Introduction
Materials and Methods
Angiosperm species, permanent voucher slides housed, and anatomical terminology
Cutting & softening wood block, microtome section, and slide observation
Results
Anatomical characteristics required for the identification of some Korean angiosperm sawn timber
An identification key on some Korean angiosperm sawn timber based on anatomical characteristics
Discussion
Introduction
The Korean Peninsula is covered by dense forests for 63.7%. Woody plants in the forests account for about 80% of the total biomass (Sundarapandian et al., 2013). The importance of forests in mitigating the climate crisis is being emphasized because trees capture and store carbon dioxide in the atmosphere through photosynthesis (Kim et al., 2023; Psistaki et al., 2024). To expand economic, social, and cultural value through dense forests, more research and sustainable management are needed though (Korea Forest Service, 2021).
The woody angiosperm plants have various physical and chemical characteristics depending on the tree species. This is because the combination of cells that both perform various functions together and micromorphology is different depending on the tree species (Chu, 2024; Yang, 2019). Therefore, both experts trained and reference materials are needed to identify the sawn timber through anatomical characteristics available only.
The first microscopic anatomical study of Korean woody plants was conducted in 1938 (Yamahayashi, 1938), and then the study of subsequent anatomy was mainly focused on specific taxa or methodologies (Chung and Lee, 1995; Ghimire et al., 2020; Lee, 1988; Lee and Chun, 2009; Park et al., 1994; Soh and Park, 1984). In particular, there is only one report on anatomical key for the identification of Korean woody plants (Lee and Eom,1987). They reported an anatomical key for 39 species of angiosperm. Our research team has recently investigated the anatomical features for useful sawn timber in Korea (Chu, 2024). In addition, there are several studies that not only identify the species and their uses of various wooden objects, but also silicified angiosperm woods from both various archaeological and paleological sites in Korea (Hwang et al., 2020; Mustoe, 2023; Park, 1992, 1993; Park et al., 2022). To conduct above researches, the reference materials of anatomical features at to extant woody angiosperm should be indispensable.
This study aims to investigate the anatomical features on some Korean woody angiosperm, and then to build an identification key for the sawn timber species. The above data might also be useful as reference material when identifying wooden household items discovered in archaeological and paleological study.
Materials and Methods
Angiosperm species, permanent voucher slides housed, and anatomical terminology
The materials used in this study are 63 species (belonging to 25 families and 63 genera) of woody angiosperms collected nationwide from 2005 to 2023 (Table 1). The above wooden materials and their permanent voucher slides were housed in the Botany Laboratory, Division of Science Education, Jeonbuk National University, Korea. The scientific names used in this study followed those of Park (2007) and National Arboretum of Korea (http://www.nature.go.kr>main/Main). The anatomical characteristics of the wood basically followed the guidelines of ‘International Association of Wood Aanatomists’ (IAWA Committee, 2004), and some were added in this study when necessary.
Table 1.
List of angiosperm species and their permanent slides used in this study.
| Family Name | Scientific Name | Voucher slide no.z |
|
Salicaceae 버드나무과 | Populus tremula L. var. davidiana (Dode) C.K. Schneid 사시나무 | JBU AngC12 25120701 |
| Salix chaenomeloides Kimura 왕버들 | JBU AngC12 25120702 | |
|
Juglandaceae 가래나무과 | Juglans mandshurica Maxim. 가래나무 | JBU AngC12 25120703 |
| Platycarya strobilacea Siebold & Zucc. 굴피나무 | JBU AngC12 25120704 | |
| Pterocarya stenoptera DC. 중국굴피나무 | JBU AngC12 25120705 | |
|
Betulaceae 자작나무과 | Alnus japonica (Thunb.) Steud. 오리나무 | JBU AngC12 25120706 |
| Betula pendula Roth 자작나무 | JBU AngC12 25120707 | |
| Carpinus laxiflora (Siebold & Zucc.) Blume서어나무 | JBU AngC12 25120708 | |
|
Fagaceae 참나무과 | Castanea crenata Siebold & Zucc. 밤나무 | JBU AngC12 25120709 |
| Castanopsis sieboldii (Makino) Hatus. Ex T. Yamaz & Mashiba 구실잣밤나무 | JBU AngC12 251207010 | |
| Corylus sieboldiana Blume 참개암나무 | JBU AngC12 251207011 | |
| Quercus mongolica Fisch. ex Ledeb. var. crispula (Blume) H. Ohashi 물참나무 | JBU AngC12 251207012 | |
|
Ulmaceae 느릅나무과 | Celtis sinensis Pers. 팽나무 | JBU AngC12 251207013 |
| Ulmus parvifolia Jacq. 참느릅나무 | JBU AngC12 251207014 | |
| Zelkova serrata (Thunb.) Makino 느티나무 | JBU AngC12 251207015 | |
|
Moraceae 뽕나무과 | Broussonetia papyrifera (L.) L'Hér. ex Vent. 꾸지나무 | JBU AngC12 251207016 |
| Cudrania tricuspidata (Carrière) Bureau ex Lavallée 꾸지뽕나무 | JBU AngC12 251207017 | |
| Ficus oxyphylla Miq. ex Zoll. 모람 | JBU AngC12 251207018 | |
| Morus bombycis Koidz. 산뽕나무 | JBU AngC12 251207019 | |
|
Magnoliaceae 목련과 | Liriodendron tulipifera L. 백합나무 | JBU AngC12 251207020 |
| Magnolia kobus DC. 목련 | JBU AngC12 251207021 | |
|
Lauraceae 녹나무과 | Laurus nobilis L. 월계수 | JBU AngC12 251207022 |
| Lindera obtusiloba Blume 생강나무 | JBU AngC12 251207023 | |
|
Platanaceae 버즘나무과 | Platanus occidentalis L. 양버즘나무 | JBU AngC12 251207024 |
|
Rosaceae 장미과 | Chaenomeles speciosa (Sweet) Nakai 명자꽃 | JBU AngC12 251207025 |
| Kerria japonica (L.) DC. 황매화 | JBU AngC12 251207026 | |
| Malus baccata (L.) Borkh. 야광나무 | JBU AngC12 251207027 | |
| Prunus tomentosa Thunb. 앵도나무 | JBU AngC12 251207028 | |
| Pseudocydonia sinensis (Thouin) C.K.Schneid. 모과나무 | JBU AngC12 251207029 | |
| Pyrus hakunensis Nakai 백운배나무 | JBU AngC12 251207030 | |
| Rhaphiolepis indica (L.) Lindl. Ex Kervar. umbellata (Thunb. Ex Murray) H.Ohashi 다정금나무 | JBU AngC12 251207031 | |
| Rhodotypos scandens (Thunb.) Makino 병아리꽃나무 | JBU AngC12 251207032 | |
| Rosa multiflora Thunb. 찔레꽃 | JBU AngC12 251207033 | |
| Rubus crataegifolius Bunge 산딸기 | JBU AngC12 251207034 | |
| Sorbaria sorbifolia (L.)A. Braunvar. stellipila Maxim. 쉬땅나무 | JBU AngC12 251207035 | |
| Sorbus commixta Hedl. 마가목 | JBU AngC12 251207036 | |
| Spiraea microgyna Nakai 좀조팝나무 | JBU AngC12 251207037 | |
| Stephanandra incisa (Thunb.) Zabel 국수나무 | JBU AngC12 251207038 | |
|
Anacardiaceae 옻나무과 | Rhus chinensis Mill. 붉나무 | JBU AngC12 251207039 |
| Toxicodendron vernicifluum (Stokes) F.A.Barkley 옻나무 | JBU AngC12 251207040 | |
|
Aquifoliaceae 감탕나무과 | Ilex macropoda Miq. 대팻집나무 | JBU AngC12 251207041 |
|
Aceraceae 단풍나무과 | Acer pictum (Thunb.) var. mono (Maxim.) Maxim. ex Franch. 고로쇠나무 | JBU AngC12 251207042 |
|
Hippocastanaceae 칠엽수과 | Aesculus turbinata Blume 칠엽수 | JBU AngC12 251207043 |
|
Actinidiaceae 다래나무과 | Actinidia arguta (Siebold & Zucc.) Planch. ex Miq. 다래 | JBU AngC12 251207044 |
|
Theaceae 차나무과 | Camellia japonica L. 동백나무 | JBU AngC12 251207045 |
| Cleyera japonica Thunb. 비쭈기나무 | JBU AngC12 251207046 | |
| Eurya japonica Thunb. 사스레피나무 | JBU AngC12 251207047 | |
| Stewartia koreana Nakai ex Rehder 노각나무 | JBU AngC12 251207048 | |
| Ternstroemia gymnanthera (Wight & Arn.) Bedd. 후피향나무 | JBU AngC12 251207049 | |
|
Thymelaeaceae 팥꽃나무과 | Daphne jejudoensis M. Kim 제주백서향 | JBU AngC12 251207050 |
|
Alangiaceae 박쥐나무과 | Alangium platanifolium (Siebold & Zucc.) Harms var. trilobum (Miq.) Ohwi 박쥐나무 | JBU AngC12 251207051 |
|
Cornaceae 층층나무과 | Aucuba japonica Thunb. 식나무 | JBU AngC12 251207052 |
| Cornus kousa Burger ex Hance 산딸나무 | JBU AngC12 251207053 | |
|
Ebenaceae 감나무과 | Diospyros lotus L. 고욤나무 | JBU AngC12 251207054 |
|
Symplocaceae 노린재나무과 | Symplocos sawafutagi Nagam. 노린재나무 | JBU AngC12 251207055 |
|
Styracaceae 때죽나무과 | Styrax japonicus Siebold & Zucc. 때죽나무 | JBU AngC12 251207056 |
|
Oleaceae 물푸레나무과 | Chionanthus retusus Lindl. & Paxton 이팝나무 | JBU AngC12 251207057 |
| Fraxinus rhynchophylla Hance 물푸레나무 | JBU AngC12 251207058 | |
| Syringa villosa Vahl subsp. wolfii (C.K. Schneid.) Y. Chen & D.Y. Hong 꽃개회나무 | JBU AngC12 251207059 | |
|
Scrophulariaceae 현삼과 | Paulownia coreana Uyeki 오동나무 | JBU AngC12 251207060 |
|
Caprifoliaceae 인동과 | Lonicera maackii (Rupr.) Maxim. 괴불나무 | JBU AngC12 251207061 |
| Viburnum odoratissimum var. awabuki (K.Koch) Zabel ex Rumpler 야외나무 | JBU AngC12 251207062 | |
| Weigela subsessilis (Nakai) L.H.Bailey 병꽃나무 | JBU AngC12 251207063 | |
|
Theaceae 차나무과 | Camellia japonica L. 동백나무 | JBU AngC12 251207045 |
| Cleyera japonica Thunb. 비쭈기나무 | JBU AngC12 251207046 | |
| Eurya japonica Thunb. 사스레피나무 | JBU AngC12 251207047 | |
| Stewartia koreana Nakai ex Rehder 노각나무 | JBU AngC12 251207048 | |
| Ternstroemia gymnanthera (Wight & Arn.) Bedd. 후피향나무 | JBU AngC12 251207049 | |
|
Thymelaeaceae 팥꽃나무과 | Daphne jejudoensis M. Kim 제주백서향 | JBU AngC12 251207050 |
|
Alangiaceae 박쥐나무과 | Alangium platanifolium (Siebold & Zucc.) Harms var. trilobum (Miq.) Ohwi 박쥐나무 | JBU AngC12 251207051 |
|
Cornaceae 층층나무과 | Aucuba japonica Thunb. 식나무 | JBU AngC12 251207052 |
| Cornus kousa Burger ex Hance 산딸나무 | JBU AngC12 251207053 | |
|
Ebenaceae 감나무과 | Diospyros lotus L. 고욤나무 | JBU AngC12 251207054 |
|
Symplocaceae 노린재나무과 | Symplocos sawafutagi Nagam. 노린재나무 | JBU AngC12 251207055 |
|
Styracaceae 때죽나무과 | Styrax japonicus Siebold & Zucc. 때죽나무 | JBU AngC12 251207056 |
|
Oleaceae 물푸레나무과 | Chionanthus retusus Lindl. & Paxton 이팝나무 | JBU AngC12 251207057 |
| Fraxinus rhynchophylla Hance 물푸레나무 | JBU AngC12 251207058 | |
| Syringa villosa Vahl subsp. wolfii (C.K. Schneid.) Y. Chen & D.Y. Hong 꽃개회나무 | JBU AngC12 251207059 | |
|
Scrophulariaceae 현삼과 | Paulownia coreana Uyeki 오동나무 | JBU AngC12 251207060 |
|
Caprifoliaceae 인동과 | Lonicera maackii (Rupr.) Maxim. 괴불나무 | JBU AngC12 251207061 |
| Viburnum odoratissimum var. awabuki (K.Koch) Zabel ex Rumpler 야외나무 | JBU AngC12 251207062 | |
| Weigela subsessilis (Nakai) L.H.Bailey 병꽃나무 | JBU AngC12 251207063 |
Cutting & softening wood block, microtome section, and slide observation
The research method is identical in the article of “An identification key of some Korean gymnosperm sawn timber; based on anatomical features,” which was being back-to-back submitted to the Korean Journal of Plant Resources (Chu and Whang, 2025; KJPR 38(6) in print). Angiosperm woods comparing to gymnosperm woods are relatively difficult to section as 8~12 μm because the tissue is heterogeneous and harder due to the development of fiber cells that are absent in gymnosperm woods. Therefore, before cutting the experimental wood, the material was boiled using a pressure cooker to make softened. The boiling time varied slightly, depending on the wood strength of species.
Results
Anatomical characteristics required for the identification of some Korean angiosperm sawn timber
The processes of creating an identification key using the anatomical features of Korean woody angiosperm sawn timber are as follows; (1) the anatomical characters and their character states were observed by a compound microscopy, (2) a detailed description was provided along with micrographs (Figs 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15), (3) the observed characters and their character states were organized and summarized in a data table (Table 2), and (4) an taxonomic key based on the above data was designated to identify Korean woody angiosperm species and sawn timber.

Fig. 3.
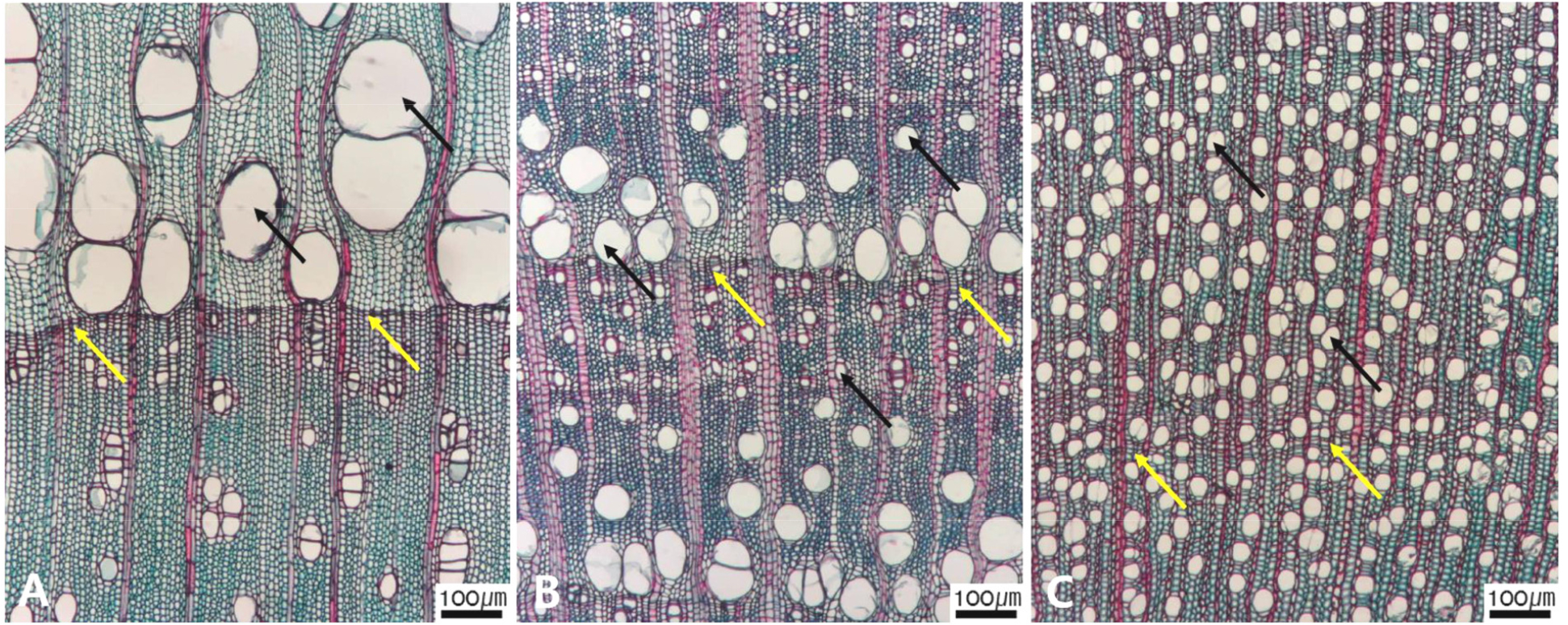
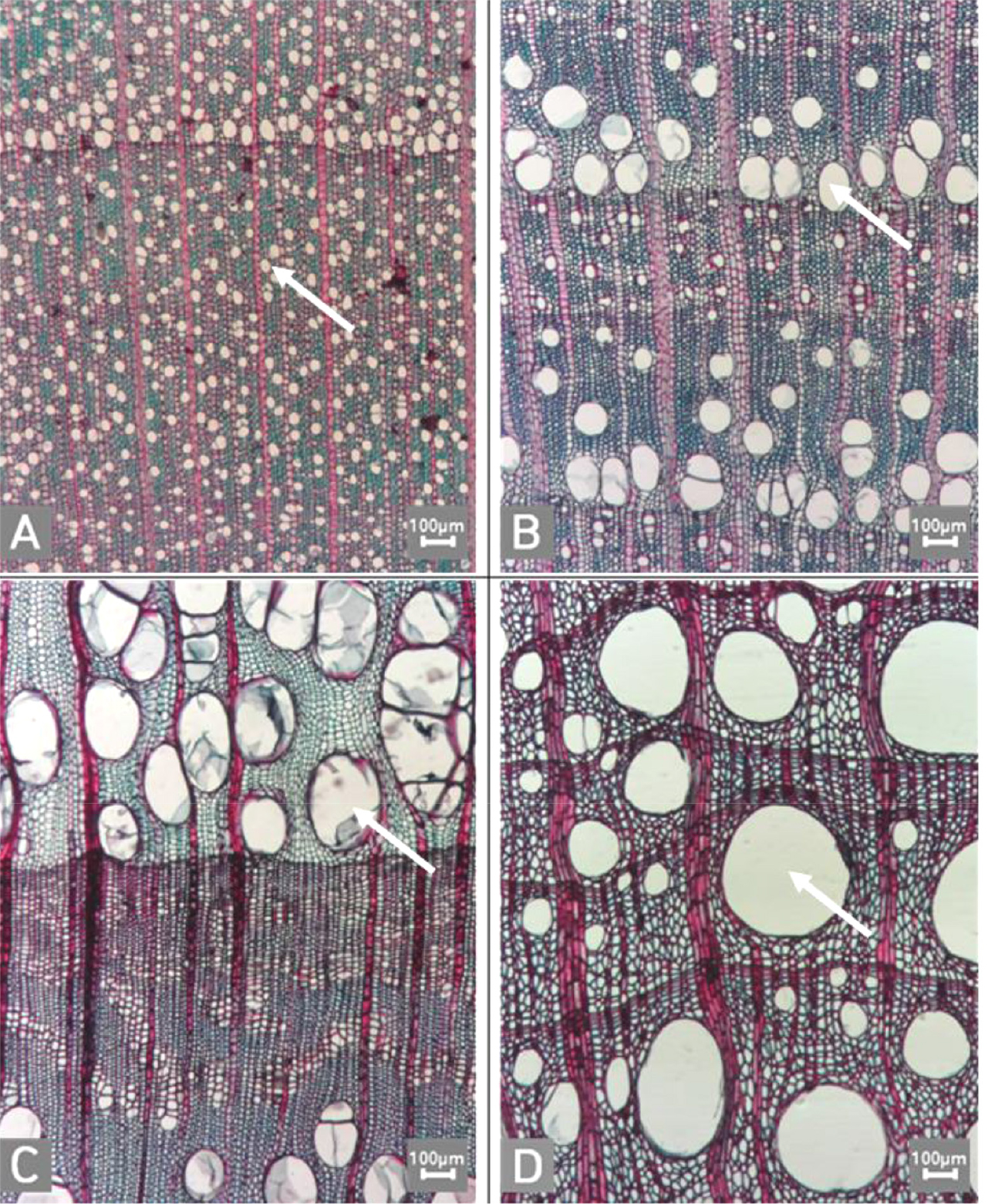
Micrographs showing the vessel grouping in the transverse view. A. Exclusively solitary as 90% or more (Symplocos sawafutagi); B. Radial multiples having two to four in general (Styrax japonicus); C. Clusters (Styrax japonicus); D. Solitary vessel having outline angular (Lonicera maackii). Arrows = vessel grouping types.

Fig. 9.
Micrographs showing the cellular composition of ray parenchyma. A. Procumbent cells only (Alnus japonica); B. Upright or square cells only (Stephanandra incisa); C. Procumbent cells except for having one row of upright or square at marginal (Toxicodendron vernicifluum); D. Procumbent at the multi-cells part except for having a single row of upright cells at marginal (Diospyros lotus); E. Procumbent at the multi-cells part except for having over four rows of upright cells at marginal (Styrax japonicus); F. Procumbent, square and upright cells mixed throughout the ray (S. japonicus).
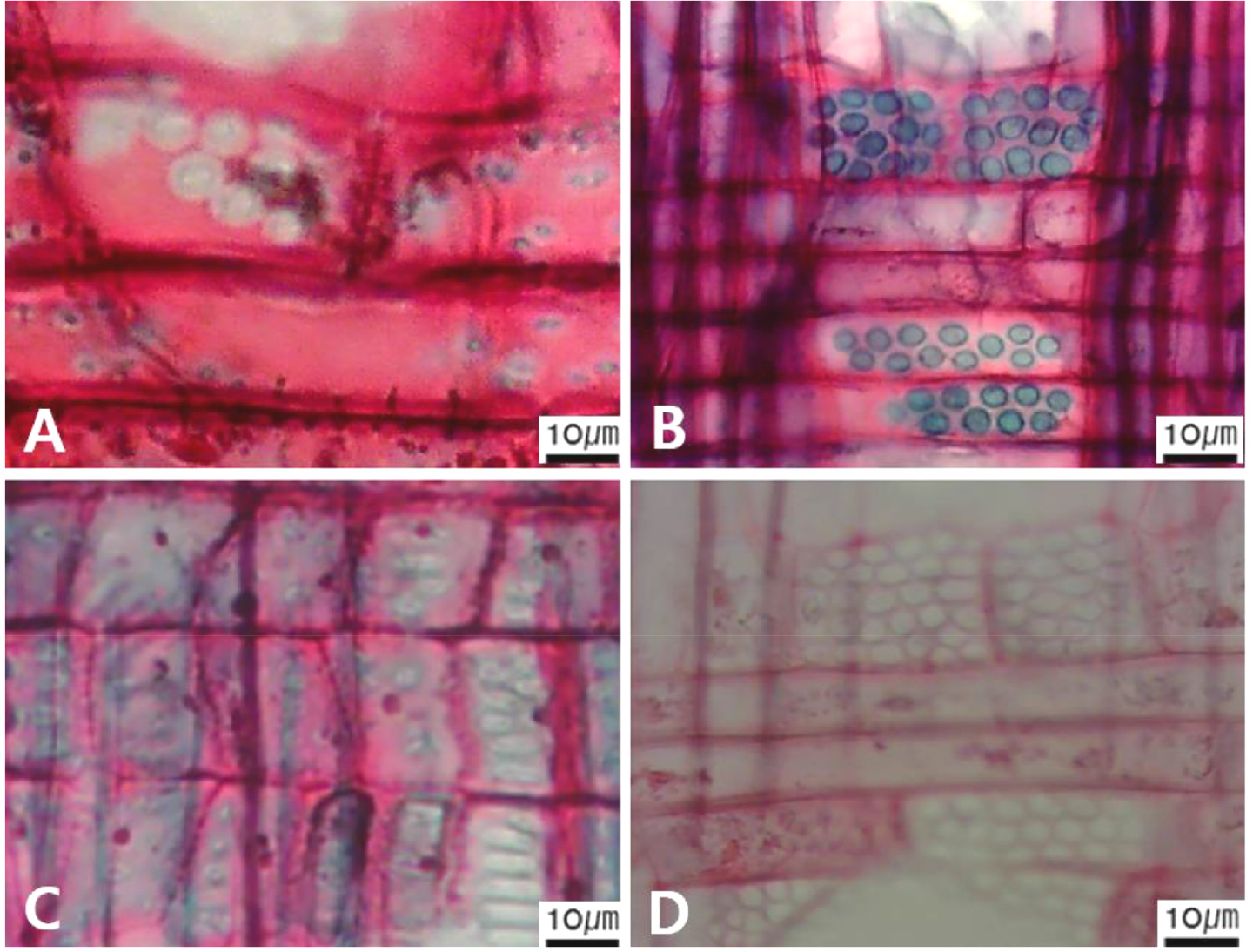
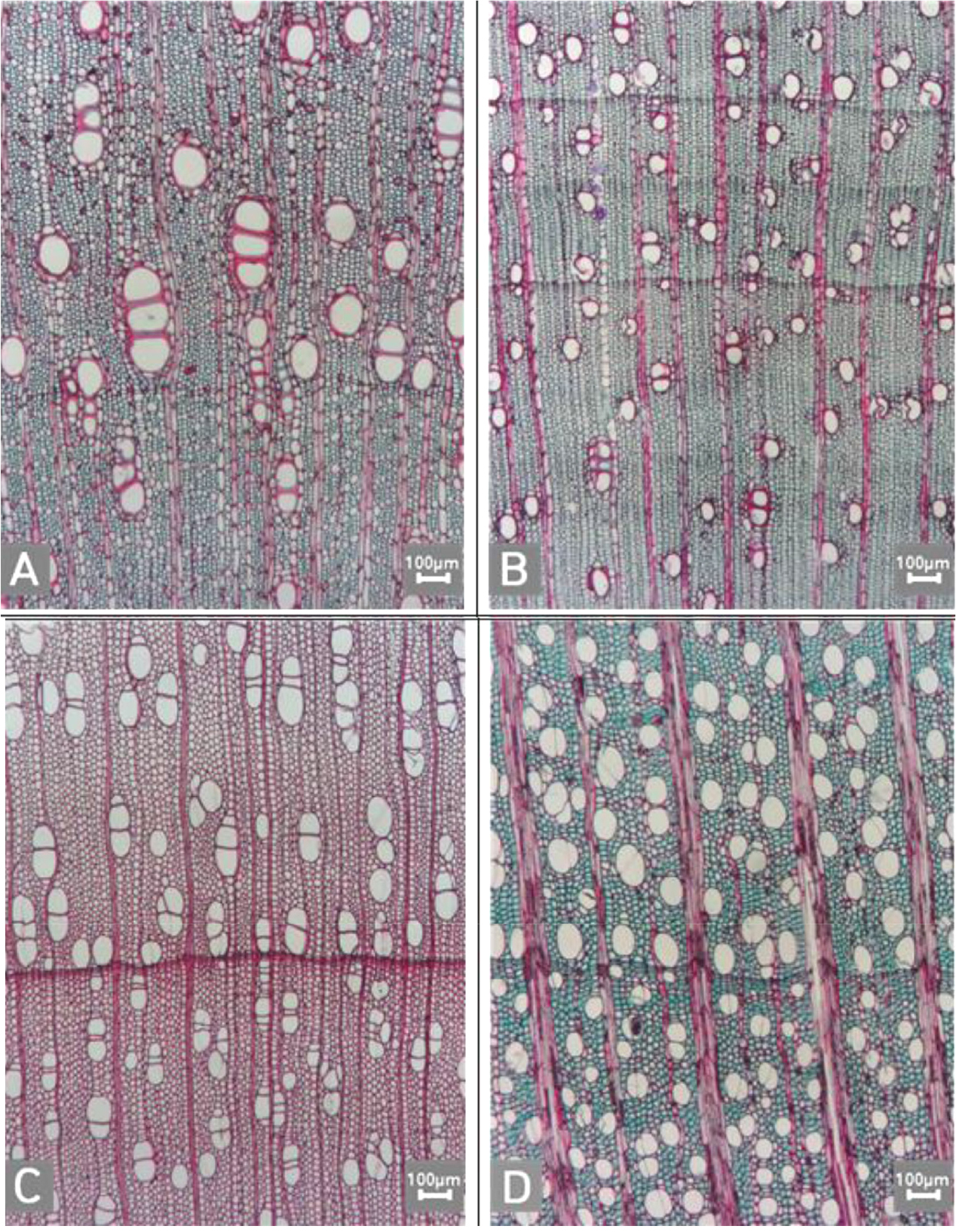
Fig. 10.
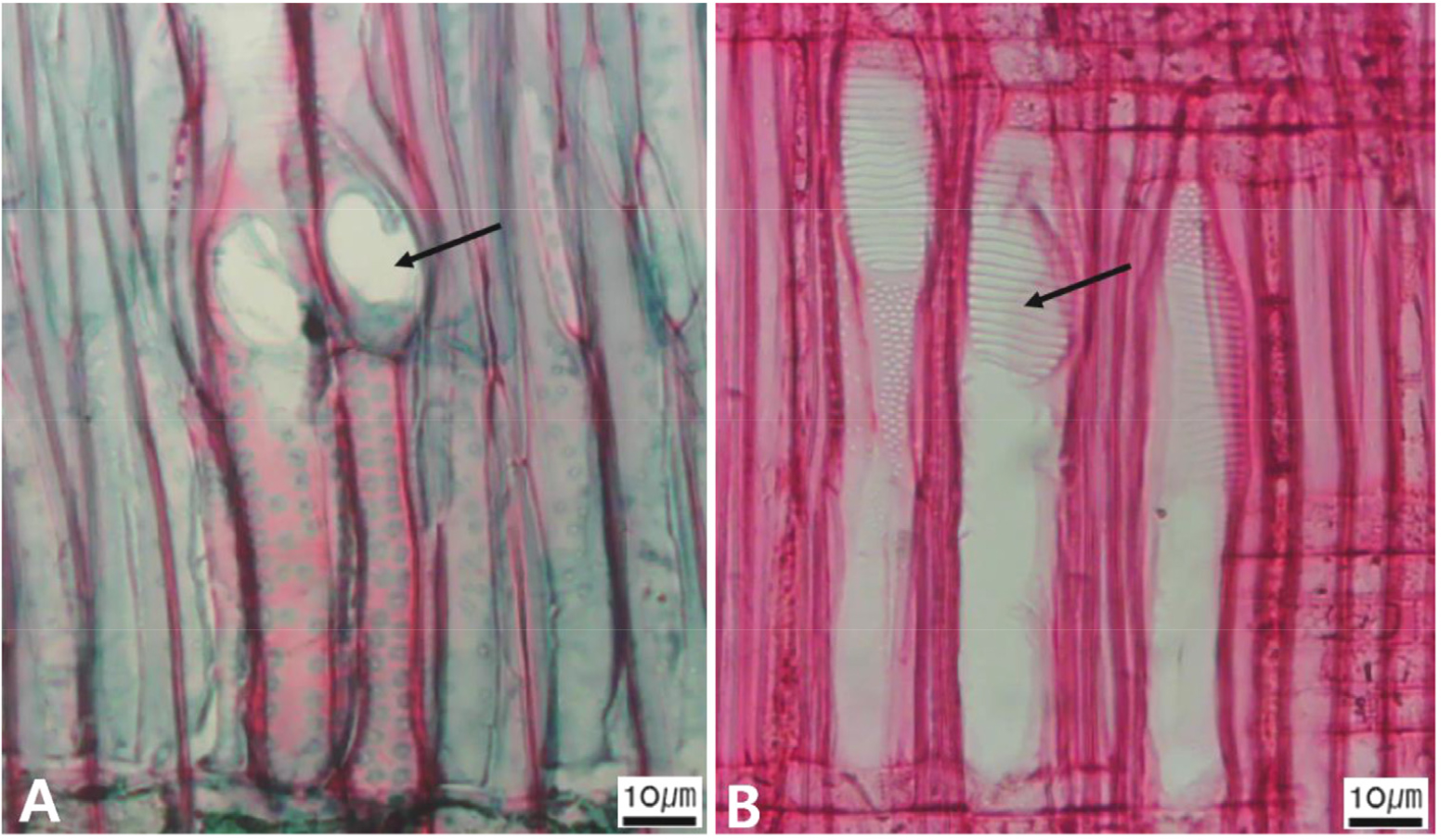
Micrographs showing cross pitting between vessel members and ray parenchyma. A. Distinct borders (Pyrus ussuriensis var. macrostipes); B. Rounded or angular pits with much reduced borders (Populus davidiana); C. Scalariform arrangement of long elliptical or circular pits (Malus baccata); D. Circular or elliptical pits at the margin of ray (Salix pierotii).
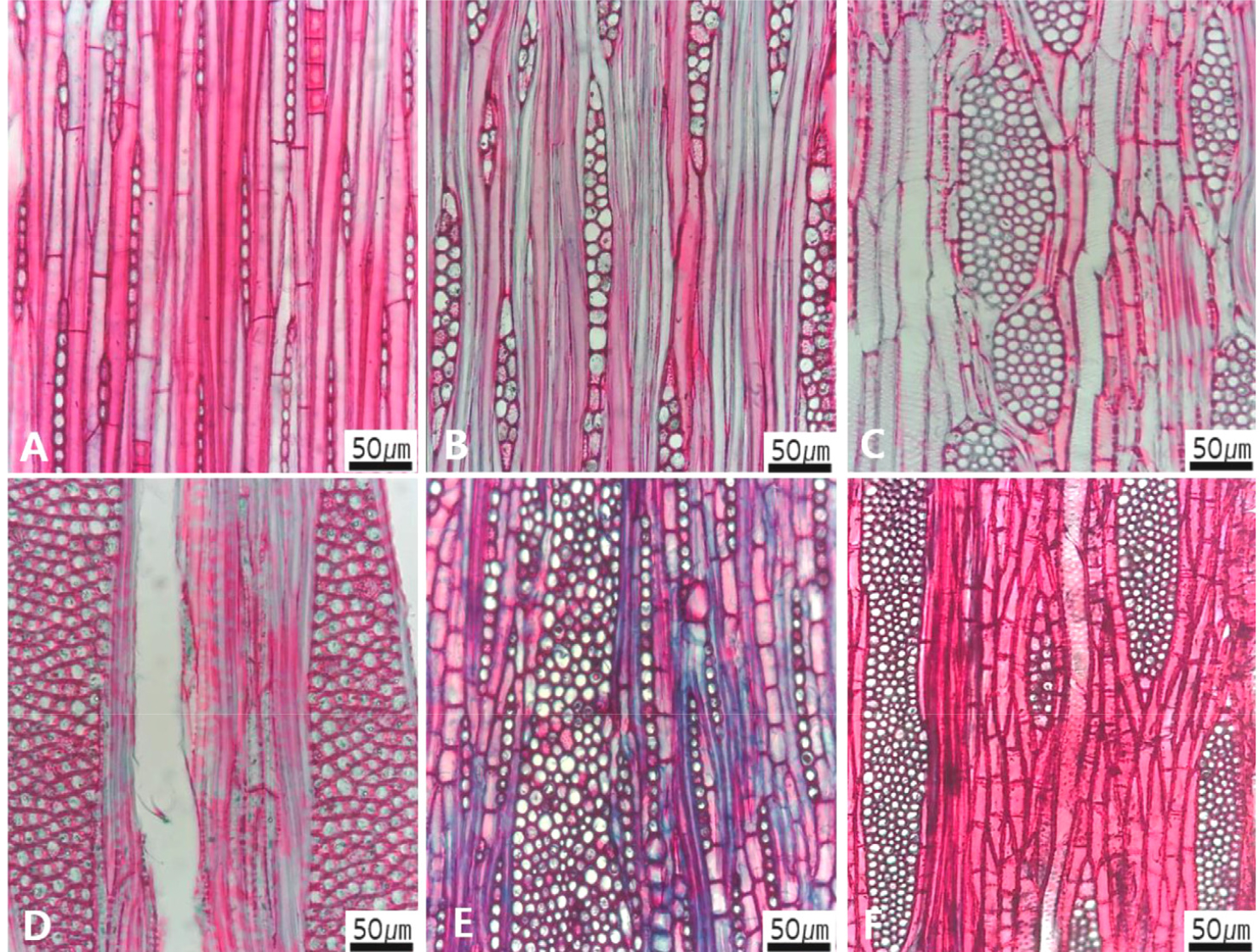
Fig. 11.
Micrographs showing the height and width of ray in the tangential view. A. Rays exclusively uniseriate (Castanopsis sieboldii); B. Ray width 1~3 cells (Lindera obtusiloba); C. Large rays commonly 4- to 10-seriate (Zelkova serrata); D. Large rays commonly >11-seriate (Platanus occidentalis); E. Mixed ray with multiseriate portion as wide as uniseriate portions (Quercus mongolica). F. Ray height >1 ㎜ (Morus bombycis).
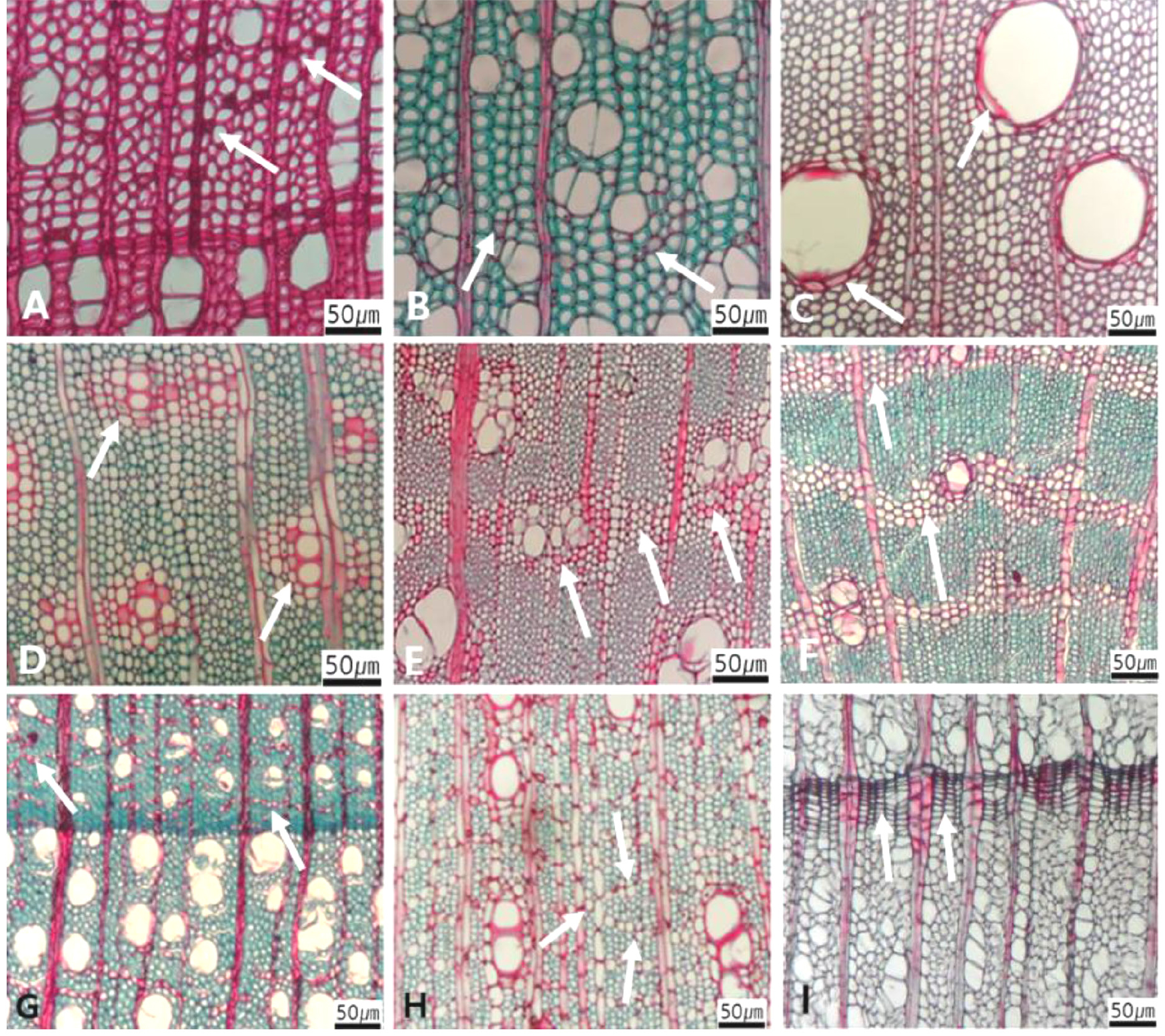
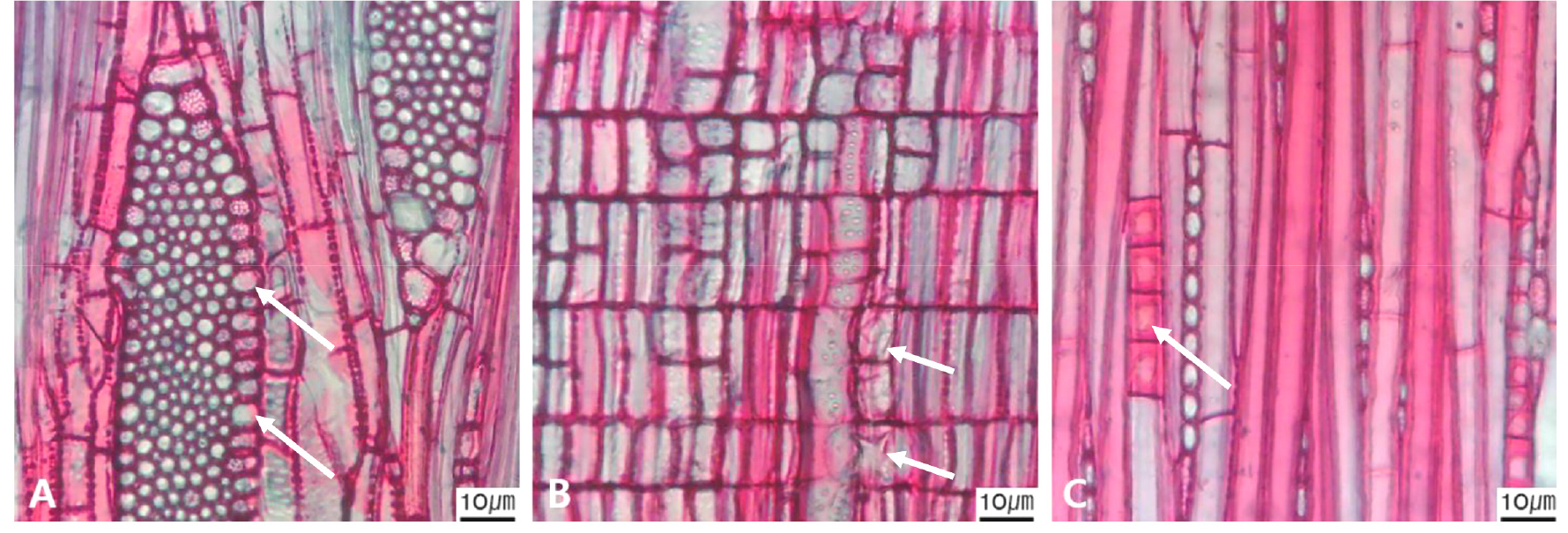
Fig. 13.
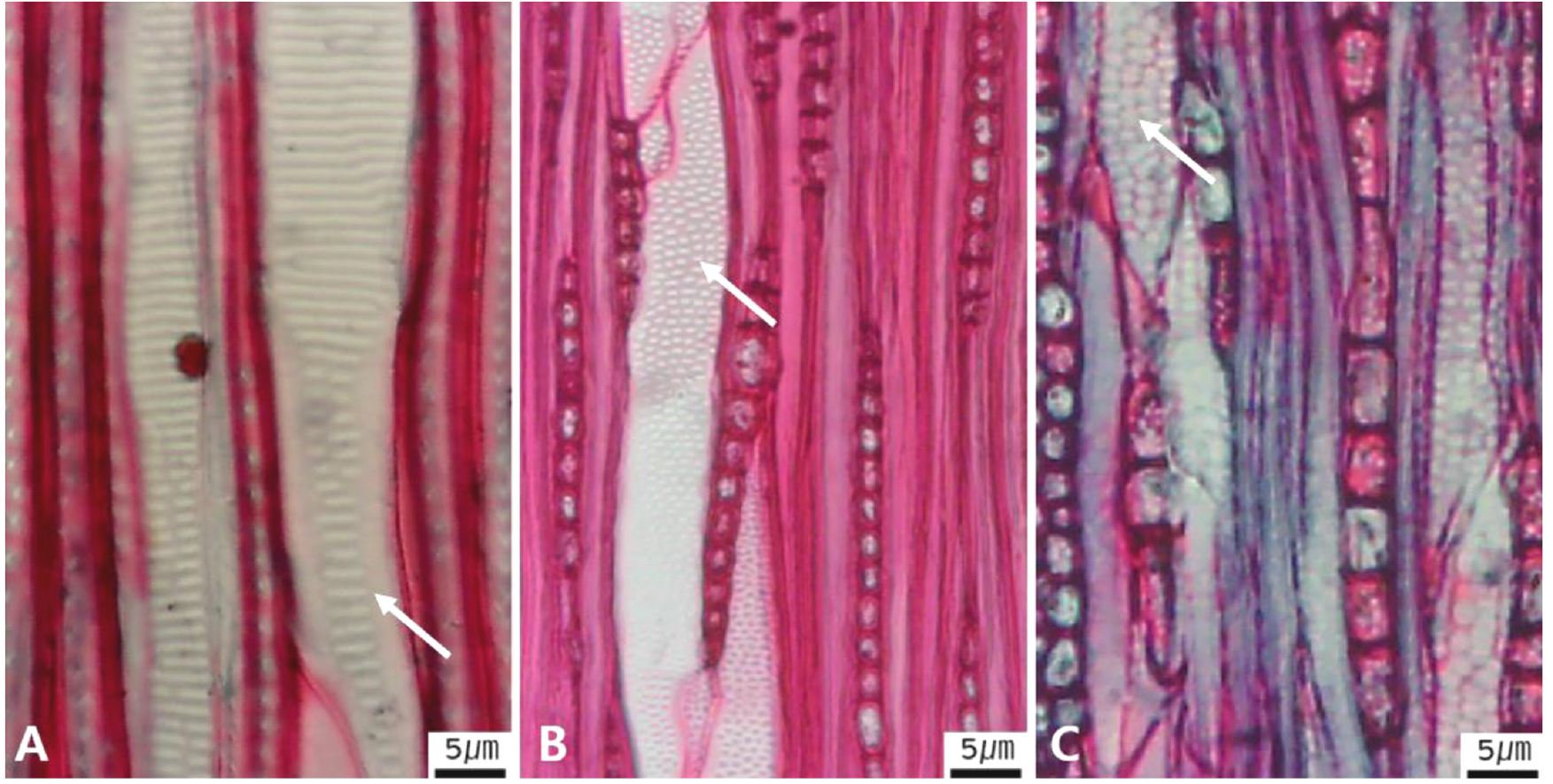
Micrographs showing axial parenchyma such as apotracheal, paratracheal, banded in the transverse view. A-B. Apotracheal axial parenchyma, A. Arrows indicate diffuse cell (Alnus japonica); B. Arrows diffuse-in-aggregates (Ilex macropoda); C-F. Paratracheal axial parenchyma; C. Arrows vasicentric (Juglans regia); D. Arrows aliform (Fraxinus lanuginosa); E. Arrows confluent (Celtis sinensis); F-I. Banded parenchyma; F. Arrows bands more than three cell wide (Ficus oxyphylla); G. Arrows narrow bands or lines up to three cells wide (Stewartia pseudocamellia); H. Arrows reticulate (Diospyros lotus); I. Arrows marginal or in seemingly marginal bands (Liriodendron tulipifera).
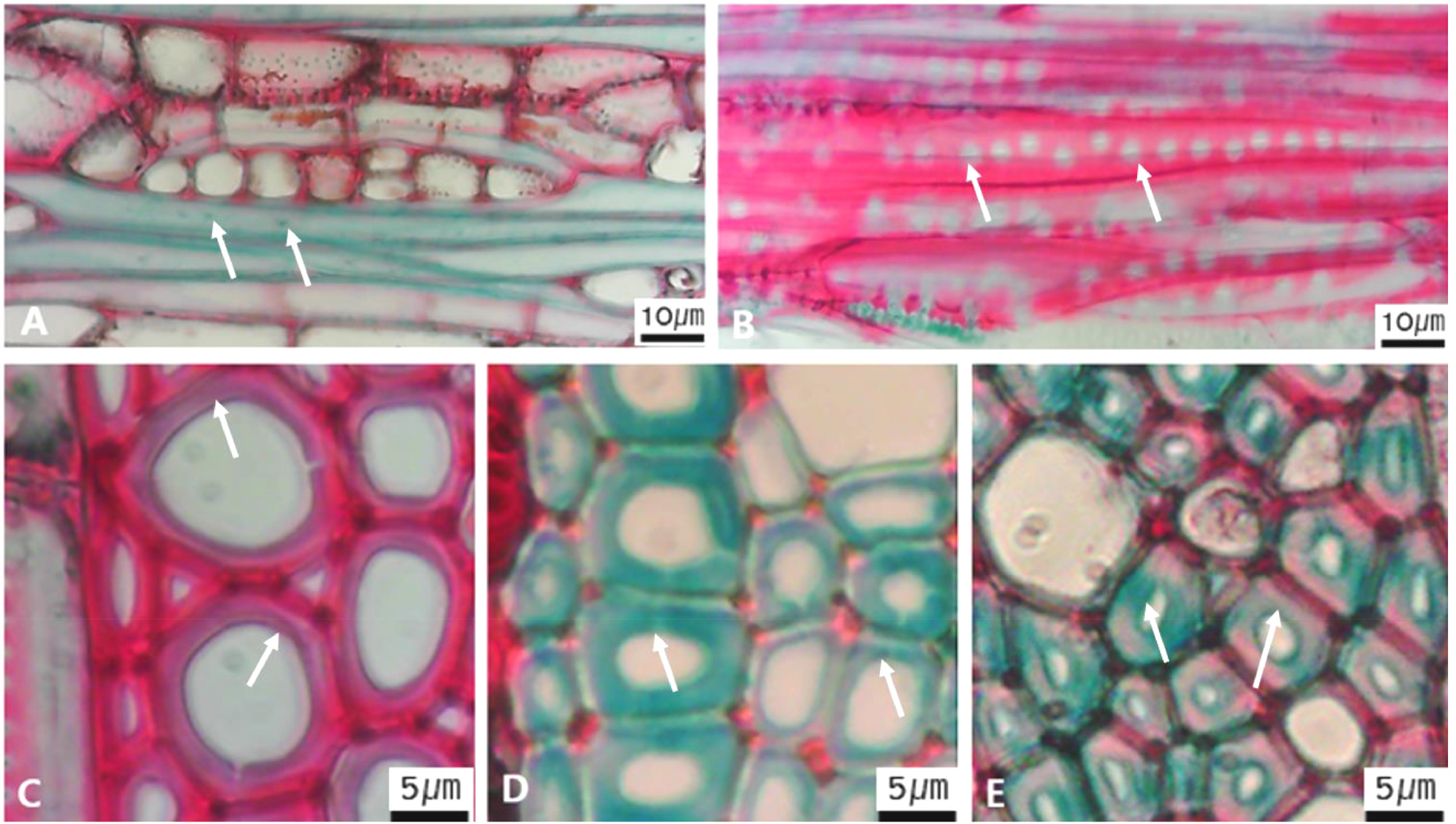
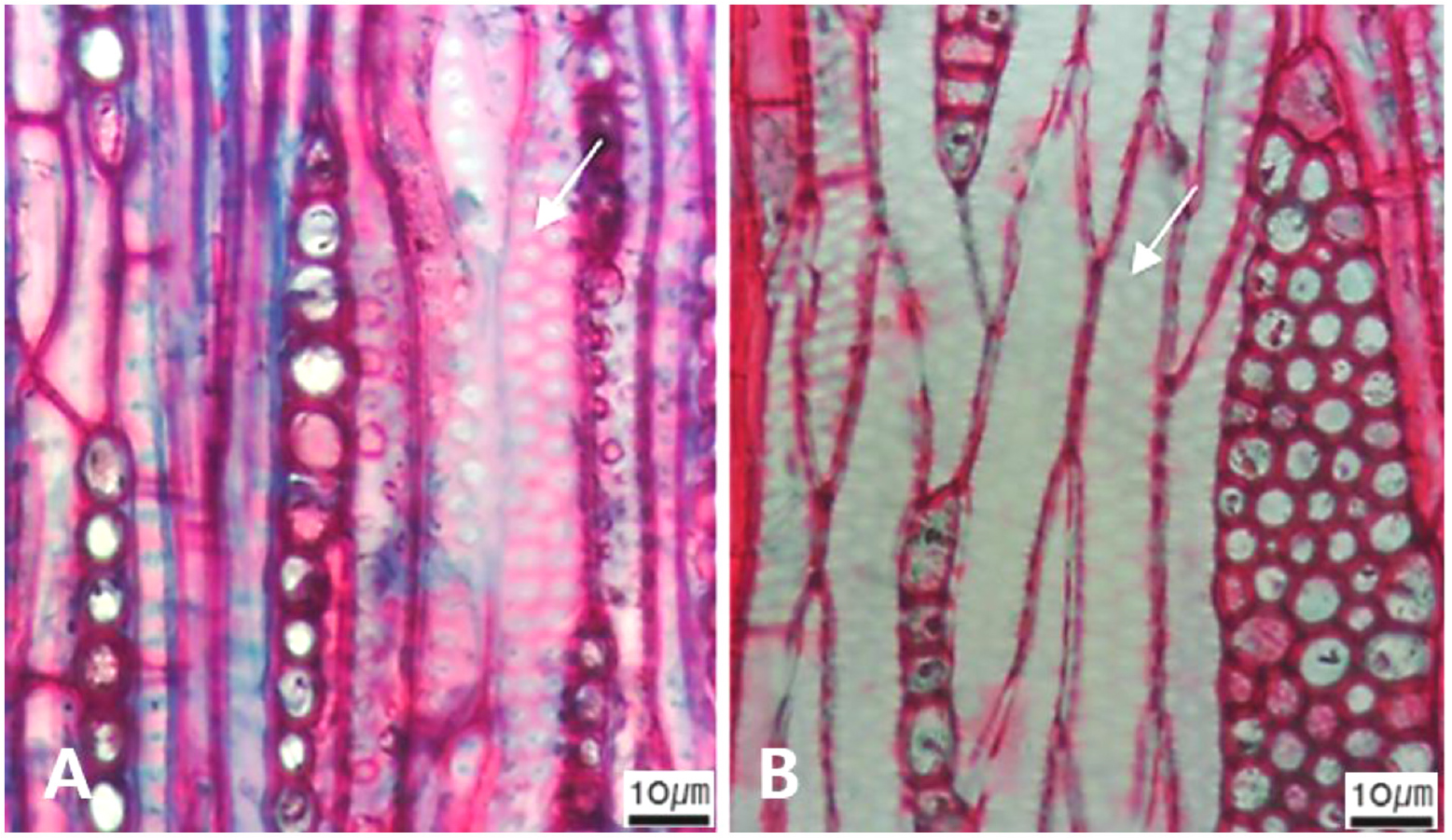
Fig. 15.
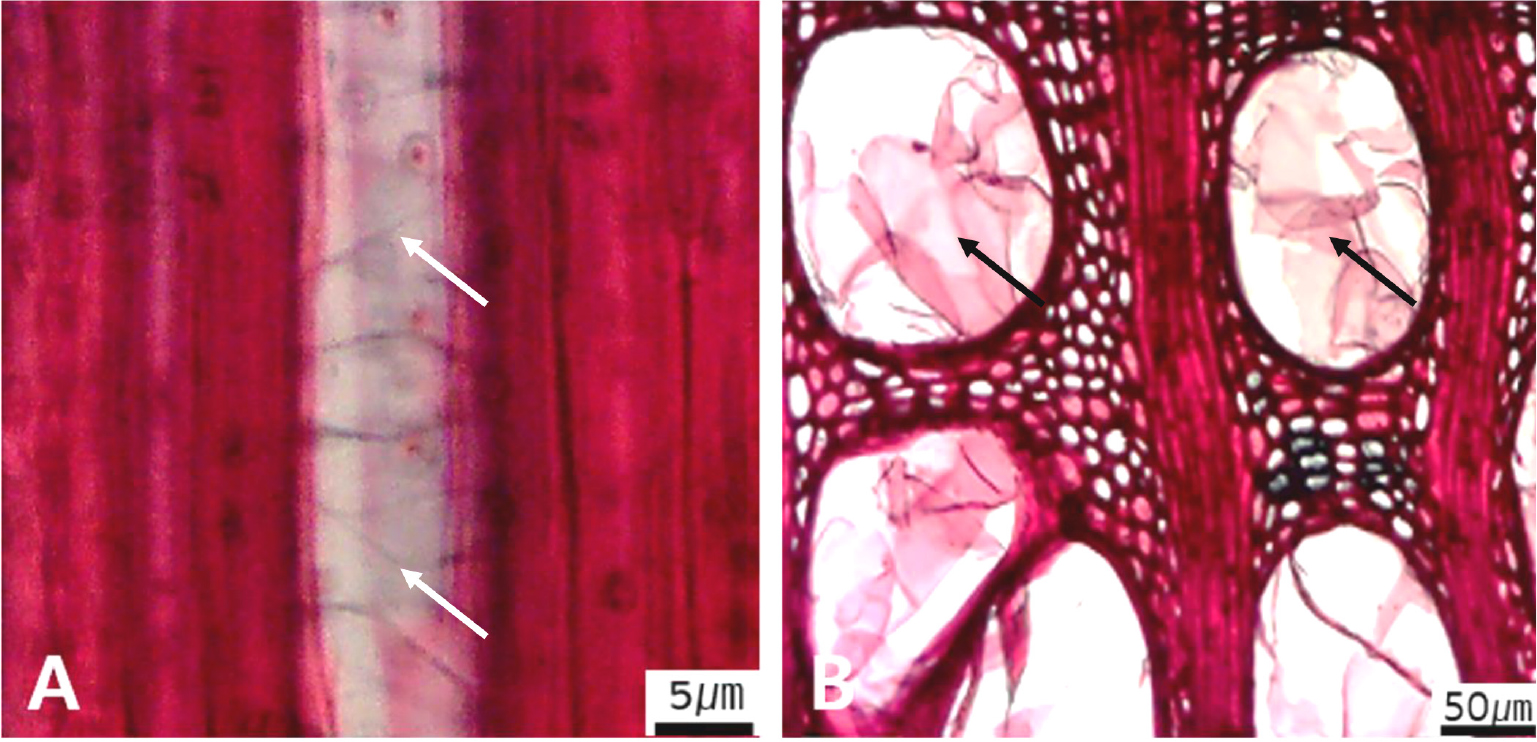
Micrographs showing the fiber pits and its wall thickening. A. Arrows indicate simple to minute bordered (Diospyros lotus); B. Arrows distinctly bordered (Platanus occidentalis); C. Arrows thin-walled fiber (Magnolia Kobus); D. Arrows thin to thick (Rhaphiolepis umbellate); E Very thick-walled (Lonicera maackii).
Table 2.
Characteristics in creating an identification key of some Korean angiosperm sawn timbers.
| Character | Detailed characters | Character statesz | |
| Vessels | Porosity | Ring-/semi-ring-/diffuse-porous | |
| Arrangement | Vessels in tangential bands/in diagonal and radial pattern/in dendritic pattern | ||
| Groupings | Vessels in exclusively solitary (90% or more)/solitary outline angular/radial multiples of four or more common/clusters common | ||
| Perforation plates | Simple perforation plates/ | ||
| Scalariform perforation plates | ≤10/10–20/20–40/≥40 bars | ||
| Reticulate perforation plates | |||
| Inter-vessel pits | Scalariform/opposite/alternate | ||
| Helical thickening | Throughout body of vessel element/only in vessel element tails/narrower vessel elements | ||
| Tyloses | Present/absent | ||
| Mean tangential diameter of vessel lumen | ≤50/50–100/101~200/≥200 μm | ||
| Vessels no. per square mm | ≤5/5~20/21~40/41~100/≥101 | ||
| Ray parenchyma | Cellular composition | All ray cells procumbent/all ray cells upright and square | |
| Body ray cells procumbent with one row of upright and square marginal cells/body ray cells procumbent with mostly 2–4 rows of upright and square marginal cells/body ray cells procumbent with over 4 rows of upright and square marginal cells/rays with procumbent, square, upright cells mixed throughout the ray | |||
| Vessel-ray pitting | Distinct borders/rounded or angular/horizontal to vertical/restricted to marginal rows | ||
| Ray parenchyma | Width | Exclusively uniseriate/1~3 cells/4~10 cells/commonly >10-seriate/multiseriate same as uniseriate portions | |
| Height | >1 ㎜, aggregate rays, and two distinct sizes | ||
| No./1 ㎜ | ≤4/5-12/≥12 | ||
| Sheath cells | Present/absent | ||
| Prismatic crystals | Present/absent | ||
| Axial parenchyma | Apotracheal | Diffuse/diffuse-in-aggregates | |
| Paratracheal | Vasicentric/aliform/confluent/unilateral paratracheal | ||
| Banded | Bands more than three cells wide/narrow bands or lines up to three cells wide/reticulate/scalariform/marginal or in seemingly marginal bands | ||
| No. of cell/strand length | Fusiform/≤2/3-4/5-8/≥9 | ||
| Prismatic crystals | Present/absent | ||
| Fiber | Pits | Simple to minutely bordered/distinctly bordered/common in both radial and tangential walls | |
| Helical thickenings in ground tissue | Present/absent | ||
| Septate fibers | Present/absent | ||
| Cell wall thickness | Very thin-walled (at least three times rather than those of inner wall)/thin to thick (almost wall thickening same as lumen diameter)/very thick (almost absent of lumen) | ||
| Etc. | Rays having storied structure | Present/absent | |
| Crystals in enlarged cells | Present/absent | ||
| Oil and mucilage cells | Present/absent | ||
| Disjunctive ray cell wall | Present/absent | ||
Primary characters, selected for creation an identification key of Korean angiosperm sawn timber, include vessels (Figs 1, 2, 3, 4, 5, 6, 7, 8), rays (Figs 9, 10, 11, 12), axial parenchyma (Fig. 13), tracheid (Fig. 14), and fibers (Fig. 15). The above five primary characters were further divided into sub-characters (Figs 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15). The sub-characters of the vessels include its porosity (Fig. 1), arrangement (Fig. 2), grouping (Fig. 3), perforation plate (Fig. 4), inter-vessel pits (Fig. 5), helical thickening and tyloses (Fig. 6), tangential lumen diameter (Fig. 7), and vessel frequency (Fig. 8). The sub-characters of the rays include the type of rays (Fig. 9), the cross pitting between vessel and rays (Fig. 10), the height and width of ray (Fig. 11), the sheath cell and prismatic crystals within rays (Fig. 12). The sub-characters of the axial parenchyma include apotracheal, paratracheal, and banded type (Fig. 13). The sub-character of fibers includes pitting pattern and cell wall thickness (Fig. 15).
The following is a description of the character and its states (Table 2). The types of vessel porosity have three characters states as ring- (Fig. 1A), semi-ring- (Fig. 1B), and diffuse- porous (Fig. 1C). There are three characters states in the vessel arrangement, tangential bands (Fig. 2A), diagonal (Fig. 2B), radial (Fig. 2C), and dendritic pattern (Fig. 2D). The vessel grouping has also four states, exclusively solitary as 90% or more (Fig. 3A), radial multiples having two to four in general (Fig. 3B), clusters (Fig. 3C), and solitary vessel having outline angular (Fig. 3D). The perforation plate has two states, simple (Fig. 4A) and scalariform (Fig. 4B). The inter- vessel pits have three states, scalariform (Fig. 5A), opposite (Fig. 5B), alternate (Fig. 5C). The helical thickening of vessel is divided into two states, present or absent (Fig. 6A). The tyloses are also divided into two states, present or absent (Fig. 6B). The mean transvers diameter of vessel lumen can be divided into four states, ≤51 μm (Fig. 7A), 51~100 μm (Fig. 7B), 101~20 μm (Fig. 7C), and ≥200 μm (Fig. 7D). The vessel frequency (no/1 ㎟) can be also divided into four state, 5~20 (Fig. 8A), 21~40 (Fig. 8B), 41~100 (Fig. 8C), and ≥100 (Fig. 8D). The cellular composition of ray parenchyma has six states, such as procumbent cells only (Fig. 9A), upright or square cells only (Fig. 9B), procumbent cells except for having one row of upright or square at marginal (Fig. 9C), procumbent at the multi-cells part except for having a single row of upright cells at marginal (Fig. 9D), procumbent at the multi-cells part except for having over four rows of upright cell at marginal (Fig. 9E), and procumbent, square and upright cells throughout the ray (Fig. 9F). The cross pitting between vessel members and ray parenchyma has four states, distinct borders (Fig. 10A), rounded or angular pits with much reduced borders (Fig. 10B), scalariform arrangement of long elliptical or circular pits (Fig. 10C), and circular or elliptical pits at the margin of ray (Fig. 10D). The height and width of ray has six states, such as rays exclusively uniseriate (Fig. 11A), ray width 1~3 cells (Fig. 11B), large rays commonly 4- to 10-seriate (Fig. 11C), large multiseriate portion as wide as uniseriate portions (Fig. 11D), mixed ray with multiseriate portion as wide as uniseriate portions (Fig. 11E), and ray height >1 ㎜ (Fig. 11F). The sheath cell and prismatic crystals have three states, sheath cells making boundary of ray (Fig. 12A), crystal within ray cells (Fig. 12B), and crystal within parenchyma cell (Fig. 12C). The axial parenchyma, such as apotracheal, paratracheal, and banded, have two to four states each. The apotracheal axial parenchyma has two states, diffuse cell (Fig. 13A) and diffuse-in-aggregates (Fig. 13B). The paratracheal axial parenchyma has three states, vasicentric (Fig. 13C), aliform (Fig. 13D), and confluent (Fig. 13E). The banded parenchyma has four states, bands more than three cell wide (Fig. 13F), narrow bands or lines up to three cells wide (Fig. 13G), reticulate (Fig. 13H), and marginal or in seemingly marginal bands (Fig. 13I). The tracheid types have two states, vascular (Fig. 14A) and vasicentric (Fig. 14B). The fiber pits and its wall thickening show two to three states each. The fiber pits have two states, simple to minute bordered (Fig. 15A) and bordered (Fig. 15B). The fiber thickening has three states, thin-walled (at least three times rather than those of inner wall) (Fig. 15C), thin to thick (almost wall thickening same as lumen diameter) (Fig. 15D), and very thick (almost absent of lumen) (Fig. 15F).
An identification key on some Korean angiosperm sawn timber based on anatomical characteristics
1A. Porosity of vessel ring-porous in transverse view 2
2A. Arrangement of vessel tangential band pattern 3
3A. Ray width 1~3 seriate in tangential view Broussonetia papyrifera
3B. Ray width 4~10-seriate in tangential view Morus bombycis
2B. Arrangement of vessel dentritic Castanea crenata
2C. Arrangement of vessel diagonal or radial pattern 4
4A. Present of sheath cell in tangential view Celtis sinensis
5B. Absent of tyloses in transverse view Zelkova serrata
5A. Present of tyloses in transverse view 6
4B. Absent of sheath cell in tangential view 5
6A. All ray cells procumbent Ulmus parvifolia
6B. All ray cells procumbent, and upright or square at marginal cells Rhus chinensis
2D. Arrangement of vessel radial & dentritic pattern 7
7A. Rays exclusively uniseriate in tangential view Castanopsis sieboldii
7B. Rays width 1- to 3-seriate in tangential view Chionanthus retusus
7C. Larger ray width 4- to 10-seriate in tangential view Platycarya strobilacea
7D. Larger rays commonly >10-seriate in tangential view Quercus mongolica
2E. Absent of vessel arrangement in transverse view 8
8A. Vessels exclusively solitary with circular or elliptical Alangium platanifolium var. trilobum
8B. Vessels exclusively solitary with elliptical 9
9A. Rays width 1- to 3-seriate in tangential view Syringa villosa subsp. wolfii
9B. Larger ray width 4- to 10-seriate in tangential view Actinidia arguta
8C. Mixed with solitary and radial multiples vessels 10
10A. All ray cells procumbent Fraxinus rhynchophylla
10B. Body ray cells procumbent, and one row of upright and square marginal cells Toxicodendron vernicifluum
8D. Mixed with solitary and radial cluster vessel Cudrania tricuspidata
1B. Porosity of vessel semi-ring-porous in transverse view 11
11A. Simple perforation plates in radial view 12
11B. Scalariform perforation plates in radial view Camellia japonica
12A. All ray cells procumbent 13
12B. Body ray cells procumbent, and one row of upright and square marginal cells Juglans mandshurica
13A. Axial parenchyma diffuse-in-aggregates Pterocarya stenoptera
13B. Axial parenchyma short aliform or confluent Paulownia coreana
1C. Porosity of vessel diffuse-porous in transverse view 14
14A. Arrangement of vessel dentritic pattern in transverse view Daphne jejudoensis
14B. Arrangement of vessel non-dentritic 15
15A. Ray height aggregate in tangential view 16
16A. Simple perforation plates Carpinus laxiflora
16B. Scalariform perforation plates with below 10 bars Corylus sieboldiana
16C. Scalariform perforation plates with 11~20 bars Alnus japonica
15B. Ray height same in both uniseriate and multi- seriate width 17
17A. Vessels mainly solitary with circular or elliptical Symplocos sawafutagi
17B. Vessels mainly solitary with angular Eurya japonica
17C. Vessels in radial multiples of four or more Diospyros lotus
17D. Vessels in radial multiples, and solitary vessels angular Styrax japonicus
15C. Ray height of two distinct sizes in tangential view 18
18A. Vessels mainly solitary with circular or elliptical 19
19A. Number of Rays ≤4/1 ㎜ in tangential view 20
20A. Prismatic crystals present in ray cells Kerria japonica
20B. Prismatic crystals absent in ray cells Rosa multiflora
19B. Number of Rays 5~12/1 ㎜ in tangential view Spiraea microgyna
19C. Number of Rays ≥13/1 ㎜ in tangenticl view Stephanandra incisa
18B. Vessels mainly solitary with angular 21
21A. Simple perforation plates 22
22A. Present of helical thickening on vessel walls Prunus tomentosa
22B. Absent of helical thickening on vessel walls Sorbaria sorbifolia var. stellipila
21B. Scalariform perforation plates Aucuba japonica
18C. Vessels clusters common Rubus crataegifolius
15D. Ray width with exclusively uniseriate in tangential view 23
23A. All ray cells procumbent in radial view 24
24A. Hierarchical rays in tangential view Aesculus turbinata
24B. No hierarchical rays in tangential view Populus tremula var. davidiana
23B. Body ray cells procumbent, and one row of upright or square marginal cells Salix chaenomeloides
23C. Body ray cells procumbent, and 1~4 rows of upright or square marginal cells Cleyera japonica
15E. Ray width with exclusively one to three cells in tangential view 25
25A. All ray cells procumbent in radial view 26
26A. Simple perforation plates in transverse view 27
27A. Inter-vessel pits opposite in radial view Chaenomeles speciosa
27B. Inter-vessel pits alternate in radial view 28
28A. Fiber walls thickness very thin less than >3 times diameter Pyrus hakunensis
28B. Fiber walls thickness very thin and/or thick as normal Sorbus commixta
28C. Fibers very thick-walled as much as no cell cavity Pseudocydonia sinensis
26B. Scalariform perforation plates in transverse view Betula pendula
25B. Procumbent body ray with 1-4 rows of upright or square marginal cells 29
29A. Present of oil or mucilage cell in radial view Lindera obtusiloba
29B. Absent of oil or mucilage cell in radial view 30
30A. Present of disjunctively thickened 2’wall ray cell in radial view Stewartia koreana
30A. Absent of disjunctively thickened 2’wall ray cell in radial view 31
31A. Present of crystals in enlarged cells in radial view Rhaphiolepis indica var. umbellata
31B. Absent of crystals in enlarged cells in radial view 32
32A. Axial parenchyma in marginal or in seemingly marginal bands 33
33A. Vessel frequency/1 ㎜ ≥100 in transverse view Magnolia kobus
33B. Vessel frequency/1 ㎜ 5~20 in transverse view Laurus nobilis
32B. No axial parenchyma in seemingly marginal 34
34A. Vessel-ray pits with distinct borders Weigela subsessilis
34B. Vessel-ray pits with rounded or scalariform borders Malus baccata
15F. Large rays commonly 4- to 10-seriate in tangential view 35
35A. Solitary vessel outline angular in transverse view 36
36A. Vessel-ray pits with distinct borders in radial view 37
37A. Fiber wall thickness very thin and/or thick as normal 38
37B. Fiber wall very thickened as no cell cavity Lonicera maackii
38A. Present of crystals in enlarged ray cells Ilex macropoda
38B. Present of crystals in enlarged ray cells 39
39A. Present of helical thickenings of vessel elements Ternstroemia gymnanthera
39B. Absent of helical thickenings of vessel elements Cornus kousa
36B. Vessel-ray pits with rounded or scalariform border Viburnum odoratissimum var. awabuki
35B. Solitary vessel outline angular or elliptical in transverse view 40
40A. Vessels exclusively solitary, 90% or more Acer pictum
40B. Vessels in radial multiples ≥4 common Ficus oxyphylla
40C. Vessel solitary and/or radial multiple Liriodendron tulipifera
15G. Larger rays commonly >10-seriate in tangential view 41
41A. All ray cells procumbent in radial view Platanus occidentalis
41B. Body ray cells procumbent with one row of upright with one row of upright and/or square marginal cells in radial view Rhodotypos scandens
Discussion
The anatomical features of woody angiosperms vary across species. This is because the physical and chemical properties of cell and tissue among species are different (Yang, 2019). The anatomical features, such as cell and tissue microstructures, of woody angiosperms can be identified by trained anatomists under a microscopy (Soh and Park, 1984). In the era of climate crisis, accurately identifying woody species sawn timber and processing and using them according to their characteristics will have the pivotal effect for the mitigation of the concentration of carbon dioxide in the atmosphere (Chu, 2024; IPCC, 2006). This study selected the major anatomical characters and their character states of some Korean angiosperm sawn timber, and then described their characteristics together with micrographs in detail. Based on obtained data above, an identification key was created to enable rapid identification of woody angiosperm. These are expected that this would be at first helpful for identification of tree species and sawn timber, and further in the development of the wood industry (Kim et al., 2023). It would be also used as a reference material when creating an anatomical database for all Korean woody angiosperms in the future.
The identification key, based on the anatomical characteristics on some Korean woody angiosperms, created in this study has two major features. First, it described the right sectioning direction among three dimensions that can better explain the characteristics of both cells and tissues of species. In general, the arrangement of the axial parenchyma is easy to observe through transverse view, and the shape of the terminal wall of the same tissue is easy to observe through tangential and radial views. However, in order to create a classification key using anatomical features for many species of woody angiosperms, three dimensional features were investigated and described all characters and their character states. Second, we developed a classification key using anatomical features that are easy to observe under a microscope. For example, Ulmus parvifolia and Zelkova serrata, belonging to Ulmaceae, have anatomically similar structure, and they can be easily distinguished by the present or absent of extended intracellular crystals within the cell. However, the identification key created here, they were distinguished by the present or absent of tyloses, which are easier to observe. Rather than tissues that are difficult to observe or features that easily confuse, we mainly applied characters and character states that can be easily observed under a microscope.
The character and character states, which appear only in some genera, were adopted in the classification key to enable quick and accurate in identifying species and wood. For example, species belonging to Alnus and Carpinus in Betulaceae are characterized by having aggregate rays, which are relatively short and small rays that appear as a single wide ray height. Species belonging to Celtis (Ulmaceae) have distinctly sheathed cells making boundary of ray. Species belonging to Aucuba (Cornaceae) are characterized by scalariform perforation plate as the very high number with more than ≥40 bars. Also, the vessel arrangements of Chionanthus (Oleaceae) and Castanea (Fagaceae) have distinct dendritic pattern in transverse view (IAWA Committee, 2004; Chu, 2024).
Meanwhile, several multidisciplinary studies related to the wood features from archaeological sites in Korea have been recently conducted (Kang et al., 2024; Lee and Kim, 2023; Park, et al., 2022). For example, wooden artifacts during the Baekje period excavated in a submerged state from the archaeological site of Ssangbuk-ri in Buyeo had identified and preserved (Kang et al., 2024). The wooden artifacts reported above are four as follow: (1) the surface coating of the lacquerware was confirmed to be from Castanea spp., (2) the square container with a handle from Juglans spp., (3) the arrow-shaped wooden object from Pyrus spp., and (4) the Y-shaped and drill-shaped wooden artifacts were crafted from hard pine. As appeared in the above study, the lack of anatomical reference data for Korean woody angiosperm makes it possible to identify plants up to the genus, but difficult to identify the species. Therefore, this study would be expected to serve as a helpful reference for future plant species identification in situations like the above.