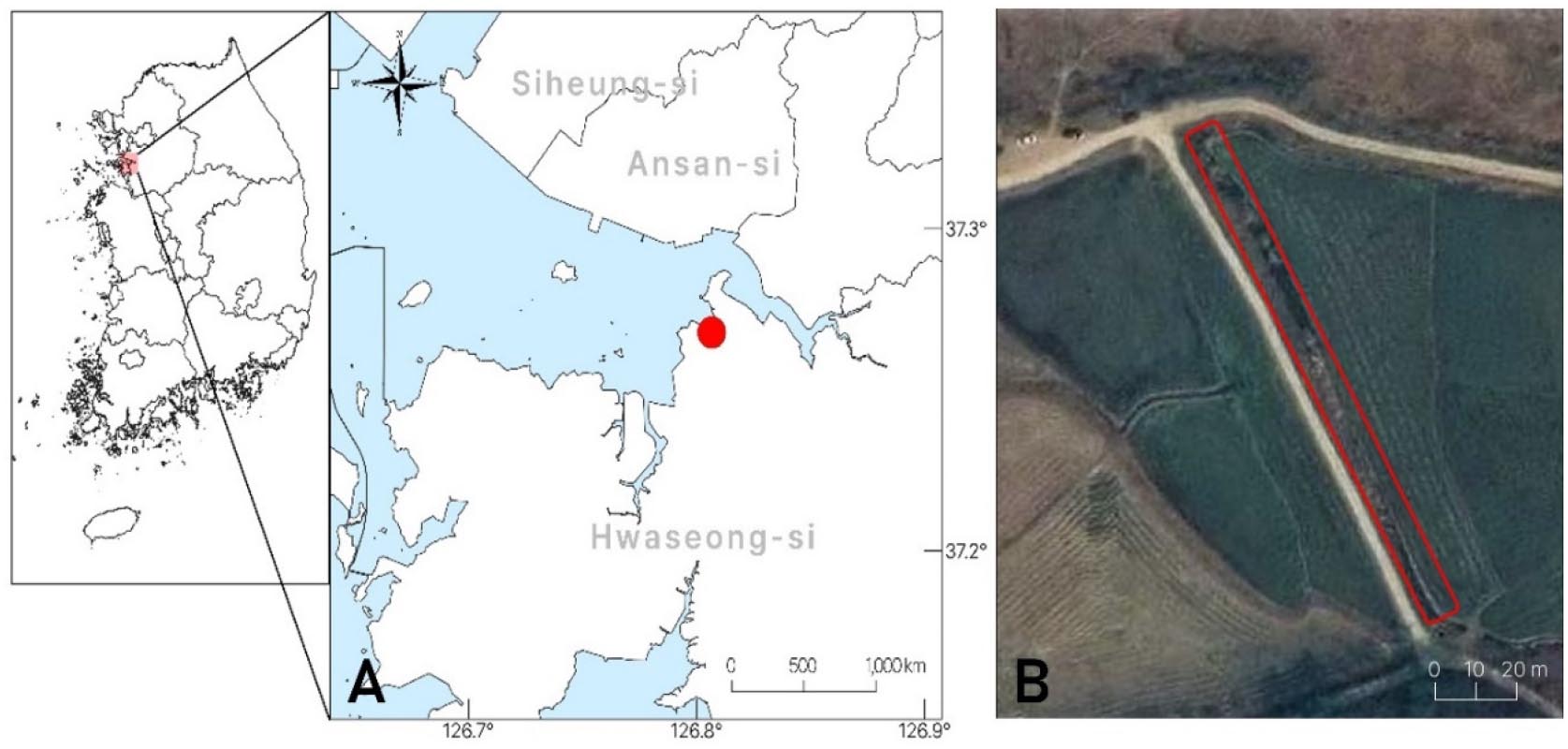
In this study, the identity and surrounding growth environment of Melilotus population found in Namyang-eup, Hwaseong-si, Gyeonggi-do were examined (Fig. 1). Based on the results, we provided the detailed species descriptions, photograph of Melilotus dentatus, and an identification key for all species of Melilotus that are found in Korea.
Materials and Methods
We examined the external morphological characteristics of live, immersed, and dried specimens collected between August 2021 and August 2022 from Namyang-eup, Hwaseong-si, Gyeonggi-do. An electronic Vernier caliper (Mitutoyo 500-196-30 absolute digimatic Vernier caliper; Mitutoyo, Kanagawa, Japan) was used to measure the plant characters, and the growth site and morphological features of plants were recorded using a digital camera (Nikon D810, Nikon 105 ㎜ AF Micro-Nikkor; Nikkon, Tokyo, Japan) and a stereo microscope (Leica MZ7.5, CLS 150X Microscope Cold Light Source; Leica, Wetzlar, Germany) to prepare photographs. We reviewed related literature for the identification of the species (Choi, 2018; Kim et al., 2019; Langran et al., 2010; Ohwi, 1965) and generated the species description and key to taxa based on our observations. The voucher specimens were stored in the Korea National Arboretum (KH).
Results
Taxonomic Description
Melilotus dentatus (Waldst. & Kit.) Desf., Tabl. École Bot. 188, 1804 (Fig. 2, 3).
Trifolium dentatum Waldst. & Kit., Descr. Icon. Pl. Hung. 1:41, 1801; Sertula dentata (Waldst. & Kit.) Kuntze, Revis. Gen. Pl. 1:205, 1891; Medicago dentata (Waldst. & Kit.) E.H.L.Krause, J.Sturm, Deutschl. Fl. Abbild., ed. 2. 9: 124, 1901; Trigonella dentata (Waldst. & Kit.) Coulot & Rabaute, Mesogene Biocosm 31: 149, 2014.
Herb biennial. Stems erect, branched, terete, longitudinally ridged, 50-140 ㎝ tall, red or green. Leaves alternate, 3-foliolate; stipules lanceolate, 1-2 pairs on petiole base, 6-8.4 ㎜ long, green, sometimes red at apex; petioles 6.2-10.8 ㎜ long, hair; leaflets oblong to oblanceolate, 2-3 × 0.4-0.5 ㎝, green, margin needle-shaped teeth, 9-12 pairs on a side, longest apex teeth. Inflorescence raceme, 2.4-5 ㎝ long, 28-43 flowers; peduncle and pedicel hair. Flowers calyx 1.8-2.2 ㎜ long, hair, lobes 5, lanceolate; petals 3.4-4.3 ㎜ long, standard slightly longer than wings and keel, yellow; stamen 10, diadelphous; pistil 1; ovary elliptic, ovule 2. Fruits legume, ovate to globular, 3.3-3.5 × 2-2.4 ㎜, green or partially pale red, surface minute reticulate nerved or smooth. Seeds 1-2, broadly elliptic to rounded, 1.8-2.4 × 1.6-1.8 ㎜, dark green or greenish brown.
Korean name: Seo-yang-jeon-dong-ssa-ri (서양전동싸리)
English name: Banat sweet clover
Flowering and fruiting season: August to October
Distribution: Europe, Central Asia, China / North America, Japan, Korea (introduced)
Distribution in Korea: Gyeonggi-do (Hwaseong)
Specimens examined: South Korea. Gyeonggi-do: Hwaseong-si, Namyang-eup, munho-ri, 18. Aug. 2021, J. S. Kim KJS938 (3 sheets, KH); same locality, 26 Aug. 2021, K. H. Lee and B. K. Park munhori 210826 (2 sheets, KH); same locality, 16 Aug. 2022, E. S. Kang and S. R. Lee munhori 220816 (8 sheets, KH).
Habitat and Ecology
Melilotus dentatus mainly inhabits areas affected by frequent disturbances, such as grasslands, farmlands, parks, and industrial facilities (Tabaka et al., 1988). In Korea, M. dentatus was found on the roadside between paddy fields in moist soil and sunny environments (Fig. 1, 2A). Regarding surrounding plants, Rubus parvifolius L., Morus bombycis Koidz., Morus alba L., Rosa multiflora Thunb., and Robinia pseudoacacia L. form a shrub layer. In the herbaceous layer, Conyza canadensis (L.) Cronquist, Artemisia indica Willd., Artemisia codonocephala Diels, Oenothera biennis L., Pennisetum alopecuroides (L.) Spreng., Sonchus brachyotus DC., Eleusine indica (L.) Gaertn., Rubia cordifolia L., Symphyotrichum pilosum (Willd.) G.L.Nesom, Cirsium japonicum Fisch. ex DC. var. maackii (Maxim.) Matsum., Leonurus japonicus Houtt., Lythrum salicaria L., Calystegia dahurica (Herb.) Choisy, Metaplexis japonica (Thunb.) Makino, Commelina communis L., Aegilops cylindrica Host, and Rumex crispus L. grow together.

Fig. 2.
Photographs of Melilotus dentatus (Waldst. & Kit.) Desf. A. Habitat, B. Stem, C. Branched stem (the red arrow indicates longitudinally ridges in the stem), D. Inflorescence, E. Stipule (the red circle indicates a stipule at the base of the petiole), F. Adaxial leaf, G. Abaxial leaf, H. Leaflet, I. Flower (standard slightly longer than wings and keel), J. Diadelphous stamens, K. Pistil, L. Fruit, M. Seed.
Key for identification of the genus Melilotus in Korea
1. Flowers white M. albus Medik. 흰전동싸리
1. Flowers yellow.
2. Plants 20-50 ㎝ tall; flowers 2-3 ㎜ long M. indicus (L.) All. 좀전동싸리
2. Plants 50-150 ㎝ tall; flowers 3.4-7 ㎜ long.
3. Stipules lanceolate; leaflets needle-shaped teeth; fruits ovate to rounded; seeds dark green or greenish brown M. dentatus (Waldst. & Kit.) Desf. 서양전동싸리
3. Stipules linear; leaflets serrate; fruits ovate-elliptic; seeds yellowish brown.
4. Leaflets oblanceolate to obovate; flowers 3.5-5 ㎜ long; calyx lobes shorter than calyx tubes; fruits reticulated nerved surface M. suaveolens Ledeb. 전동싸리
4. Leaflets elliptic to oblong; flowers 5-7 ㎜ long; calyx lobes same as calyx tube; fruits transverse nerved surface M. officinalis (L.) Lam. 주름전동싸리
Discussion
Most Melilotus species are native to Europe, Central Asia, and China. Currently, M. albus, M. officinalis, and M. dentatus have been introduced into North America in the United States and into Canada (GBIF Secretariat, 2021; Tabaka et al., 1988; Turkington et al., 1978). Melilotus species are mainly found in artificial environments, such as cultivated fields, parks, and gardens (POWO, 2019).
Common characteristics among Korean Melilotus taxa include 3-foliolate leaves, stipules on petiole base, long racemes, and elliptic to globular legumes. M. dentatus plants are 50-140 ㎝ tall, display yellow flowers, and the leaves and flowers are less than 1 ㎝ long when viewed with the naked eye. M. suaveolens cannot be easily distinguished from M. officinalis. However, the oblong leaflets of M. dentatus can be distinguished from the leaflets of M. suaveolens (Fig. 2H). Furthermore, M. dentatus has a calyx length of 1.8-2.2 ㎜ and reticulated or smooth fruit surface, whereas M. officinalis has a calyx length of 2.5-3 ㎜ and a transverse nerved fruit surface. M. dentatus has lanceolate stipules (Fig. 2E), needle-shaped leaflet margin, and dark green or greenish brown seeds (Fig. 2M) which can be distinguished from features of other species of Melilotus (Bobrov, 1945; Ohwi, 1965; Choi, 2018; Kim et al., 2019).
In comparison with other legumes, M. dentatus has high resistance to drought, cold damage, and pests, and it has a low coumarin content, which makes it suitable for fodder production. M. dentatus is particularly useful for re-cultivation and soil restoration in salty soils (Muntyan et al., 2012). However, few studies have raised concerns regarding the environmental impacts of Melilotus. M. albus and M. officinalis have been reported to adversely affect native species in the Rocky Mountains of Colorado, USA, leading to a decrease in biodiversity (Wolf et al., 2003). Additionally, seeds can germinate in soil without dormancy for at least 20 years, even in low-temperature regions like Alaska. The seeds spread rapidly along the water system to form a new population, and controlling their spread is difficult (Conn et al., 2008; Conn and Seefeldt, 2009).
Although there are no domestic ecological risk studies on M. dentatus, it is presumed that M. dentatus was first introduced unintentionally together with crop seeds from arable land, considering the cases of overseas spread and the surrounding environment. It is highly likely to spread across arable land. Since there is a concern that M. dentatus may have a direct impact on the production of crops in arable land in the future, additional research on the spread and inflow is necessary, and the preparation of a continuous monitoring and management plan is urgently required.