Introduction
Materials and Methods
Study Site and Plant Materials
Phenotypic Characterization
Statistical Analysis
Results
Quantitative Traits
Qualitative Traits
Correlation Among Quantitative Traits
Principal Component Analysis (PCA)
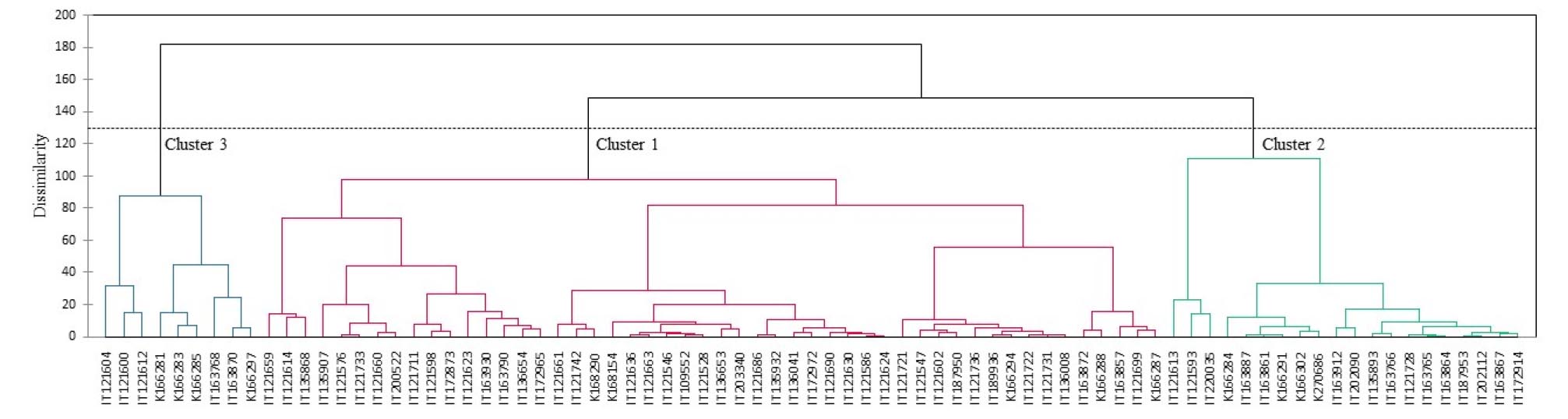
Cluster Analysis
Discussion
Introduction
Onions (Allium cepa L.) are one of the most widely grown vegetables worldwide (FAOSTAT, 2022). Southwest Asia is the gene center for onions (Bagci et al., 2021; Gokce, 2022), and the Mediterranean region is the secondary center of origin (Castell and Portas, 1994). The economic importance of onion is based on their culinary use, nutritional value, and health benefits (Benkeblia, 2005). Despite the importance of onions, research on their genetic improvement is limited compared to that of other major vegetables (McCallum et al., 2008). One reason might be improper genetic resource identification (Cross, 1998). Normally, variety improvement depends on the genetic variability of the germplasm (Mallor et al., 2014; Mallor Gimenez et al., 2011; Manjunathagowda, 2022). Exploration of new onion genetic resources and their characterization and exploitation have been hampered by out-breeding and biennial growth habits (McCallum, 2007).
Short and intermediate-day onion cultivars were first introduced in the southern parts of Korea in the 1950s and are currently predominantly grown in Korea. Intermediate-day onion cultivars are widely grown in southern South Korea. In 2022, onions were cultivated across 17,655 ㏊ with a total production of 1.2 million tonnes, but production is decreasing (Korea Agro-Fisheries and Food Trade Corporation, 2022). In Korea, the low seed self-sufficiency rate (~30%) and high seedling costs (~18% of the production costs) have increased the financial burden on onion growers. By 2022, ~1, 800 onion cultivars were registered for production, import, and distribution and most of the cultivated varieties were F1 hybrids (Korea Seed Association, 2022). Most F1 hybrids are imported from Japanese seed companies, although the origin of these cultivars has not yet been identified (Lee et al., 2020). Currently, farmers typically grow onion cultivars that are recommended by seed distributors or obtained from neighboring onion growers rather than from formalized seed distribution schemes. F1 hybrid varieties have a narrow genetic base and can replace old domestic varieties that contain potential adaptive genes. Therefore, the evaluation of novel onion genetic resources is important for selecting better traits and enhancing the competitiveness of domestic cultivars in Korea.
Variety improvement and hybrid breeding depend on the genetic variability and diversity of the germplasm (Manjunathagowda, 2022; Ojuederie et al., 2014). Knowledge of the genetic variation present in the gene pool of onions is important for breeders, and helps them to select genotypes based on their distinctiveness for variety development (Dangi et al., 2018; Manjunathagowda and Selvakumar, 2021). A selection of genetically diverse parents is necessary for successful recombination in breeding programs (Akter et al., 2015).
Plant genetic materials adapt to a region over time and substantial changes occur in their genetic structures because of environmental conditions. Therefore, studying morphological variation and phenotypic diversity is important for breeding programs (Yuguda et al., 2017). Many researchers (Azimi et al., 2020; Bagci et al., 2022; Lee et al., 1996; Mallor Gimenez et al., 2011; Mousavizadeh et al., 2006; Sunil et al., 2014) in different countries have performed studies to collect, characterize, and determine the phenotypic diversity of onion genetic resources. They reported high levels of morphological variation in the bolting rate, bulb diameter, bulb shape index, bulb weight, bulb shape and base color of the dry skin in onion accessions.
The Allium Vegetable Research Institute (AVRI) has mandated research on breeding new cultivars of onion, garlic, and green onion as well as increasing the self-sufficiency rate of domestic cultivars. Both the AVRI and the National Agrobiodiversity Center (NAC) have conducted research on the regeneration and trait identification of temporary onion genetic resources. Furthermore, these germplasms must be characterized to remove redundant accessions and to identify genetically distinct accessions for onion breeding programs. This study will help in generating information for creating a database for this purpose. Morphological characterization is the first step in the description and classification of genetic resources (Smith and Smith, 1989). The analysis of germplasm morphological traits provides useful information to breeders for preliminary evaluation and determination of the diversity status (Asare et al., 2011). Previous researchers (Lee et al., 1996; 2020) evaluated onion cultivars and their classification using multivariate analysis. Multivariate analysis was also used to study morphological diversity and other relationships among garlic germplasm (Lee et al., 2021). For selective breeding, knowledge of the association between the bulb yield and characteristics contributing to yield is crucial (Arya et al., 2017). Knowledge of the correlations among different characteristics and their relative contributions to the yield is useful for selecting multiple traits (Arya et al., 2017). Principal component analysis (PCA), a multivariate method, can be used to reveal important traits for genotype differentiation. Using PCA, breeders can understand the impact and associations among different traits of genotypes (Kovacic, 1994). Furthermore, cluster analysis can be used to select potential genotypes for breeding and commercial onion production. Characterizing the onion germplasm is essential for developing new cultivars using existing genetic variability. Therefore, the aim of this study was to identify the phenotypic traits of temporary genetic resources and find the best accessions for their use in the development of onion breeding programs.
Materials and Methods
Study Site and Plant Materials
This study was conducted at the AVRI research field, Muan-Gun (34° 59' 25.63 44" N, 126° 28' 54.0696" E, 31.48 meters above sea level), Jeollanam‐Do, Republic of Korea from September 2020 to June 2021. The study site is characterized by a humid subtropical climate; it receives 1,000-1,800 ㎜ of precipitation annually (Korea Meteorolgical Administration, 2021). The soil type at the site is clay, with a pH value ranging from 5.5 to 7.2 (http://soil.rda.go.kr). The seeds of 79 onions (Allium cepa L.) were obtained from the National Agrobiodiversity Center of Jeonju, Korea (Appendix 1). Plug trays containing 338 cells (26 × 13 cells; FAMWIN, Bumong Co. Ltd., Jeong‐eup, Korea) were filled with a commercial growing medium (Plantworld, Nongwoobio Co. Ltd., Suwon, Korea) and 50 seeds from each germplasm were sown in September 9, 2020. Cattle manure compost (20 ㎎ ㏊-1) and pre-plant fertilizers (80 ㎏ ㏊-1 N as urea, 33.6 ㎏ ㏊-1 of P2O5 as fused phosphate, and 58 ㎏ ㏊-1 K2O as potassium sulphate) were applied to the plots according to the fertilizer recommendation of the Rural Development Administration (RDA, 2010). Terbufos and fosthiazate were applied as pesticides to reduce maggot and nematode damage. The planting bed was prepared at a center-to-center distance of 1.50 m with a 1.0 m bed width and 0.20 m height. Alchlor (43.7%) and pendimethalin (31.7%) were sprayed on the bed. Subsequently, on November 11, 2020, black plastic was mulched on the bed, and an average of 25-30 seedlings of each accession were transplanted into the plot, each with a plant geometry of 20 ㎝ × 10 ㎝. We followed the recommended cultivation practices for onions outlined by the RDA.
Phenotypic Characterization
Nine quantitative traits (bolting rate, plant height, polar and equatorial bulb diameters, bulb shape index (BSI), bulb weight, degree of splitting into bulblets, bulb neck width, and bulb size) and seven qualitative traits (leaf color, foliage attitude, foliage waxiness, bulb shape at root end, base color of dry skin in bulb, bulb shape, and color of the epidermis of fleshy scales at the bulb) were selected using modified descriptors of the International Union for the Protection of New Varieties of Plants (UPOV, 2008), and reference descriptors of Allium spp. (IPGRI, 2001). Quantitative measurements for each trait were performed on ten random samples and averaged. Qualitative characteristics were recorded from plant and bulb observations in the field. The detailed quantitative and qualitative traits and measurement methods are provided in Appendix 2.
Statistical Analysis
Quantitative and qualitative data were subjected to descriptive statistics using SPSS Statistics (version 20.0; SPSS Inc., Chicago, IL, USA). Frequency statistics were also used to analyze 16 phenotypic traits of the 79 accessions. Pearson correlation coefficients were calculated for the nine quantitative traits, and principal components analysis (PCA) and agglomerative hierarchical cluster analysis were performed for all 16 phenotypic traits using the XLSTAT software (Addinsoft, Paris, France).
Results
Quantitative Traits
Descriptive statistics for nine quantitative traits of the 79 accessions and their variations are presented in Table 1. The full dataset for the nine quantitative traits is presented in Appendix 3. The bolting rate varied from 0.0% to 80.0%, with an average value of 7.5%. The plant height ranged from 37.9 to 91.2 ㎝, with an average of 68.6 ㎝. The average polar bulb diameter was 6.0 ㎝, ranging from 3.4 to 9.0 ㎝. The equatorial bulb diameter ranged from 3.7 to 9.5 ㎝, with an average of 6.9 ㎝. The BSI varied from 0.5 to 1.4, with an average value of 0.9. The maximum and minimum bulb weights were 368.4 and 38.5 g, respectively, with an average of 173.2 g. The degree of splitting into bulblets ranged from 0.0% to 100%, with an average of 28.0%. The coefficient of variation ranged from 16.3% (low) for the plant height to 230.1% (high) for the bolting rate, followed by the degree of splitting into bulblets (141.7%) and bulb weight (50.7%). Bulb width of neck and bulb size showed 24.0% and 33.9% variation, respectively.
Table 1.
Phenotypic variation of quantitative traits in the 79 onion germplasms evaluated at Muan-Gun, Jeolla‐Do, Korea, from 2020-2021
| Quantitative traits | Maximum | Minimum | Mean | SD (±)z | CV (%)y |
| Bolting rate (%) | 80.0 | 0.0 | 7.5 | 17.3 | 230.1 |
| Plant height (㎝) | 92.1 | 37.9 | 68.6 | 11.2 | 16.3 |
| Polar bulb diameter (㎝) | 9.0 | 3.4 | 6.0 | 1.4 | 23.2 |
| Equatorial bulb diameter (㎝) | 9.5 | 3.7 | 6.9 | 1.5 | 21.5 |
| Bulb shape index | 1.4 | 0.5 | 0.9 | 0.2 | 19.3 |
| Bulb weight (g) | 368.4 | 38.5 | 173.2 | 87.9 | 50.7 |
| Degree of splitting into bulblets (%) | 100.0 | 0.0 | 28.0 | 39.7 | 141.7 |
| Bulb neck width (3-7) | 7.0 | 3.0 | 6.1 | 1.5 | 24.0 |
| Bulb size (3-7) | 7.0 | 3.0 | 4.9 | 1.7 | 33.9 |
The frequency distribution of the nine quantitative traits among the onion accessions is shown in Fig. 1. Among the 79 accessions, 61 did not bolt (Fig. 1A). The plant height ranging from 61.0‒80.0 ㎝ had the highest number of accessions (50; Fig. 1B). The bulb polar diameters ranging from 3.6 to 7.0 ㎝ contained the highest number of accessions (55; Fig. 1C). The bulb equatorial diameter ranging from 5.6 to 7.0 ㎝ had the highest number of accessions (28), followed by 22 accessions within the range of 7.1 to 8.5 ㎝ (Fig. 1D). The BSI ranging from 0.8 to 0.9 contained the highest number of accessions (33), followed by 26 accessions within were in the range of 0.10 to 0.11 ㎝ (Fig. 1E). The bulb weight ranging from 101.0-150.0 g had the highest number of accessions (19), followed by the range of 151.0 to 200.0 g (16 accessions; Fig. 1F). Regarding the degree of splitting into bulblets, the highest number of accessions (42) were observed for non-splitting types. The degree of splitting into bulb-lets ranged from 81.0% to 100.0% in 15 accessions (Fig. 1G) and the highest number of accessions (51) had a large bulb neck width (Fig. 1H). The highest number of accessions (29) had a small bulb size, whereas nine accessions had a large bulb size (Fig. 1I).
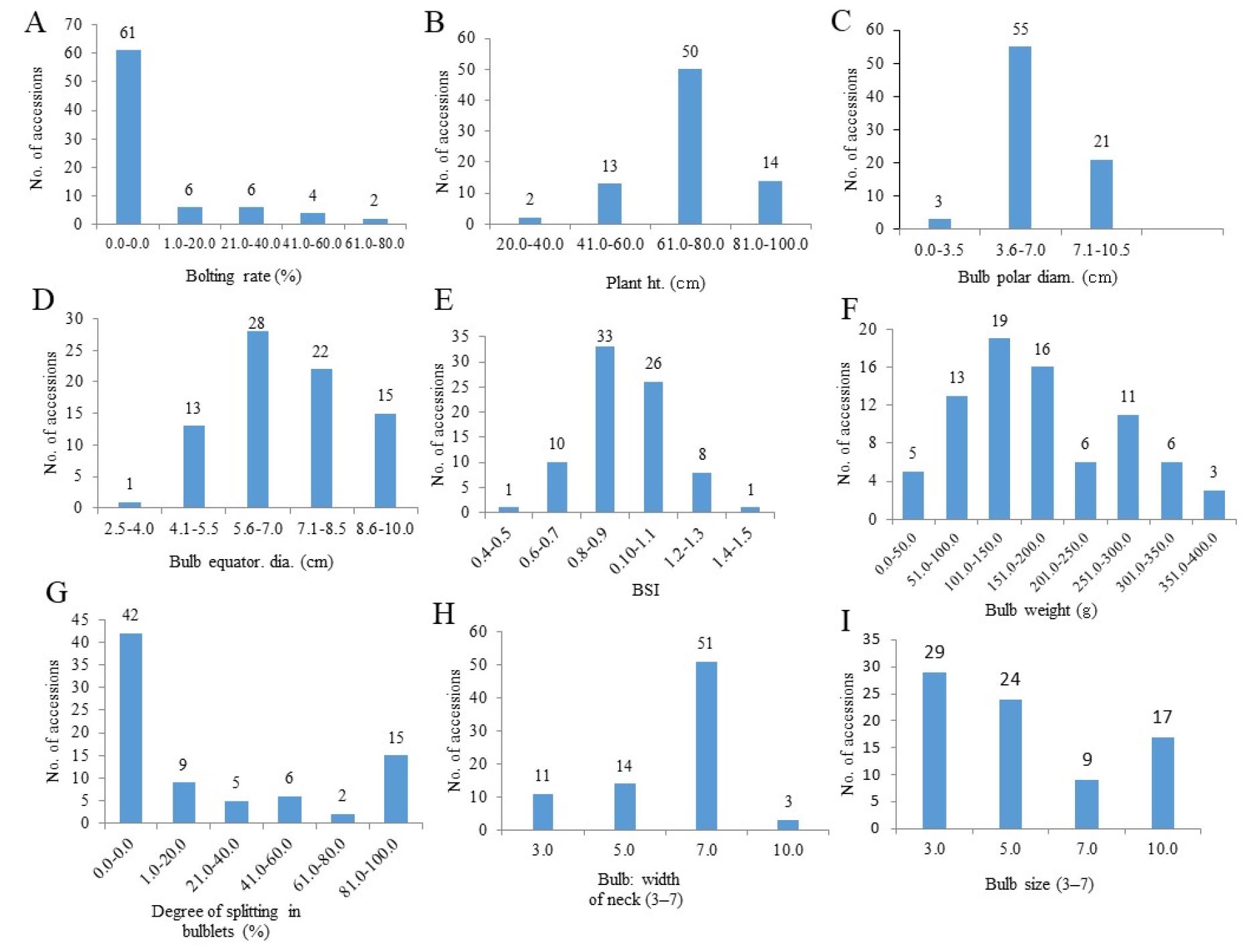
Fig. 1.
Frequency distribution of nine quantitative traits among the 79 onion germplasms. A, bolting rate (%); B, plant height (㎝); C, bulb polar diameter (㎝); D, bulb equatorial diameter (㎝); E, bulb shape index (BSI); E, bulb weight (g); F, degree of splitting in bulblets (%); H, bulb neck width (3-7): 3, small; 5; medium; 7; large; 10, mix; I, bulb size (3-7): 3; small; 5; medium; 7; large; 10; mix.
Qualitative Traits
The assessment of the seven qualitative characteristics of 79 accessions is presented in Appendix 4. The seven qualitative traits exhibited a wide variation among the accessions (Fig. 2). Green leaves were predominantly observed among the accessions, although they varied in color (Fig. 2A). The foliage attitude varied from erect to semi-erect to mixed (Fig. 2B). A broad variability was observed for the foliage waxiness (Fig. 2C), bulb shape at the root end (Fig. 2D), and color of the dry skin at the base of the bulb (Fig. 2E). The circular bulb shape had the highest number of accessions, followed by the oval shape (Fig. 2F). Most accessions for epidermal flesh scales were white, followed by red (Fig. 2G).
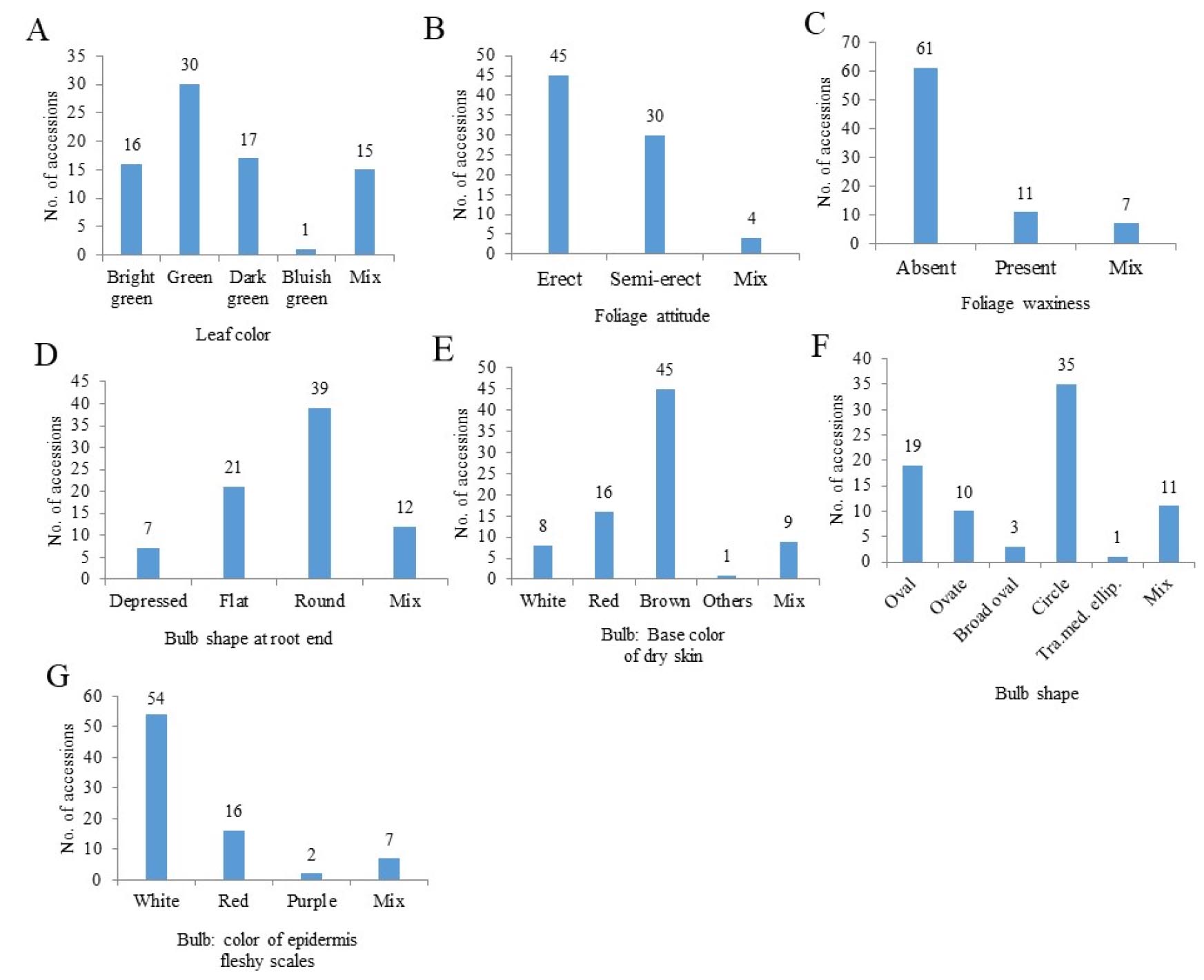
Fig. 2.
Frequency distribution of seven qualitative traits among 79 onion germplasms. Tra. med. ellip., transverse medium elliptic; A, leaf color; B, foliage attitude; C, foliage waxiness; D, bulb shape at root end; E, bulb: base color of dry skin; F, bulb shape; G, bulb: color of epidermis fleshy scales.
Correlation Among Quantitative Traits
Pearson correlation coefficients for the nine quantitative traits are presented in Table 2. Plant height was significantly (p < 0.01) positively correlated with the bulb polar diameter, bulb equatorial diameter, and bulb weight. Bulb polar diameter was significantly positively correlated with bulb equatorial diameter, BSI, and bulb weight, but negatively correlated with degree of splitting into bulblets (p < 0.01). Bulb equatorial diameter was significantly positively correlated with bulb weight and bulb size (both p < 0.01) but negatively correlated with degree of splitting into bulblets (p < 0.01). The bulb weight was significantly negatively correlated with the degree of splitting into bulblets, but significantly positively correlated with the bulb size (both p < 0.01).
Table 2.
Correlation coefficients among the nine quantitative traits of the 79 onion germplasms evaluated at Muan‐Gun, Jeolla‐Do, Korea, from 2020-2021
Principal Component Analysis (PCA)
PCA was used to calculate the correlations among the 16 phenotypic traits of the 79 onion accessions. PCA showed that the first six components (PC1 to PC6) accounted for almost three-quarters of the total variation (Table 3). PC1 accounted for approximately one-quarter of the phenotypic variance, with bulb weight, bulb equatorial diameter, polar diameter, and plant height (0.34) being the main contributors. Bulb color of the epidermal fleshy scales and the color of the bulb base dry skin, contributed to phenotypic variance in PC2 (Appendix 5). The third PC explained just over 11.0% of the total variation and was mainly associated with the BSI, bulb polar diameter, bolting rate, and foliage waxiness. Similarly, in PC4, the foliage attitude, bolting rate bulb neck width, foliage waxiness, and leaf color contributed close to 9.0% of the phenotypic variance. PC5 contributed to 7.0% of the phenotypic variance and was associated with the bulb shape, bulb shape at root end, and leaf color. PC6 accounted for 7.0% of the total variation and was associated with bulb neck width and plant height.
Table 3.
Eigenvector and eigenvalues generated by PCA applied to the 16 morphological characteristics of the 79 onion germplasms evaluated at Muan‐Gun, Jeolla‐Do, Korea, from 2020-2021
Cluster Analysis
A dendrogram of genetic distances based on morphological characteristics was constructed using agglomerative hierarchical clustering. Overall, three clusters were obtained from the 79 onion accessions. Clusters 1, 2, and 3 contain 50, 20, and 9 accessions, respectively (Fig. 3). Cluster 1 was further separated into three subgroups and clusters 2 and 3 each separated into two subgroups.
The means and standard deviations of the quantitative traits in the three clusters are presented in Table 4. Most accessions were grouped in cluster 1, which showed a lower plant height, bulb polar diameter, bulb equatorial diameter, and bulb weight than that of clusters 2 and 3. The bulb polar diameter, bulb equatorial diameter, and bulb weight were higher in cluster 2 than in clusters 1 and 3. Accessions in cluster 2 showed lower bolting rate than those in clusters 1 and 3. The degree of splitting into bulblets was higher in cluster 1 than in clusters 2 and 3. Clusters 2 and 3 did not contain small bulb neck widths. The bulb size of cluster 1 accessions consisted of small, medium, large, and mixed types, whereas cluster 2 accessions contained medium and large onions, and cluster 3 accessions had small‐to‐medium bulb sizes.
Table 4.
Means and standard deviations of quantitative traits used for the classification of three onion clusters evaluated at Muan‐Gun, Jeolla‐Do, Korea, from 2020-2021
| Traits | Cluster 1 | Cluster 2 | Cluster 3 | |||
| Mean | SDz | Mean | SD | Mean | SD | |
| ny | 50 | 20 | 9 | |||
| Bolting rate (%) | 7.4 | 17.0 | 3.8 | 11.3 | 16.7 | 26.9 |
| Plant height (㎝) | 64.7 | 10.2 | 74.8 | 7.6 | 77.3 | 13.5 |
| Polar bulb diameter (㎝) | 5.6 | 1.3 | 7.1 | 0.9 | 6.1 | 1.4 |
| Equatorial bulb diameter (㎝) | 6.4 | 1.4 | 8.4 | 0.6 | 6.9 | 1.5 |
| BSI | 0.9 | 0.2 | 0.9 | 0.1 | 0.9 | 0.2 |
| Bulb weight (g) | 135.5 | 67.3 | 268.1 | 52.6 | 171.4 | 97.7 |
| Degree of splitting into bulblets (%) | 38.8 | 43.3 | 0.8 | 3.4 | 28.9 | 35.8 |
| Bulb neck width (3-7) | 3, 5, 7 | - | 5, 7, 10 | - | 5, 7 | - |
| Bulb size (3-7) | 3, 5, 7, 10 | - | 5, 7, 10 | - | 3, 5, 10 | - |
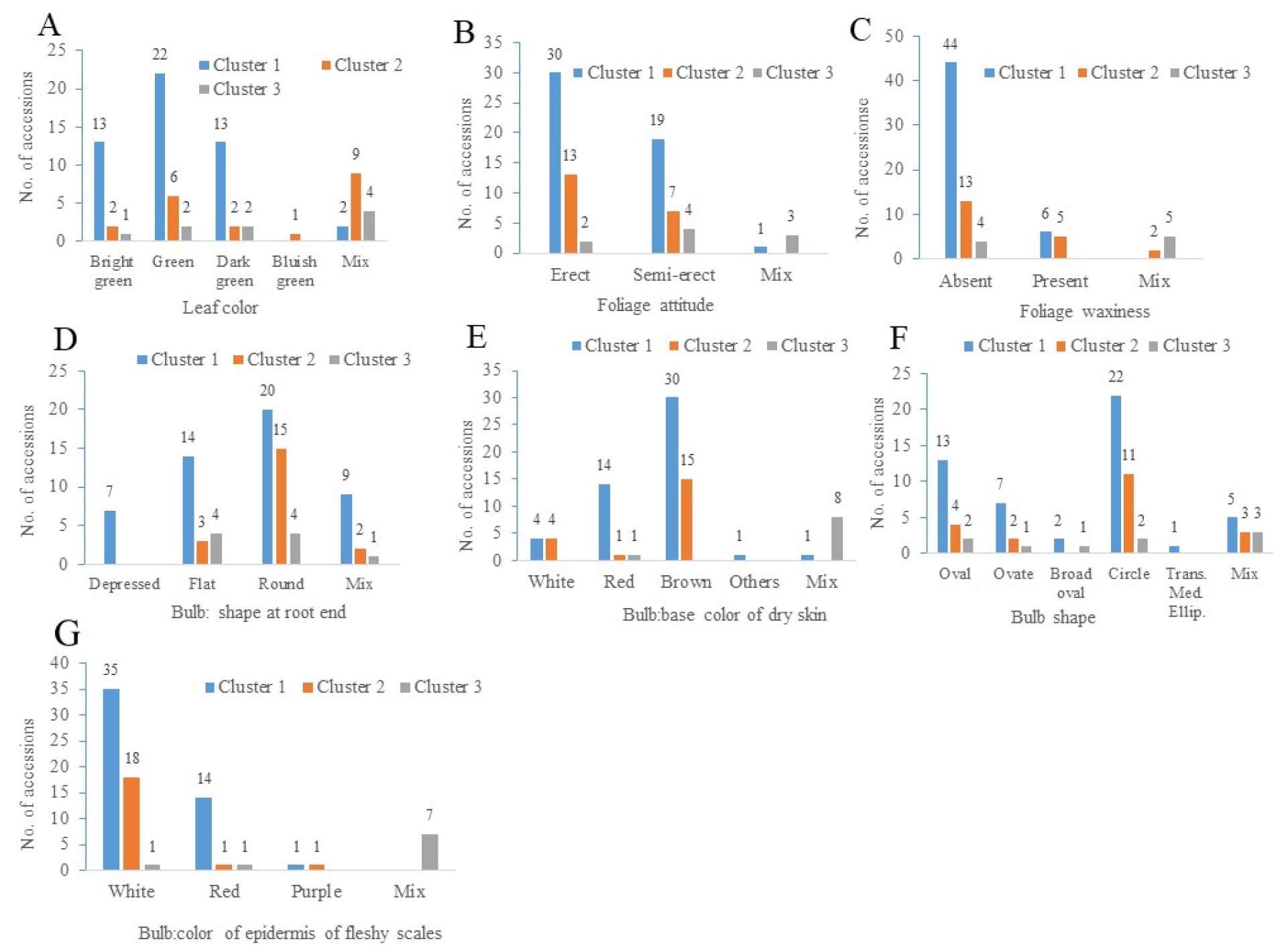
A summary of the qualitative characteristics of the three clusters is given in Fig. 4. Cluster 1 contained the highest number of accessions with green leaf color, whereas clusters 2 and 3 contained the highest number of accessions with mixed leaf color (Fig. 4A). Clusters 1 had the most accessions with erect and semi-erect foliage attitude, followed by Cluster 2 (Fig. 4B). Clusters 1 and 2 had the highest number of accessions that contained no foliage waxiness (Fig. 4C). Clusters 1 and 2 contained the highest number of accessions with round bulb shapes at the root end (Fig. 4D). Similarly, the color of the bulb base dry skin was predominantly brown in clusters 1 and 2, but cluster 3 contained the highest number of accessions with a mixed color (Fig. 4E). Clusters 1 and 2 had the highest number of accessions with circular bulb shapes (Fig. 4F). The white epidermis of fleshy scales was predominant in clusters 1 and 2 accessions, but a mixed color of the epidermis of fleshy scales was predominant in cluster 3 accessions (Fig. 4G).
Discussion
In this study, marked phenotypic trait variations were identified among the germplasms, which could provide accessions for use in the development of onion breeding programs. Variations in phenotypic traits are of paramount importance in onion breeding. Owing to the high variability in the bolting rate, degree of splitting into bulblets, bulb weight, bulb polar and equatorial diameters in the onion germplasms of this study, the selection of superior genotypes based on these traits could be effective. Arya et al. (2017) also reported variations in the plant height, polar and equatorial bulb diameters, BSI, and bulb weight in 26 onion germplasms.
Bolting is an undesirable trait in the production of onion bulbs. In our study, the bolting rate exhibited a high degree of variation, followed by the degree of splitting into bulblets, and then bulb weight. Monpara et al. (2005) also reported a high variation (133.9%) in the bolting traits when evaluating 106 onion germplasms. Hanci and Gokce (2016) reported a 68.1% and 61.6% variation in the degree of splitting into bulblets and bulb weight, respectively. The high coefficients of variation obtained for the bolting rate, degree of splitting into bulblets, and bulb weight in this study are similar to the findings of previous studies (Ghetia and Singh, 2000; Hanci and Gokce, 2016; Monpara et al., 2005). Genotype selection is a well-known approach for avoiding bolting traits. Selecting genotypes that are resistant to bolting is a major breeding objective for onions (Hu et al., 2003).
Bulb shape, weight, and size are commercially important traits. The results of this study showed a wide variation in these traits. The bulb shape also affects consumer preferences and varies across different markets. Notable differences in the bulb shape among onion cultivars, which are important to consumers, have been reported by Grant and Carter (1991). In this study, marked variation was observed in the base color of dry skin among onion accessions. Phenotypic diversity in the onion germplasm for the base color of dry skin has also been reported (Azimi et al., 2020; Gvozdanović et al., 2013). In this study, circular bulb‐ shaped accessions were identified, which might be important for onion breeding. Azimi et al. (2020) also reported differences in the bulb shape. Plant morphology can be used as a genetic marker to characterize onion germplasms (Ahmed et al., 2013). Morphological traits are useful for the protection, registration, and patenting of varieties. Accordingly, the variation in qualitative traits observed in the study could be used to identify accessions. Phenotypic traits are also useful for characterizing other crops such as lettuce germplasm (Sung et al., 2016).
The positive association of plant height, bulb polar diameter, bulb equatorial diameter with bulb weight suggests that these traits are yield determining factors and could be directly selected. Manjunathagowda (2022) also reported a strong positive correlation among bulb weight, plant height, and bulb polar and equatorial diameters which concurs with our findings. Other studies have found bulb weight positively and significantly (p < 0.01) correlated with the equatorial and polar bulb diameters (Dhotre et al., 2010; Patil et al., 1990). Conversely, bulb weight negatively correlated with the degree of splitting into bulblets, which indicated that selecting genotypes with a high‐degree of splitting results in a low bulb weight. Raghuwanshi et al. (2016) reported that plant height is a yield‐determining trait. In the present study, we found that the quantitative traits contribute more to diversity than qualitative traits. Hanci and Gokce (2015) reported that the nine PCs contributed to 71.84% of the variability (high eigenvalues > 1), and that the bulb weight and pseudostem diameter positively contributed to PC1. However, in this study, six PCs contributed to most of the variability (high eigenvalues > 1). The bulb weight, bulb equatorial and polar diameters, and plant height positively contributed to PC1. Dangi et al. (2018) reported that five principal components PC1-PC5 contributed to 78.5% of the total variation in short‐day onion genotypes and they identified that leaf diameter, bulb diameter, and number of leaves are the major contributors to PC1. The results of our study show that bulb weight, bulb equatorial and polar diameters, plant height, degree of splitting into bulblets, and bulb size are the most useful traits for PC1, followed by bulb color of the epidermal fleshy scales and bulb base color of the dry skin (PC2). Mousavizadeh et al. (2006) stated that the axes of the first three PCs had 83.42% of the total phenotypic diversity in Iranian onion landraces and that bulb dry weight, diameter, and bulb weight were the major contributors to PC1. Arya et al. (2017) observed that three PCs accounted for 95.61% of the variation. The high positive loading related to the bulb weight in PC1 in this study agrees with the results of Arya et al. (2017), where the major contributing traits were the bulb weight, bulb equatorial and polar diameters, and plant height in the positive direction, and the degree of splitting into bulblets (in the negative direction). In contrast, the traits contributing the least were BSI, foliage attitude, bulb color of the epidermal fleshy scales, bulb neck width and bolting rate. Most accessions were clustered together, indicating morphological similarities, whereas a few accessions showed diversity. Differences in traits, such as the bolting rate, degree of splitting into bulblets, bulb weight, bulb size, and color of the epidermal fleshy scales might have created the variations in these accessions. Based on morphological traits, Lee et al. (1996) classified 60 onion varieties into four groups, most of which were not associated with their geographical origins. This study classified 79 onion germplasms into three clusters based on 16 morphological traits. Manjunathagowda (2022) also reported five clusters from 62 Indian onion accessions, and Arya et al. (2017) classified 26 onion accessions into four clusters. Prior studies have reported that clustering does not signify geographical origin, which is consistent with our findings.
In this study, marked phenotypic trait variations were identified among the 79 onion germplasms. These variations could provide information to breeders and gene bank curators. The phenotypic traits, including a low bolting rate, low degree of splitting into bulblets, and high bulb weight, along with circular bulb-shaped onion accessions could be important for improving onion breeding. The significant correlation between plant and bulb traits in onion accessions could improve their genetics. The strong positive values in the principal components can aid in the precise selection of onion accessions. In particular, cluster 2 onion accessions can be effectively utilized in variety improvement. The history and collection area for each accession may help to form the clusters, and examine their relationship with morphological characteristics. However, the present study reveals little explanation for the relationships between closely related or geographically distant accessions. Therefore, exploring the geographical origin of onion germplasms using molecular characterization is recommended for further research.