Introduction
General aspects of conserved terpenoids between Brassicaceae and Solanaceae
Major terpenoids commonly produced in Brassicaceae and Solanaceae
Combined roles of terpenoids
Conclusion
Introduction
Plants produce various types of primary and secondary metabolites during their life cycle. Terpenoids constitute the largest and most diverse class of metabolites, comprising over 80,000 compounds that play various roles in a wide range of ecological niches (Boncan et al., 2020). As knowledge of the functions of terpenoids accumulates, their utilization has been extended to a wide range of applications across diverse field including agricultural, pharmaceutical, and medical industries. The high structural diversity of terpenoids is attributed to various precursors, intermediates, and biosynthetic enzymes (e.g., terpenoid synthase and P450) present in plants (Boutanaev et al., 2015; Ninkuu et al., 2021; Roba, 2020; Singh et al., 2015; Vivaldo et al., 2017). Terpenoids are synthesized from the five-carbon precursor, isopentenyl pyrophosphate (IPP) and its isomer dimethylallyl pyrophosphate (DMAPP). Subsequently, IPP and DMAPP are condensed by isoprenyl diphosphate synthase (prenyltransferase), which forms isoprenyl diphosphate/pyrophosphate (ID C5n). Subsequently, IDs are converted into intermediates including geranyl pyrophosphate (GPP, C10), farnesyl pyrophosphate (FPP, C15), and geranylgeranyl pyrophosphate (GGPP, C20), which are further diversified by terpene synthases (TPSs). Further modifications, mainly mediated by cytochrome P450 oxygenases, yield terpenoids and their oxidized derivatives. Terpenoids are generally classified based on the number of carbon atoms present in their skeleton: hemiterpenoids (C5), monoterpenoids (C10), homoterpenoids (C11,16), sesquiterpenoids (C15), diterpenoids (C20), sesterpenoids (C25), triterpenoids (C30), tetraterpenoids (C40), and high-order polyterpenoids.
In plants, terpenoids play various roles such as serving as building blocks, contributing to allelopathy and plant defense, acting as insecticides/repellents, attracting pollinators, and interfering with plant hormones. Terpenoids are primary metabolites and mainly function as phytohormones (i.e., gibberellins, cytokinins, auxins, brassinosteroids, and strigolactones), regulating developmental processes, plant growth, and adaptation to various biotic and biotic stresses (Roba, 2020). Terpenoids are secondary metabolites that play diverse roles in plant defense against herbivores, disease resistance, attraction of mutualists (e.g., pollinators), and plant-to-plant communications (Ninkuu et al., 2021). The beneficial effects of terpenoids have also been reported in belowground ecological niches (Huang and Osbourn, 2019). Moreover, the production of terpenes by plants in response to biotic and abiotic stress has been extensively studied. Terpenoids are the key bioactive compounds in plant essential oils and are widely used in the medical and food industries to improve human health (Masyita et al., 2022).
Regarding the functional lineage of plant families, some terpenoids exert distinct functions that are restricted to certain species (Zhou and Pichersky, 2020). Therefore, the identification of unique terpenoids from the entire terpenome for each family or species could enhance our understanding of the role of a certain terpenoid in the growth and development of plants. However, many terpenoids play several common roles in a broad range of plant species. Terpenoids are commonly produced across genera owing to their evolutionary conservation in biosynthetic pathways and responses to environmental cues. The identification of terpenoids commonly present in different plant families might provide insights into the cross-talk associated with terpenoids across plant families.
Brassicaceae (or Cruciferae) and Solanaceae are major economically important families of Angiosperms. Brassicaceae comprises 372 genera and 4,060 species, including vegetables such as Brassica oleracea (cabbage, kale, cauliflower), Brassica rapa (turnip, Chinese cabbage, etc.), Brassica napus (rapeseed), Raphanus sativus (radish), Armoracia rusticana (horseradish), and Arabidopsis thaliana (thale cress). The most distinct developmental morphology in this family is the formation of four free sepals and petals. Many species belonging to Brassicaceaeare economically important sources of vegetables, oils, fodder, and ornamental products. In addition, several species are utilized for medicinal purposes, such as asthma, hemorrhage, and syphilis. Similarly, Solanaceae is another large family comprising economically important members. This family includes 98 genera and 2,700 species, such as Solanum lycopersicum (tomato), Solanum tuberosum (potato), Solanum melongena (eggplant), Capsicum annuum (chili peppers), and Nicotiana tabacum (tobacco). They are also utilized and studied in various industrial fields such as food, medicine, narcotics, and ornamentation (Lee et al., 2024a; Lee et al., 2024b).
In this study, we explored the Plant Secondary Metabolite Database (http://pscdb.appsbio.utalca.cl, PSC-db, Valdés-Jiménez et al., 2021) and recent literatures on plant terpenoids. Although the complete terpenome of the plant kingdom is yet to be analyzed, we extracted terpenoids commonly produced by Brassicaceae(or Cruciferae) and Solanaceae. In addition, we explain the general functions of the selected terpenoids that have been experimentally investigated in the literature. Furthermore, we discuss the cross-talk among terpenoids and their roles in phytohormone-mediated signaling pathways.
General aspects of conserved terpenoids between Brassicaceae and Solanaceae
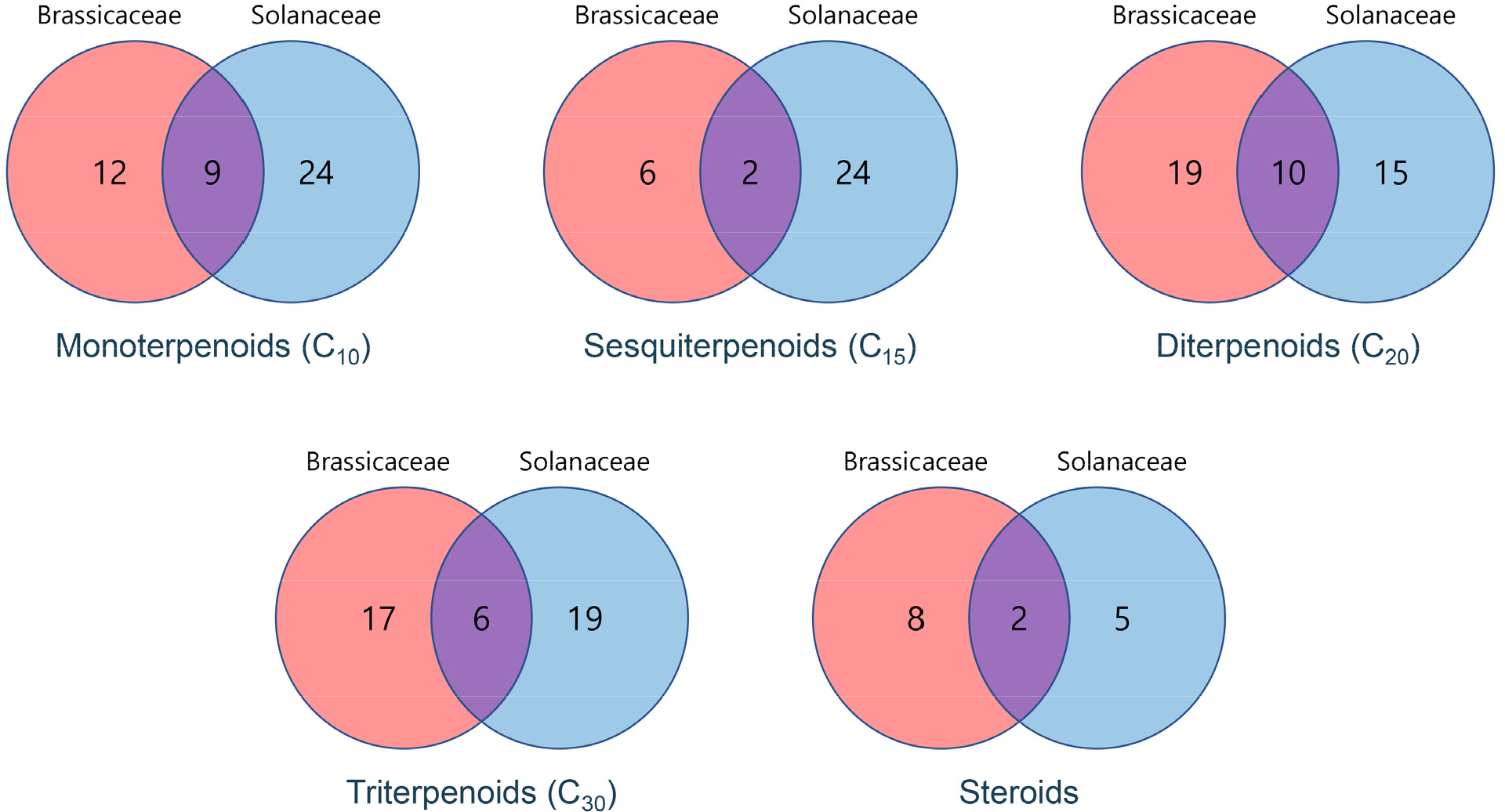
Currently, the PSC-db contains information on 2,853 chemicals derived from 12,096 plant species, as reflected in 85,677 resources (Valdés-Jiménez et al., 2021). Although PSC-db represents the most comprehensive resource to identify plant terpenoids to date, it provides limited information insufficient for covering the entire terpenome. Nevertheless, we focused on terpenoids commonly detected in Brassicaceae and Solanaceae in the PSC-db. Overall, the number of terpenoids identified from PSC-db was 62 and 87 chemicals for Brassicaceae and Solanaceae, respectively. Among these, 29 chemicals have been listed in both plant families (Fig. 1), which included nine monoterpenoids (C10), 2 sesquiterpenoids (C15), 10 diterpenoids (C20), 6 triterpenoids (C30), and 2 steroids were commonly identified (Table 1). Among these, several terpenoids are the derivatives of gibberellins (diterpenoids) and steroids/sterols (tetraterpenoids), which are primary metabolites. In the case of sesqui-, di-, and mono-terpenoids, chemicals such as β-caryophyllene, phytol, limonene, 1,8- cineole, α-/β-pinene, linalool, terpineol, sabinene, and myrcene were disclosed to be conserved between two plant families. The potential biological roles of these terpenoids are discussed as follows (Table 2).
Table 1.
The list of terpenoids commonly identified in PSC-db.
Table 2.
The biological roles of several selected terpenoids commonly produced in Brassicaceae and Solanaceae.
| Terpenoid | Reported functions | References |
| β-Caryophyllene |
•Recruited natural enemy against pests, pollinators, and herbivores •Overexpression of caryophyllene synthase gene attracted parasitoids against pests in transgenic rice | Chiriboga et al., 2017Cheng et al., 2007 Zhang et al., 2020 Tamiru et al., 2017 |
|
•Exhibited insecticidal activity against wheat weevil •Attracted rice gall midge and repelled multiple pests | Plata-Rueda et al., 2018 Winnacker and Rieger, 2015 | |
| •Showed antimicrobial activity against Pseudomonas syringae | Huang et al., 2012 | |
|
•Allelopathic activity in pines against herbaceous plants •Modulated JA/SA signaling and biotic resistance in receiver plants | Santonja et al., 2019Frank et al., 2021 Rosenkranz et al., 2021 Kong et al., 2021 | |
| Limonene | •Attracted pollinators | Byers et al., 2014Chen and Song, 2008 |
| •Showed insecticidal activity | Hollingsworth, 2005 | |
| •Modulated the balance between SA and JA signaling in plants | Maffei et al., 2001Chaimovitsh et al., 2017 Rodríguez, 2014 | |
| 1,8-Cineole |
•Attracted pollinators •Defended plants against mechanical damages from insects •Recruited parasitic wasps in cabbage | Dodson et al., 1969Chacón-Fuentes et al., 2019 Ahuja et al., 2010 Korankye et al., 2017 |
|
•Involved in senescence in plant cells •Exhibited allelopathic effects on weed plants | Romagni et al., 2000Scogmamiglio et al., 2013 | |
| α/β-piene | •Showed insecticidal activity | Arthur and Hain, 1987Plata-Rueda et al., 2018 |
| •Attracted pollinators or repellent insects | Chen et al., 2008Ali et al., 2011 Naidoo et al., 2018 | |
| •Modulated stress responses and senescence in plants | Singh et al., 2006Weir et al., 2004 | |
| Linalool | •Recruited pollinators such as wasp and syrphid flies | Chen and Song, 2008Majetic et al., 2009 Dobson, 2006 |
| •Attracted pest predator or repellent insects | Xu et al., 2015 | |
| •Served to defend plants against pests and pathogens | Ogah et al., 2017 Boncan et al., 2020 | |
| Myrcene |
•Altered interaction with pollinators •Acting as toxic compound against pests | Terry et al., 2007Arthur and Hain, 1987 |
Major terpenoids commonly produced in Brassicaceae and Solanaceae
1) β-Caryophyllene
(E)-β-Caryophyllene is a natural bicyclic sesquiterpene produced in a wide range of plant species. Attacks by herbivores induce the biosynthesis of (E)- β-caryophyllene to attract natural enemies or predators to combat them (Ninkuu et al., 2021). For example, (E)-β-caryophyllene recruited entomopathogenic nematodes against rootworm in corn (Chiriboga et al., 2017). Moreover, the overexpression of genes, such as OsTPS3 and GhTPS1, which encode biosynthetic enzymes (e.g., caryophyllene synthase), negatively affects multiple pests while attracting parasitoids in transgenic rice and cotton (Cheng et al., 2007; Zhang et al., 2020). (E)-β-caryophyllene released under the ground by maize roots recruits entomopathogenic nematodes (Tamiru et al., 2017). (E)-β-caryophyllene also attracts detrimental herbivores such as Cotesia sesamia (larval parasitoid) of Chilo partellus (spotted stalk borer), which reduced the productivity of cereals (Tamiru et al., 2017). Practically, these properties of caryophyllene can be utilized as a decoy to trap and control herbivores.
Furthermore, the insecticidal activity of (E)-β-caryophyllene was reported. In cinnamon and clove, caryophyllene exhibits insecticidal activity against Sitophilus granarius (Plata-Rueda et al., 2018). Rice-derived caryophyllene attracts the African rice gall midge, which repels pests in a concentration-dependent manner (Ogah et al., 2017). The essential oils from Cannabis sativa, comprising 45.4% (E)-β-caryophyllene, exhibited efficient insecticidal activities against various aphids, worms, house flies, and mosquitoes (Winnacker and Rieger, 2015). Additionally, (E)-β-caryophyllene inhibited a rod-shaped Gram-(-) Pseudomonas syringae on Solanaceae and Arabidopsis plants, which reflected its antimicrobial activity (Huang et al., 2012).
(E)-β-Caryophyllene exhibits allelopathic behavior and is involved in plant-to-plant interactions. For instance, (E)-β-caryophyllene excreted from the roots and pines of Pinus halepensis inhibited the vegetative growth of the plants (Santonja et al., 2019). In the receiver leaves, β-caryophyllene exposure induced jasmonic acid (JA) signaling and an associated resistance reaction (Frank et al., 2021). It also regulates belowground plant-to-plant cues, adjusting the germination, growth, and defense of sympatric neighboring plants of Centaurea stoebe (Gfeller et al., 2019; Huang and Osbourn, 2019). (E)-β-caryophyllene is strongly correlated with JA-mediated immunity. The biosynthesis of (E)-β-caryophyllene was enhanced in Arabidopsis upon the activation of MYC2, a transcription factor that was central to JA signaling response (Rosenkranz et al., 2021). (E)-β-Caryophyllene released by tomato triggered defense responses in neighboring plants, strongly enhancing the salicylic acid (SA) levels in the root exudates of receiver plants (Kong et al., 2021).
2) Limonene
Limonene is a cyclic monoterpene and a major component of the aromatic scents and resins of many coniferous and broadleaved plants. In Oryza sativa (rice), limonene synthesis was enhanced by high UV-B, gamma irradiance, and H2O2 treatments (Lee et al., 2015). In terms of function, D-limonene is largely involved in interactions with insects. For instance, D-limonene attracts bumblebees and wasps to monkey flowers and fig (Ficus hispida) to facilitate pollination (Byers et al., 2014; Chen and Song, 2008). It also exhibits antimicrobial, antioxidant, anti-inflammatory, antinociceptive, anticancer, and insecticidal properties (Erasto and Viljoen, 2008). As an insecticide, (+)-R-limonene from citrus extracts deters mealybugs and aphids (Hollingsworth, 2005). In addition, limonene accumulates in the resin ducts found in needles, twigs, and trunk poses, acting as a toxic compound against serious conifer pests such as bark beetles and balsam woolly adelgids (Arthur and Hain, 1987). In contrast, limonene functions as a semiochemical or allelopathic agent that influences plant development processes, including leaf expansion, floral organ formation, and fruit ripening. When cucumber seedlings were introduced to limonene containing essential oils, the plants underwent senescence, which was considerably driven by membrane depolarization (Maffei et al., 2001). In addition, limonene, which is abundantly produced by transgenic Arabidopsis, exhibits strong antimicrobial activity (Chaimovitsh et al., 2017). Interestingly, D-limonene accumulation in orange peels modulated the balance between SA and JA. High levels of D-limonene are positively correlated with the pathogen- induced priming of SA defenses, whereas reduced D-limonene levels are associated with JA immunity (Rodríguez et al., 2014). In many cases, limonene, a volatile compound, act as a signal that triggers its functions at a distance. Owing to its high vapor density, limonene easily penetrates soil, adversely affecting underground organisms (Erasto and Viljoen, 2008).
3) 1,8-Cineole
1,8-cineole, also known as eucalyptol, is a monoterpenoid mainly present in eucalyptus, rosemary, and camphor laurel. Their primary role is to mediate the mutualistic and parasitic interactions with insects. In tropical orchids, secreted 1,8- cineole attracts male euglossine bees serving as pollinators (Dodson et al., 1969). This volatile compound is involved in the defense of murtilla plants against insect-induced mechanical damage (Chacón-Fuentes et al., 2019). 1,8-cineole recruited parasitic wasps (Cotesia glomerata) to lay eggs in the caterpillars that attack cabbage (Ahuja et al., 2010; Dicke et al., 2004).
1,8-cineole is also involved in the stress-responsive senescence in plant cells (Korankye et al., 2017). For instance, membrane integrity and polarization were disrupted in cucumber seedlings exposed to 1,8-cineole containing essential oil (Maffei et al., 2001). Additionally, the analogs of 1,8-cineole severely inhibited the growth of roots and shoots, causing a cork-screw shaped morphological distortion. It also inhibited the germination in the weedy species Cassia obtusifolia and Echinochloa crus-galli (Romagni et al., 2000). In particular, 1,8-cineole reduced photosynthetic yield and mitosis at all growth stages. 1,8-cineole also exhibits allelopathic effects, prolonging the germination time and suppressing the development of weeds (Scogmamiglio et al., 2013). It inhibited plant cell proliferation and meristematic DNA synthesis in neighboring plants (Abd-Elgawad et al., 2021; Martino et al., 2010). In contrast, 1,8-cineole has been utilized in traditional medicine, which exhibits anti-inflammatory, antioxidant, antimicrobial, bronchodilatory, analgesic, and pro-apoptotic effects (Hoch et al., 2023).
4) α/β-Pinene
Pinene is a group of bicyclic monoterpene which has two isomers, α-pinene and β-pinene. These isomers are mainly found in conifers in Douglas fir, their synthesis is strongly induced by drought and high salt conditions (Kleiber et al., 2017; Nyamwihura and Ogungbe, 2022). Their main biological role is to inhibit various insects. They are toxic to conifer pests, including bark beetles and balsam woolly adelgids (Arthur and Hain, 1987). In cinnamon and clove, α-pinene exhibited insecticidal activity against wheat weevil (Sitophilus granarius) (Plata-Rueda et al., 2018). Pinene also serves as pollinator-recruiting agent and contributes to the interaction between fig (Ficus hispida) and wasps (Ceratosolen solmsimarchali) during the floral stages of host plants (Chen and Song, 2008). β-pinene was induced by Diaprepes abbrevatus larvae and recruited entomopathogenic nematodes in swingle (Ali et al., 2011). α-pinene from Eucalyptus grandis also attracted pest Leptocybe invasa (Naidoo et al., 2018).
Moreover, pinene is involved in the plant stress response and senescence. A high concentration of pinenes is detrimental to plant cell membranes. α-pinene inhibited radicle growth in Cicer arietinum by promoting solute leakage from the roots and enhancing the levels of malondialdehyde (MDA), proline, and hydrogen peroxide (Singh et al., 2006). It also inhibits seed germination in Pisum sativum and Zea mays. Furthermore, β-pinene was reported to inhibit the shoot and root growth in Phalaris minor, Echinochloa crus-galli, and Cassia occidentalis. Specifically, β-pinene reduced Cr (VI)- induced accumulation of reactive oxygen species in maize. α-pinene inhibited early root growth and caused oxidative damage in the root tissue through enhanced reactive oxygen species (ROS) generation and lipid peroxidation, disrupted membrane integrity and elevated antioxidant enzyme levels (Singh et al., 2006; Weir et al., 2004). Both α- and β- pinenes repress nuclear and organelle DNA synthesis in the root apical meristem of Brassica campestris (Nishida et al., 2005).
In contrast, pinenes exhibit a broad spectrum of pharmacological activities, including the modulation of antibiotic resistance and anticoagulant, antitumor, antimicrobial, antioxidant, anti-inflammatory, and analgesic effects. They also exhibit cytogenetic, gastroprotective, anxiolytic, cytoprotective, anticonvulsant, neuroprotective effects and protective effects against oxidative stress (Salehi et al., 2019).
5) Linalool
Linalool, an acyclic monoterpene, is one of the most common flavor-associated volatiles in the plant kingdom (Zhang et al., 2023). Linalool-emitting flowers attract a wide range of pollinators, although some species exhibit a strong preference for these flowers. Linalool, derived from fig (Ficus hispida), recruits wasps (Ceratosolen solmsi marchali) and acts as a signal for pollinators to identify the floral stages of the host plants (Chen and Song, 2008). Linalool protects plants against insect pests and pathogens. Heterologous overexpression of the linalool synthase gene decreases aphid attacks on strawberries (Boncan et al., 2020). Notably, the two linalool enantiomers exhibit distinct functions, (S)-(+)- Linalool mainly attracts pollinators, whereas (R)-(-)-linalool acts as an insect repellent. Linalool recruits various pollinators, such as syrphid flies in sweet rocket (Majetic et al., 2009; Dobson, 2006). Linalool attracts the pest predator Chrysopa phyllochroma to promote oviposition (Xu et al., 2015). In rice, linalool repels pests in a concentration- dependent manner (Ogah et al., 2017). Furthermore, linalool exhibits allelopathic activity and promotes the germination of stifled weeds (Abd-Elgawad et al., 2021).
6) Myrcene
Myrcene, a monoterpene derived from geranyl pyrophosphate (GPP) is abundant in hops and cannabis. Especially, β-myrcene is known to enrich the aroma of beer. Myrcene synthesis is promoted in rice exposed to high UV-B, gamma irradiation, and hydrogen peroxide treatment (Lee et al., 2015). β-Myrcene from Pineapple zamia altered the interactions with pollinators, thrips (Cycadothrips chadwicki) (Terry et al., 2007). In terms of insecticidal activity, myrcene acts as a toxic compound against important pests such as bark beetles and balsam woolly adelgids (Arthur and Hain, 1987).
7) Phytol and terpineol
Phytol is an acyclic hydrogenated diterpene alcohol often used as a precursor during the manufacture of synthetic forms of vitamins E and K1. Phytol acts as a chemical inhibitor for several insects in plants. Interestingly, insects, such as the sumac flea beetle, use phytol originating derived from their host plants to expel predators. In water primrose, phytol exhibits insecticidal activity against webbers (Mitra et al., 2017).
Terpineols are a group of isomeric monoterpenoids commonly used in perfumes, cosmetics, and flavors owing to their pleasant odor. Khaleel et al. (2018) reported that terpineols exert various biological effects including antihypertensive, antioxidant, anticancer, antinociceptive, antiulcer, anticonvulsant, sedative, antibronchitis, and insecticidal activities.
Combined roles of terpenoids
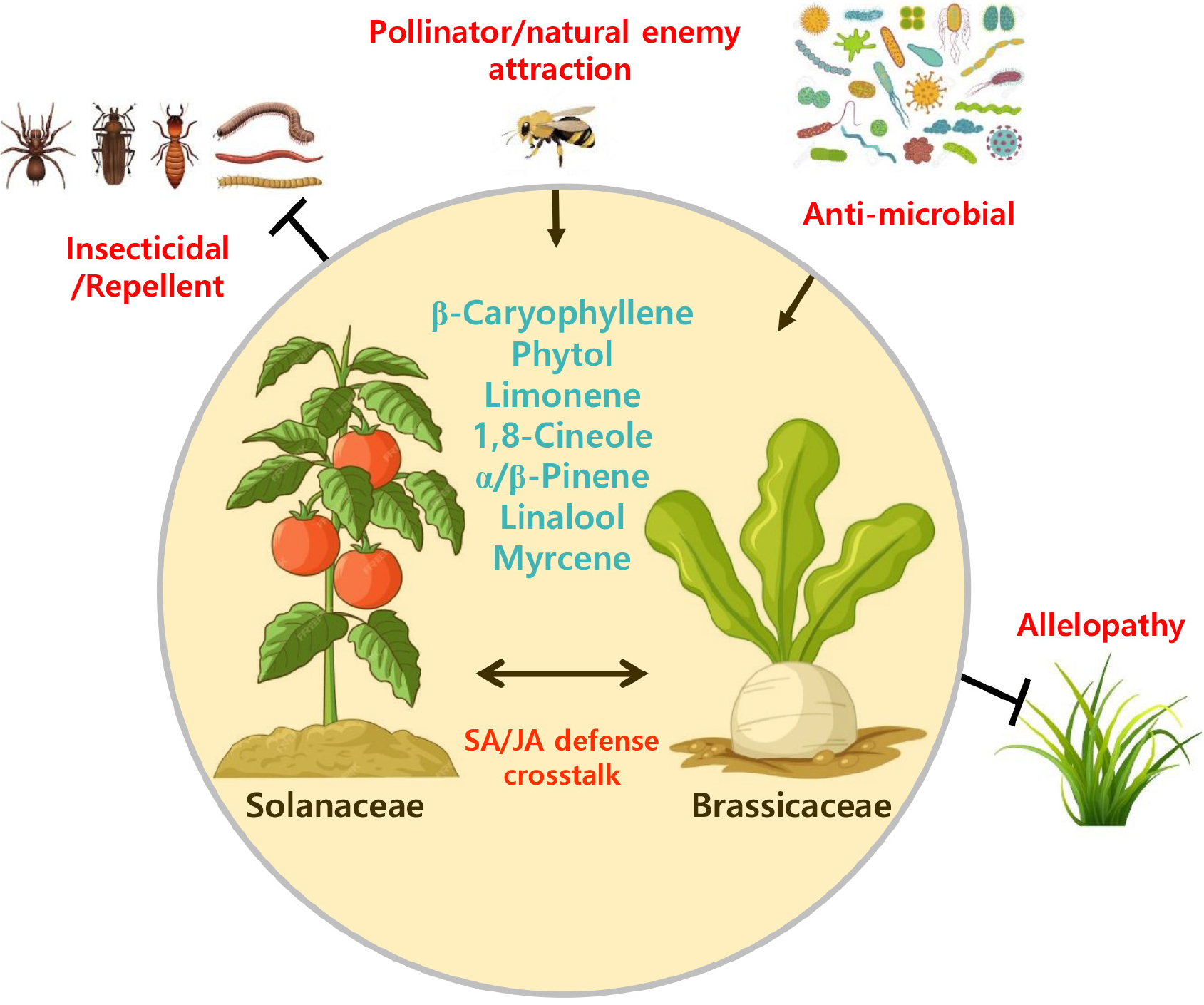
In this article, we assessed and briefly introduced the suggested functions of β-caryophyllene, limonene, 1,8-cineole, α/β-pinene, linalool, myrcene, phytol, terpineol, the representative terpenoids commonly produced in members of Brassicaceae (or Cruciferae) and Solanaceae. All terpenoids introduced in the current study were largely involved in interactions with insects, such as pollinators, herbivores, and/or their natural enemies (Fig. 2). In some cases, insecticidal or antimicrobial activities have been reported. In addition, most terpenoids modulate the growth and development of neighboring plants in a phenomenon known as allelopathy. Terpenoids stimulate or trigger SA or JA signaling in plants exposed to biotic stresses. The intensity of the outcome of each function has been suggested to be dependent on the concentration of terpenoids, plant species, developmental stages of plants, and their interactors, such as insects. Overall, terpenoids showed versatile functions with the great potential as semiochemicals, biostimulators, or biocontrol agents in agricultural practice. In addition to intra-family signaling, several studies have demonstrated that terpenoids mediate cross-family communications between Brassicaceae and Solanaceae plants through phytohormonal cross-talk. For instance, both A. thaliana and Solanum lycopersicum release (E)-β-caryophyllene in response to the attack by herbivores via JA-dependent pathways. Airborne β-caryophyllene emitted from tomato induces salicylic acid accumulation and defense priming in neighboring Arabidopsis roots (Kong et al., 2021). Similarly, monoterpenes such as linalool and α/β-pinene differentially modulate JA and SA-dependent antibacterial responses in Arabidopsis (Huang et al., 2012; Rodríguez et al., 2014). These findings indicate that terpenoids act as intra-species defense signals as well as contribute to the ecological cross-talk between Brassicaceae and Solanaceae through shared volatile-mediated networks that influence both hormone signaling and rhizosphere interactions.
Conclusion
Our study reflects that the members of, Brassicaceae (or Cruciferae) and Solanaceae commonly produce multiple terpenoids via under genetically and environmentally regulated process. These terpenoids play various biological roles by creating barriers between plants and other organisms, such as insects and, microbes. The terpenoids can manipulate and optimize plant responses to biotic and biotic environmental changes at the community level. The apparent connections between different terpenoids and phytohormone signaling cascades regulate the fine-tuning of the overall plant responses to multiple stresses through phytohormone cross-talk. Despite the availability of limited information, an understanding of the synthesis and functions of terpenoids in plants belonging to Brassicaceae (or Cruciferae) and Solanaceae would facilitate the development of effective strategies to improve crop productivity and support the sustainable maintenance of green ecosystems.