Introduction
Materials and Methods
Animals and reagents
Preparation of PG
Experimental colitis model construction and PG treatment
Disease activity index
Histological evaluation
Cytokine assay
Western blot analysis
Statistical analysis
Results
Beneficial effect of PG on DSS-induced body weight loss in mice
Beneficial effect of PG on DSS-induced colon shortening in mice
Regulatory effect of PG on DSS-induced DAI and colonic injury in mice
Regulatory effect of PG on DSS-induced inflammatory cytokine levels in colon tissue
Regulatory effect of PG on DSS-induced HIF-1α activation in colon tissue
Discussion
Introduction
Ulcerative colitis (UC), an inflammatory bowel disease (IBD), is a public health problem that can lead to an increased risk of colorectal cancer (Yashiro, 2014). The pathogenesis of UC is believed to result from the interaction of genetic, environmental and immune factors (Baumgart and Carding, 2007; Danese et al., 2004). The current drugs for UC include antibiotics and immunosuppressive drugs (Domenech, 2006; Sandborn and Targan, 2002). However, these available therapies are associated with adverse drug reactions in long-term use. Thus, herbal medicines have gained attention for developing treatments for UC.
UC progress is well-known to be associated with increased levels of inflammatory cytokines (Sands and Kaplan, 2007). Inflammatory cytokines including tumor necrosis factor (TNF)-α and interleukin (IL)-6 play crucial roles in the progression of colitis (Mueller, 2002). Clinical studies have shown that blood and colon tissues from patients with UC exhibited increased TNF-α and IL-6 levels (Park et al., 2017). Thus, therapeutic agents that reduce TNF-α and IL-6 levels may effectively ameliorate intestinal inflammation including UC.
Inflammation and hypoxia are hallmarks of inflammatory colon disorder. It was reported that hypoxia in mucosal tissue damage resulting from both acute and chronic inflammation (Taylor and Colgan, 2007). Hypoxia-inducible-factor (HIF)-1α is one of the transcription factors that links various inflammatory pathways (Van Uden et al., 2008). Clinical reports have verified HIF-1α upregulation in colon tissue from patients with UC (Scaldaferri et al., 2009). These findings suggest that HIF-1α is involed in controlling the levels of inflammatory mediator involved in the mucosal tissue damage in UC. Consequently, drugs that effectively regulate HIF-1α activation would be beneficial for the treatment of UC.
Platycodon grandiflorum (PG), belong to the Campanulaceae family, has been used in traditional herbal medicine for the treatment of inflammation, obesity and cancer (Choi and Lee, 2018; Kim et al., 2001, 2005; Park et al., 2007). However, whether PG regulates intestinal inflammation including UC in unknown. In the present study, to provide experimental evidence that PG might be a useful therapeutic for UC, we determined the improving effects and mechanism of PG in a dextran sulfate sodium (DSS)-induced colitis in mouse model. The specific goals were: (I) to evaluate the effect of PG on the main clinical features of colitis such as weight loss, colon length shortening and diarrhea and (II) to investigate the regulatory effect of PG on the expression levels of inflammatory cytokines as well as the activation of HIF-1α in colon tissues.
Materials and Methods
Animals and reagents
BALB/c mice (male, 7- weeks) were obtained from Hyo-chang Science (Daegu, Korea). The mice were maintained in a specific pathogen-free environment for at least one week to allow them to adapt to the environment. DSS was procured from MP Biomedical. Specific antibodies against HIF-1α andβ-actin were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Enhanced chemiluminesence (ECL) and bicinchoninic acid (BCA) assay kit were procured from Pierce Thermo Scientific (Rockford, IL, USA). ELISA assay kits for IL-6 and TNF-α were obtained from BD Biosciences (San Diego, CA, USA). Hematoxylin and Eosin (H&E) and other reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA).
Preparation of PG
We procured the dried root of PG from an oriental drug store, Daehak Oriental Drugstore (Iksan, Korea). The PG (100 g) was pulverized into a fine powder and decocted in 1 L of a distilled water for 3 h, and then concentrated under a vacuum. The extract was filtered and lyophilized (Ilshinbiobase, ILSHINBIOBASE-FD, Korea), then kept at 4℃ (yield, 11.8%).
Experimental colitis model construction and PG treatment
Colitis was induced in mice by administering 3% (w/v) DSS in drinking water ad libitum for 14 days. The mice were inspected daily for body weight loss, stool consistency and gross bleeding. The mice were randomized into 4 groups (n=7/ group): vehicle, DSS, DSS plus PG (10 ㎎/㎏) and DSS plus PG (100 ㎎/㎏). PG was orally administered once a day during DSS treatment.
Disease activity index
Intestinal disease activity was evaluated based on body weight loss, the presence of diarrhea accompanied by blood and colonic length shortening (Hendrickson et al., 2002). The degree of colitis was determined by disease activity index (DAI), a scoring system of three elements as described by Wirtz et al. (2017): degree of stool consistency (0-4), intestinal bleeding (0-4) and weight loss (0-4). The DAI values were measured by investigators blinded to the protocol.
Histological evaluation
Colon tissue was fixed in 10% neutral buffered formalin, dehydrated in a graded ethanol series, and embedded in paraffin. Tissue sections were cut by a rotary microtome, mounted on glass slides, and dried. The sections were stained with H&E using standard protocols.
Cytokine assay
Sections of the distal colon were excised, homogenized and extracted in protein extraction solution (Intron Biotechnology, Seoul, Korea). Following protein quantification using the BCA assay reagent, the levels in IL-6 and TNF-α of colon tissue were measured using ELISA kits according to the manufacturer’s protocols.
Western blot analysis
The distal colon were excised, homogenized and extraction by protein extraction solution. After protein quantification using BCA, the sample was mixed in sample buffer (2x), separated by electrophoresis and transferred to membranes. The membranes were blocked with skimmed milk and reacted with HIF-1α primary antibody. After washing, the membrane was reacted with secondary antibody for 1 h. After washing, protein bands were visualized using an ECL detection system.
Statistical analysis
The results are presented as the mean ± S.D. The statistical analyses were performed using an independent t-test and ANOVA with a Tukey post hoc test. P < 0.05 was considered significant.
Results
Beneficial effect of PG on DSS-induced body weight loss in mice
The DSS colitis model exhibited features of human UC (Eichele and Kharbanda, 2017). The main clinical symptoms of DSS-induced colitis (body weight loss, bloody diarrhea and colon shortening) were verified in this model. First, the improving effect of PG on the body weight loss in DSS induced colitis was determined. The results indicated that the body weight of the mice treated with DSS changed significantly compared to the control group. However, the PG group showed a significant attenuation of weight loss caused by DSS (Fig.1).
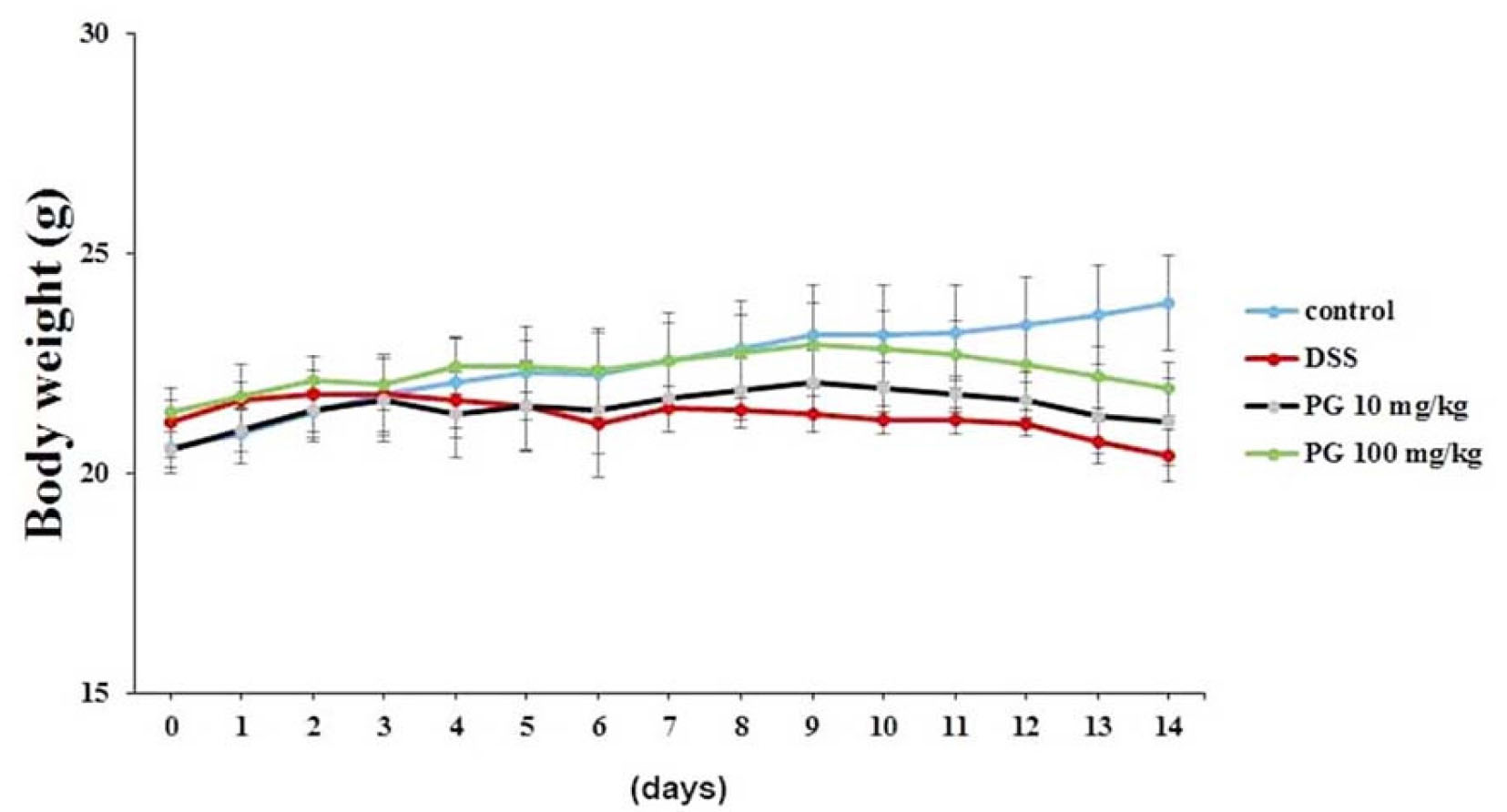
Fig. 1.
Ameliorative effect of PG on DSS-induced body weight loss in mice. Experimental colitis was induced in mice by administering 3% DSS in the drinking water for 14 days. PG (10 ㎎/㎏) or PG (100 ㎎/㎏) were orally administered. Body weight was determined at the same time on the experimental days. Data are presented as the mean ±S.D. (n = 7) and were analyzed by the Tukey post hoc test (#P < 0.05 versus control, *P < 0.05 versus DSS alone).
Beneficial effect of PG on DSS-induced colon shortening in mice
Generally, colon length shortening is a morphological parameter used to evaluate the degree of intestinal inflammation (Nunes et al., 2018). We investigated the regulatory effect of PG on colon length shortening in DSS-induced colitis. As shown in Fig. 2A, we observed that the colon length in the DSS-administered mice was significantly shorter (6.80±0.58 ㎝) than that of the controls (10.68±0.71 ㎝). However, PG (100 ㎎/㎏) treatment significantly alleviated the DSS effects on colon shortening (8.35±0.66 ㎝). The relative colon lengths are presented in Fig. 2B.
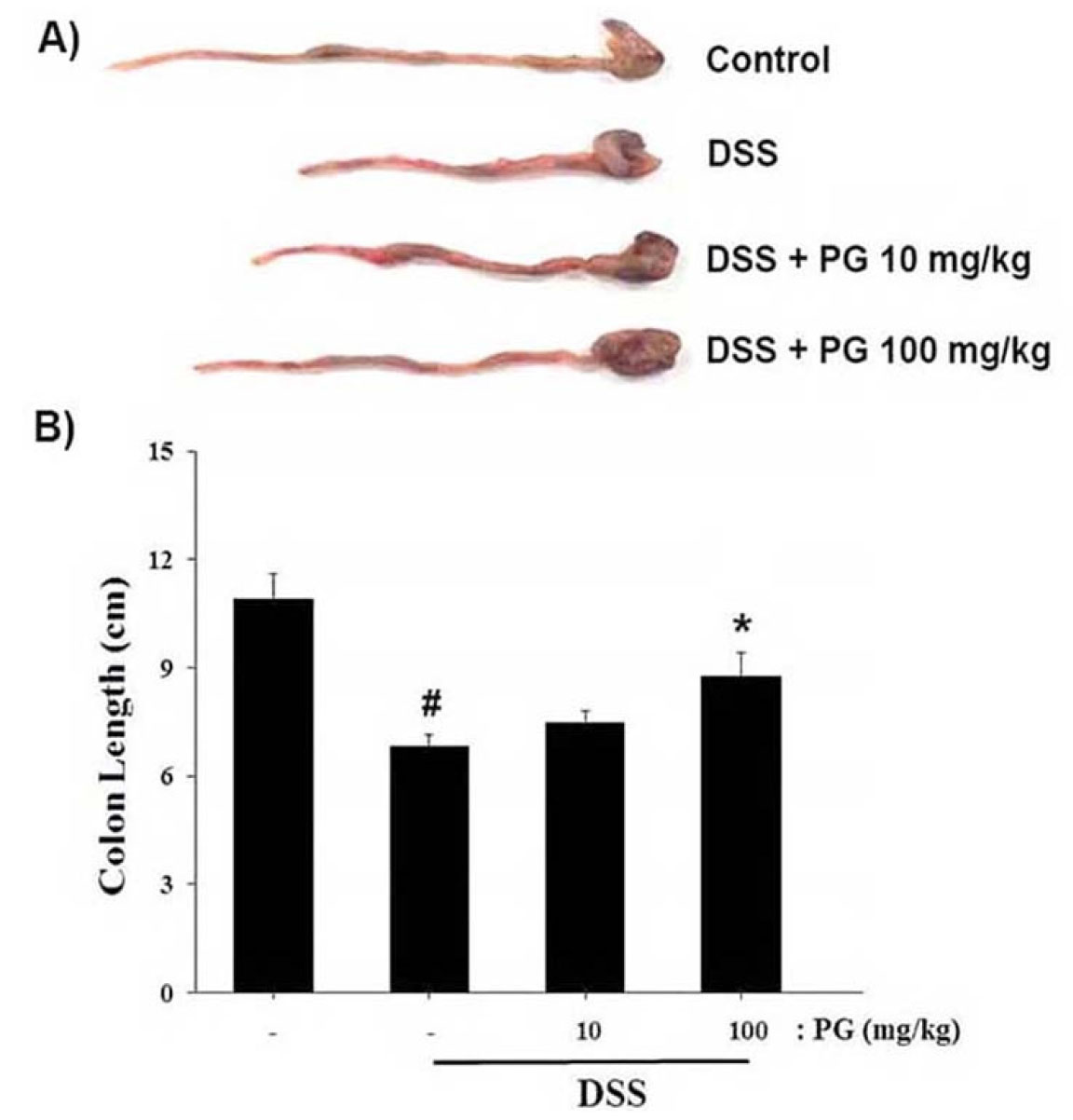
Fig. 2.
Ameliorative effect of PG on DSS-induced colon length shortening in mice. (A)Colons were removed on day 14 after DSS treatment, and the lengths were evaluated. (B) Relative colon length is presented. Data are presented as the mean ± S.D. (n = 7) and results were analyzed by the Tukey post hoc test (#P < 0.05 versus control, *P < 0.05 versus DSS alone).
Regulatory effect of PG on DSS-induced DAI and colonic injury in mice
DAI value is a parameter used to determine the severity of main colitis symptoms (Wirtz et al., 2017). To evaluate the improving effect of PG in DSS induced colitis, the DAI scores were measured. As shown in Fig. 3A, the results showed that DAI scores were significantly increased in the DSS-treated group, whereas it was attenuated by PG treatment. Additionally, histological analysis of the colons showed that DSS-induced colonic injury, such as epithelial crypt damage, was improved after PG treatment (Fig. 3B). The relative thickness of the colon muscle layer was presented in Fig. 3C.
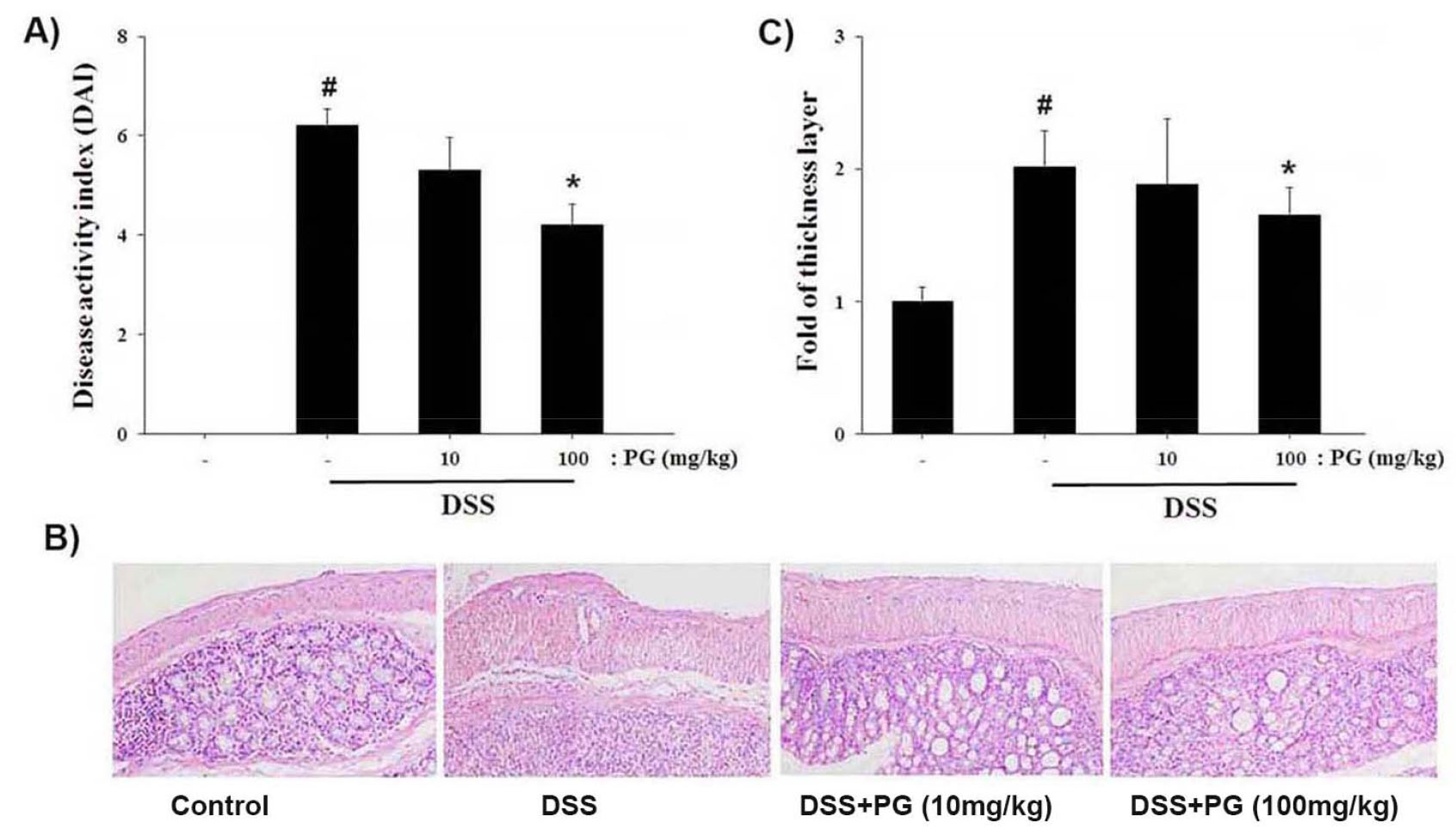
Fig. 3.
Ameliorative effect of PG on DSS-induced the DAI change and colonic injury in mice. (A) The DAI value was calculated as described in the Materials and Methods. (B) H&E staining was performed to evaluate the effect of PG on colon tissue damage. (C) The relative thickness of the muscle layer is presented. Data are presented as the mean ±S.D. (n = 7) and were analyzed by the Tukey post hoc test (#P < 0.05 versus control, *P < 0.05 versus DSS alone).
Regulatory effect of PG on DSS-induced inflammatory cytokine levels in colon tissue
Increase in inflammatory cytokine levels is associated with UC procession (Ogata and Hibi, 2003). In this study, we determined the regulatory effect of PG on inflammatory cytokines such as TNF-α and IL-6 levels in DSS-induced colon tissue. The result showed that DSS induced the expression of TNF-α and IL-6 compared to the controls. However, the enhanced levels of TNF-α and IL-6 expression in colon tissue caused by DSS were significantly attenuated by PG treatment (Fig. 4).
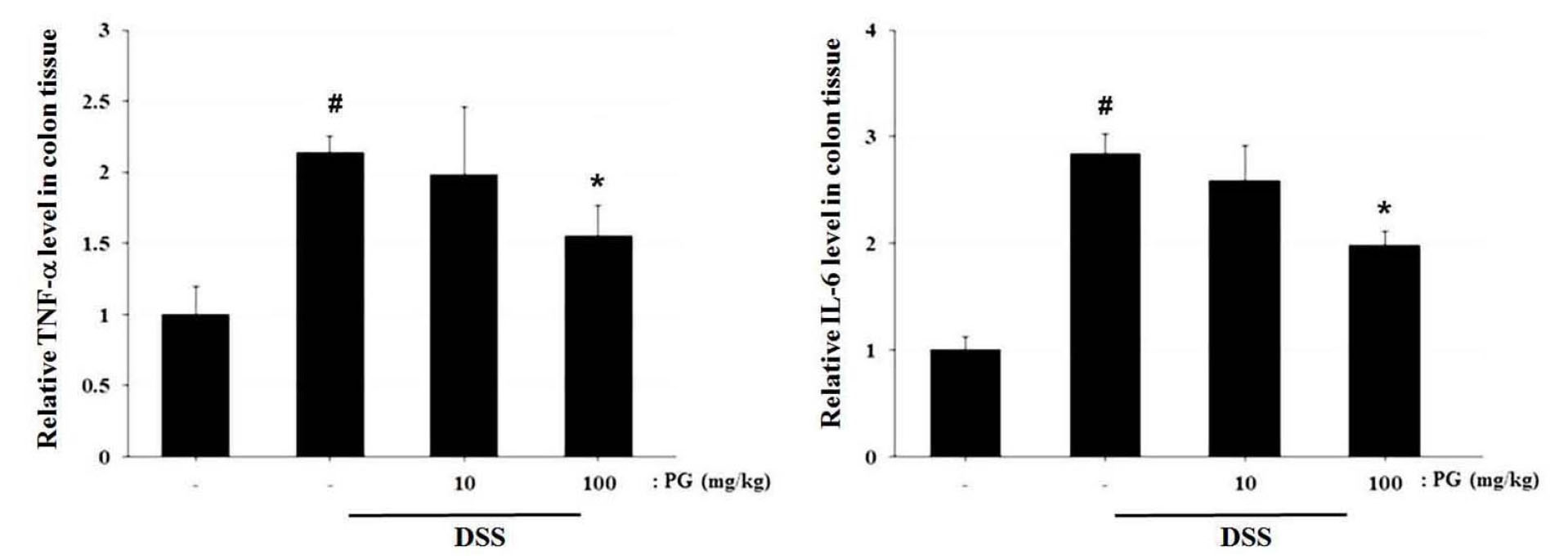
Fig. 4.
Ameliorative effect of PG on inflammatory cytokine levels in DSS-induced colitis tissue. At the end of the experiment, colon tissues were excised and homogenized. Following protein quantification, the levels of TNF-α and IL-6 in the colon tissue were assayed using ELISA assay kits. Data shown are expressed as the means ± S.D. (#P < 0.05 versus control, *P < 0.05 versus DSS alone).
Regulatory effect of PG on DSS-induced HIF-1α activation in colon tissue
HIF-1α activation is involved in UC procession (Karhausen et al., 2004). To evaluate the molecular mechanism of PG, we investigated whether PG regulated DSS-induced HIF- 1α activation in mice. As shown in Fig. 5A, we observed that HIF-1α activation was strongly enhanced in the colon tissues of DSS-treated mice, and treatment with PG suppressed DSS-induced HIF-1α activation. Fig. 5B showed the relative levels of HIF-1α observed by image analysis (Fig. 5B).
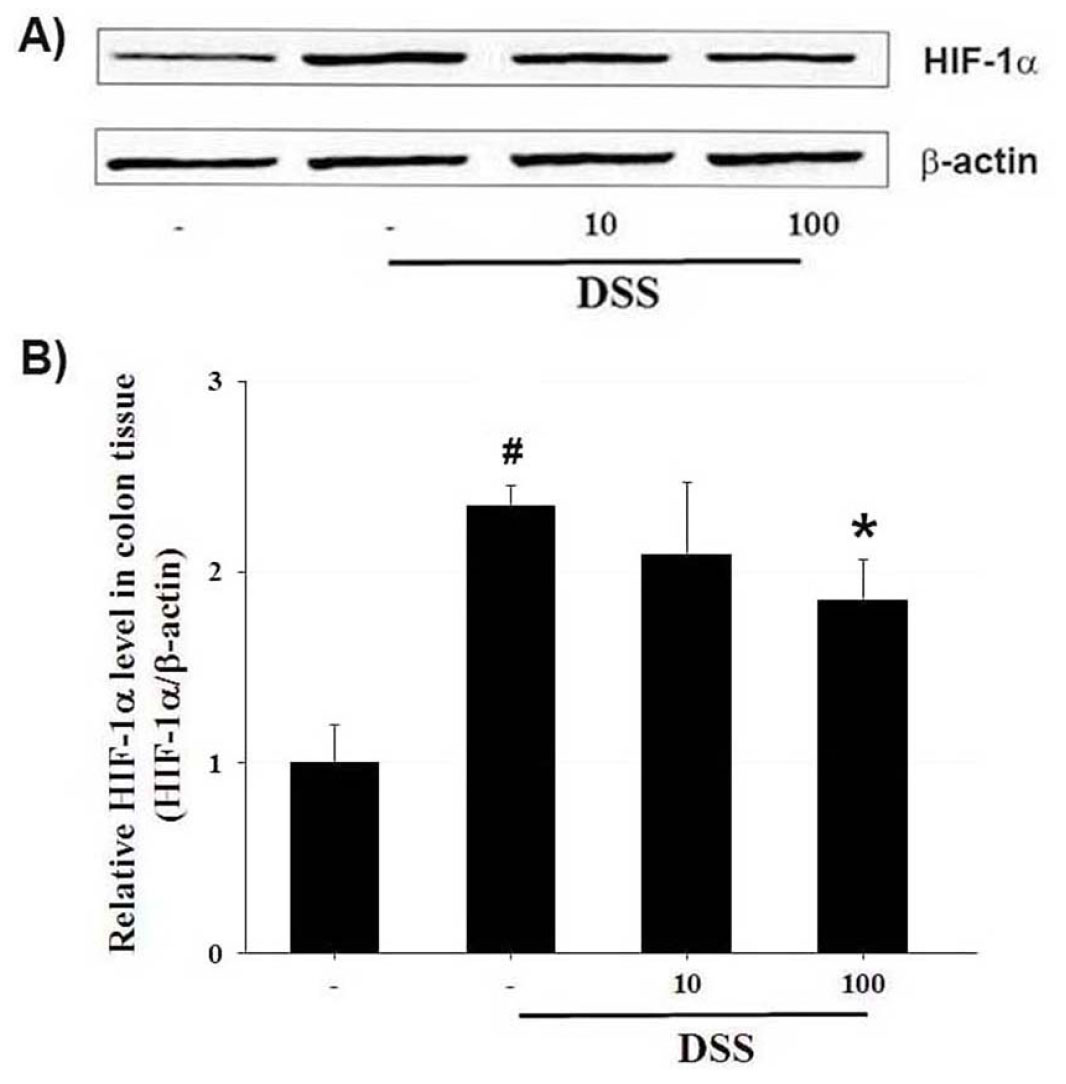
Fig. 5.
Ameliorative effect of PG on HIF- 1α activation in DSS-induced colon tissue. At the end of the experiment, the colon tissues were excised and homogenized. (A) The levels of HIF-1α in colonic tissue were assayed by Western blot analysis. (B) The relative HIF-1α level was measured using an image analyzer. Data shown are expressed as the means ± S.D. (#P < 0.05 versus control, *P < 0.05 versus DSS alone).
Discussion
Herbal medicines have received interest as complementary and alternative agents for various diseases. Although herbal medicines have been effectively employed, their exact mechanism is not completely elucidated. The findings of this study indicated that PG effectively attenuated the main clinical signs of colitis including colonic injury in mice. Additionally, we showed that the therapeutic mechanism of PG on intestinal inflammation arose from suppressing the inflammatory cytokine expression and HIF-1α activation in mice. The result suggests an important molecular mechanism by which PG ameliorates DSS-induced the intestinal inflammatory reaction.
UC is an inflammatory intestinal disorder that leads to an increased risk of colorectal cancer. The main clinical features of UC include colonic mucosal ulceration, weight loss, bloody diarrhea, anaemia, tenesmus, and abdominal pain. (Baumgart and Carding, 2007). Therapeutic treatments for UC include corticosteroids, biological therapy and immunosuppressive drugs. However, these have serious side effects in long-term use (Cottone et al., 2011). Therefore, new and effective alternative candidates for UC need to be developed. In this study, we determined the therapeutic effect of PG in DSS-induced colitis. The DSS-induced main clinical signs of UC are weight loss, bloody stools, colon length decrease and diarrhea (Ardizzone and Bianchi, 2005). The results showed that mice treated with PG suppressed body weight loss and colon shortening in mice with UC induced by DSS. The DAI values, which were scored using the main clinical signs of UC (body weight changes, diarrhea, and extent of rectal bleeding), were significantly higher than those of the controls. However, the DAI increases caused by DSS were markedly attenuated in the PG-treated groups. Additionally, histological analysis of the colon indicated that PG improved DSS-induced colonic injury, such as epithelial crypts and lamina damage. Our results demonstrated that PG exerted anti-colitis activity by regulating the main clinical symptoms of colitis and colonic injury caused by DSS in mice.
Accumulated experimental evidence has shown that inflammatory cytokines are associated with the pathogenesis of intestinal inflammation, including UC (Mueller, 2002). Clinical studies showed that inflammatory cytokines such as TNF-α and IL-6 have been reported to be elevated in blood and colon tissue of UC patients (Sands and Kaplan, 2007). Another previous study found that the overexpression of TNF-α and IL-6 was key contributor to UC carcinogenesis (Papadakis and Targan, 2000; Li et al., 2010). Therefore, we determined the ameliorative effect of PG the TNF-α and IL-6 expression levels in DSS-induced colon tissue. We confirmed that the PG inhibited the DSS-induced increase of TNF-α and IL-6 expression in colon tissue. Thus, we suggest that the anti-inflammatory activity of PG might be associated with the regulation of inflammatory cytokines expression in DSS-induced colon tissue.
Hypoxia and inflammation are hallmarks of intestinal inflammatory disorders. Other previous studies have indicated hypoxia in the mucosal tissue damage resulting from both acute and chronic inflammation. Numerous reports have shown that HIF-1α activation was involved in intestinal inflammatory reaction and pathogenesis of UC (Glover and Colgan, 2011). HIF-1α activation and the subsequent increase in inflammatory mediator are important in UC pathogenesis (Van Uden et al., 2008). Additionally, other studies showed that HIF-1α played an integral role in the disruption of epithelial barrier function (Furuta et al., 2001). Therefore, attenuation of HIF- 1α activation have been suggested as a novel strategy for UC. We evaluated whether PG regulated the DSS-induced HIF-1α activation to determine the molecular mechanism of PG action in the UC pathology. Our results revealed that DSS-induced HIF-1α activation in colitis tissue was strongly inhibited by PG treatment. These results indicated that PG attenuatesd intestinal inflammation by blocking HIF-1α activation in colitis.
Collectively, the results indicate that PG exerted anti-colitis activity by relieving the main clinical symptoms and colonic damage in DSS-induced colitis. Moreover, we demonstrated that the anti-inflammatory effect and mechanism of PG could be attributed to the suppression of HIF-1α activation and the subsequent down-regulation of inflammatory cytokines levels in colon tissue. Our results provide experimental evidence that PG might be an efficacious therapeutic candidate for treating intestinal inflammatory diseases including UC.




