Introduction
Materials and Methods
Reagents
Sample preparation
Cell culture and treatment
Cell viability assay
Phagocytic capacity
Measurement of NO production
SDS-PAGE and Western blot analysis
Reverse transcriptase-polymerase chain reaction (RT-PCR)
Statistical analysis
Results
Effect of BCS on the activation of RAW264.7 cells
Effect of BCS on activation of MAPK, NF-κB and PI3K/AKT signaling pathway in RAW264.7 cells
Contribution of AKT, JNK and NF-κB to BCS-mediated expression of immunomodulators in RAW264.7 cells
Effect of TLR2 on BCS-mediated expression of immunomodulators through the activation of AKT, JNK and NF-κB
Inhibitory effect of BCS on pro-inflammatory mediators and MAPK/NF-κB signaling activation in LPS-stimulated RAW264.7 cells
Discussion
Introduction
Innate immune surveillance system is the first line of the defensive mechanisms that protect hosts from invading external pathogens (Akira, 2009; Kawai and Akira, 2007). The innate immune response plays by recognizing pathogens, phagocytosing them, and secreting immunomodulators (Ozinsky et al., 2000) which play an important role in activating defense immunity against pathogens and their components (Kawai and Akira, 2006). Host cells express various pattern recognition receptors (PRRs) that sense diverse pathogen-associated molecular patterns (PAMPs), ranging from lipids, lipoproteins, proteins and nucleic acids (Akira et al., 2006; Akira, 2009; Labonte et al., 2014; Pluddemann et al., 2011). The regulation of the innate immune system is induced through cell receptors called pattern-recognition receptors (PRRs), of which several classes of PRRs, including Toll-like receptors (TLRs) and cytoplasmic receptors, recognize distinct microbial components and directly activate immune cells (O’Neill, 2006; Takeda and Akira, 2005). Exposure of immune cells to the ligands of these receptors activates intracellular signaling cascades that rapidly induce the expression of diverse and unique genes involved in the inflammatory and immune responses (Akira et al., 2006).
Toll-like receptors (TLRs) are one of the best knew the pattern recognition receptor (PRR) families (Jin and Lee, 2008; Lee et al., 2015). These receptors are expressed either on the cell surface or intracellular endosomes (Kawai and Akira, 2010) and are type I transmembrane receptors which most predominantly expressed in immune cells, such as macrophages (Medzhitov, 2001). To date, 11 human TLRs and 13 mouse TLRs have been identified, and TLRs are classified into several groups based on the types of PAMPs they recognize (Akira et al., 2006; Kawai and Akira, 2007; Zhang et al., 2004). Each TLR can detect distinct PAMPs from bacteria, viruses, fungi and parasites (Akira et al., 2006; Kumar et al., 2009). One of the most studied TLRs is TLR2, that is associated with TLR1 and TLR6 to form heterodimeric complex that recognizes lipoproteins, lipopeptides, peptidoglycans and lipoteichoic acid (Kang et al., 2009), but also lipoarabinomannan from Mycobacteria (Wieland et al., 2004), phenol-soluble modulin from Staphylococcus (Ozinsky et al., 2000), zymosan from fungi (Sato et al., 2003) and glycosylphosphatidylinositol from Trypanosoma cruzi (Campos et al., 2001). The recognition of microbial pathogens and their components by TLRs triggers the activation of intracellular signaling and results in production of immunomodulators such as nitric oxide (NO), inducible nitric oxide synthase (iNOS), cytokines such as interleukin-1β (IL-1β), interleukin-6 (IL-6), interleukin-12 (IL-12), and tumor necrosis factor-α (TNF-α) and chemokines (Chang, 2010; Deng et al., 2016; Kawamoto et al., 2008; Kopf et al., 2010; Liu et al., 2017; Zheng et al., 2017). The production of these immunomodulators is important for the killing of intracellular infectious agents (Kuan et al., 2012). The production of these immunomodulators has been reported to tightly regulated by several signaling pathways including nuclear factor-κB (NF-κB) (Lee et al., 2017; Ren et al., 2016), mitogen-activated protein kinases (MAPKs) and phosphatidylinositol 3-kinase/Protein kinase B (PI3K/AKT) (Cho et al., 2017; Guo et al., 2015; Hommes et al., 2003; Pearson et al., 2001).
A vegetable is a young vegetable that has been sprouted from seeds for about one to three leaves and has sprouted for about a week (Khalil et al., 2007). It is known that these sprouts contain 5 to 10 times more nutrients that grown vegetables (Khalil et al., 2007). Sprout vegetables contain a variety of bioactive substances, but studies on their efficacy are still lacking. Bok choy (Brassica. campestris var. chinensis) is one of the representative crops belonging to Brassicaceae (Kurilich et al., 1999), and brassica vegetables include cabbage, broccoli, cauliflower, brussels sprouts, kale and turnips (Soengas et al., 2011).
Bok choy is consumed all over the world and has been reported to contain various vitamins (such as vitamin C, vitamin A and β-carotene ) and minerals (such as calcium and potassium) (Soengas et al., 2011). Indole-3-carbinol in brassica vegetables have been reported to have various physiological activities such as antioxidant, anti-cancer and cardioprotective activity (Kris-Etherton et al., 2002; Maruthanila et al., 2014; Soengas et al., 2011). However, despite having these various nutrients, studies on immunostimulatory activity of bok choy sprout have not been conducted properly. Therefore, in this study, we evaluated in vitro immunostimulatory activity of bok choy sprout (BCS) using mouse macrophage RAW264.7 cells.
Materials and Methods
Reagents
Chemicals for the experiment were obtained from Sigma (St. Louis, MO, USA). Dulbecco's Modified Eagle's Medium (DMEM)/F-12 1:1 Modified medium were purchased from Lonza (Walkersville, MD, USA). Primers for immunomodulatory gene and housekeeping gene (GAPDH) were obtained from Invitrogen (Grand Island, NY, USA). Primary antibodies against IκB-α, phospho-IκB-α, IKK, phospho-IKK, ERK1/2, phospho-ERK1/2, p38, phospho-p38, JNK, phospho-JNK, AKT, phospho-AKT and β-actin were purchased from Cell Signaling (Bervely, MA, USA).
Sample preparation
The bok choy sprouts and bok choy were kindly provided from Jong Hwa Shin as a professor at the Department of horticulture in Andong National University, Korea. Briefly, 6 grams of bok choy seed were sown in petri dishes and cultivated for 9 days so that the cotyledon was unfolded and epicotyl was elongated using four-room growth chamber. Supplying distilled water at each irrigation was increased from 10 to 50 mL depending on the growth of bok choy. And irrigation interval also increased from daily to twice a day. The temperature in the chamber was maintained at 20℃ which optimum temperature for germination. The photoperiod was set equal to 12 hours each day and night. During the daytime, fluorescent lamp was used to maintain a light intensity of 150-180 μM/㎡/s at the height of the crops. Bok choy sprout extract was obtained according to our previous method (Seo and Jeong, 2020). Briefly, one gram of freeze-dried bok choy sprout and bok choy was extracted with 20 mL of distilled water for 72 h under shaking at the room temperature. After 72 h, the water-soluble extract was filtered and centrifugation at 15,000 rpm for 10 min, the supernatant was freeze-dried to obtain extracts from bok choy sprout (BCS) and bok choy (BC), which was dissolved in deionized water and kept in a refrigerator until use.
Cell culture and treatment
Murine macrophage cell line, RAW264.7 was purchased from American Type Culture Collection (Manassas, VA, USA) and grown in DMEM/F-12 media containing 10% fatal bovine serum (FBS), 100 U/mL penicillin and 100 ㎍/mL streptomycin. The cells were incubated at 37℃ in 5% CO2 atmosphere.
Cell viability assay
Effect of BCS on cell viability was assessed by MTT assay. Briefly, RAW264.7 cells were inoculated in 96-well plates, after 24 h, the cells were treated with BCS at various concentration of 0, 50 and 100 ㎍/mL to culture for another 24 h. Then, the cells were incubated with MTT solution (1 ㎎/mL) for an additional 2 h. The resulting formazan crystals were dissolved in DMSO. The plates were read on UV/Visible spectrophotometer (Human Cop., Xma-3000PC, Seoul, Korea) at 570 ㎚.
Phagocytic capacity
Effects of BCS on the phagocytosis of RAW264.7 cells was evaluated according to the modified method reported by Liu et al. (2017). After cells were cultured with various concentrations (0, 50, and 100 ㎍/mL) of BCS for 24 h, the culture medium was removed and 100 μL/well 0.1% neutral red solution was added. The cells were incubated for another 3 h and 200 μL/well lysis solutions (ethanol/acetic acid 1:1) were added, then the cells were lysis for 30 min at room temperature. The absorbance was determined at 540 ㎚ by UV/Visible spectrophotometer (Human Cop., Xma-3000PC, Seoul, Korea).
Measurement of NO production
RAW264.7 cells were treated with BCS at various concentration of 0, 50 and 100 ㎍/mL. After 24 h, NO level in the cultured supernatants was measured by Griess assay. Briefly, the supernatants were collected and mixed with an equal volume of Griess reagent (Sigma Aldrich, St. Louis, MO, USA) and followed by incubation for 15 min at the room temperature. The absorbance values were determined by microplate reader (Human Cop., Xma-3000PC, Seoul, Korea) at 540 ㎚.
SDS-PAGE and Western blot analysis
RAW264.7 cells were stimulated for various periods of time, the supernatant was removed and cells were washed twice with cold phosphate-buffered saline (PBS). Cells were then lysed, in radioimmunoprecipitation assay (RIPA) buffer (Boston Bio Products, Ashland, MA, USA) supplemented with protease inhibitor cocktail (Sigma-Aldrich) and phosphatase inhibitor cocktail (Sigma-Aldrich), and centrifuged at 15,000 rpm for 10 min at 4℃. Protein quantitative was performed with bicinchoninic acid (BCA) protein assay (Pierce, Rockford, IL, USA). The proteins were separated on SDS-PAGE and then transferred to PVDF membrane (Bio-Rad Laboratories, Inc., Hercules, CA, USA). The PVDF membranes were blocked for non-specific binding with 5% non-fat dry milk in Tris-buffered saline containing 0.05% Tween 20 (TBS-T) for 1h at room temperature and then incubated with specific primary antibodies (target proteins) at 4℃ overnight. The membranes were incubated with the optimal horseradish peroxidase-conjugated secondary antibody for 1 h. Signals were visualized with an enhanced chemiluminescence kit (Amersham Biosciences, Piscataway, NJ, USA). The density of Western blot bands was calculated using the software UN-SCAN-IT gel version 5.1 (Silk Scientific Inc. Orem, UT, USA).
Reverse transcriptase-polymerase chain reaction (RT-PCR)
After treatment, total RNA was extracted according to our previous method (Seo and Jeong, 2020). Briefly, total RNA was prepared using a RNeasy Mini Kit (Qiagen, Valencia, CA, USA) and total RNA (1 ㎍) was reverse-transcribed using a Verso cDNA Kit (Thermo Scientific, Pittsburgh, PA, USA) according to the manufacturer's protocol for cDNA synthesis. PCR was carried out using PCR Master Mix Kit (Promega, Madison, WI, USA) and mouse primers for iNOS, IL-1β, IL-6, IL-12, TNF-α, MCP-1 and GAPDH as followed : mouse iNOS: forward 5'-ttgtgcatcgacctaggctggaa-3' and reverse 5'-gacctttcgcattagcatggaagc-3', mouse IL-1β: forward 5'-ggcaggcagtatcactcatt-3' and reverse 5'-cccaaggccacaggtattt-3', mouse IL-6: forward 5'-gaggataccactcccaacagacc-3' and reverse 5'-aagtgcatcatcgttgttcataca-3', mouse IL-12: forward 5'-aaccagacccgcccaagaac-3' and reverse 5'-gatcctgagcttgcacgcaga-3', mouse TNF-α: forward 5'-tggaactggcagaagaggca-3' and reverse 5'-tgctcctccacttggtggtt-3' mouse MCP-1: forward 5'-gaaggaatgggtccagacat-3' and reverse 5'-acgggtcaacttcacattca-3', GAPDH: forward 5'-ggactgtggtcatgagcccttcca-3' and reverse 5'-actcacggcaaattcaacggcac-3'. The PCR results were visualized using agarose gel electrophoresis. The density of mRNA bands was calculated using the software UN-SCAN-IT gel version 5.1 (Silk Scientific Inc. Orem, UT, USA).
Statistical analysis
All the data are shown as mean ± SD (standard deviation). Statistical significance was determined by Student’s t-test. Differences with *P or #P <0.05 were considered statistically significant.
Results
Effect of BCS on the activation of RAW264.7 cells
Green leafy vegetables such as broccoli and bok choy (BC) are commonly known to enhance the body’s immune system. In particular it is reported that selenium contained in BC acts as important function to protect the human body from external pathogens by activating immune cells (Hoffmann and Berry, 2008). BCS used in this study was grown for 9 days in a growth chamber. The weight of BCS was 0.040 ± 0.0056 g, the shoot length of BCS was 38.3 ± 1.83 ㎜, and the root length of BCS was 37.4 ± 4.54 ㎜ (Fig. 1). Thus, in order to compare the macrophage activation of BCS and BC, we investigated the production of immunomodulators after treating BCS or BC to RAW264.7 cells. As shown in Fig. 2A, the BCS and BC were found to similar activity to induce immunomodulators such as iNOS, IL-1β, IL-6 and TNF-α. Based on these results, BCS is thought to have the function of enhancing immunity through macrophage activation like BC. Thus, we used BCS for further study. The effect of BCS on the NO production of RAW264.7 cells was determined by Griess assay (Fig. 2B). When RAW264.7 cells were treated with various concentrations (50 and 100 ㎍/mL) of BCS, NO level was significantly increased in a dose-dependent manner. This result suggested that BCS may stimulate NO production in RAW264.7 cells. MTT assay was performed to confirm the cytotoxicity of BCS (Fig. 2C). There was no significant difference between control and BCS treatment groups, which means that BCS have no cytotoxicity to RAW264.7 cells. In order to evaluate whether BCS-activated RAW 264.7 cells produced immunomodulators, we investigated the mRNA expressions of iNOS, IL-1β, IL-6, IL-12, TNF-α, and monocyte chemoattractant protein-1 (MCP-1) (Fig. 2D). The levels of all immunomodulators significantly increased in response to BCS stimulation, especially the IL-1β and IL-6 mRNA expressions were dramatically increased. This result shows that BCS significantly increased the production of iNOS, IL-1β, IL-6, IL-12, TNF-α, and MCP-1 in RAW264.7 cells. iNOS is the most crucial enzyme to synthesize NO after the activation of macrophages. Our results showed that BCS treatment increased the expression levels of iNOS, which are associated with NO production. The most typical activity of macrophage was phagocytosis as a primary immune response against pathogen. As shown in Fig. 2E, when RAW264.7 cells were treated with BCS, BCS significantly stimulated phagocytosis of RAW264.7 cells in a dose dependent manner.
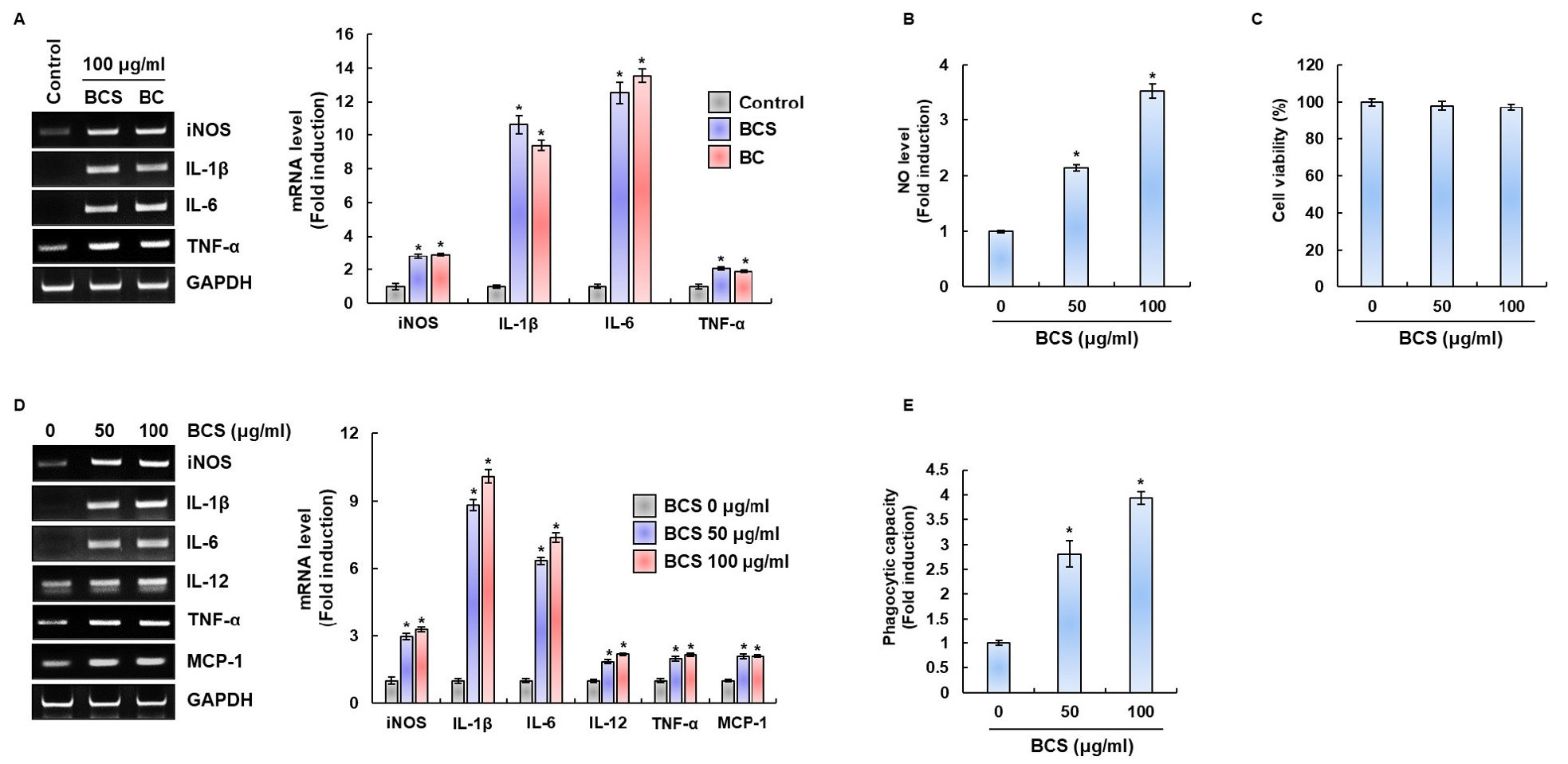
Fig. 2
Effect of BCS on the activation of RAW264.7 cells. (A) Gene expression in RAW264.7 cell treated with BCS or BC for 24 h. For RT-PCR analysis, total RNA was prepared. GAPDH was used as internal control for RT-PCR. *P<0.05 compared to the cells without the treatment. (B) NO production in RAW264.7 cell treated with BCS for 24 h. NO production was measured by Griess assay. *P < 0.05 compared to the cells without the treatment. (C) Viability in RAW264.7 cell treated with BCS for 24 h. Cell viability was measured using MTT assay. (D) Gene expression in RAW264.7 cell treated with BCS for 24 h. For RT-PCR analysis, total RNA was prepared. GAPDH was used as internal control for RT-PCR. (E) Phagocytosis capacity in RAW264.7 cell treated with various concentrations of BCS for 24 h. *P<0.05 compared to the cells without the treatment.
Effect of BCS on activation of MAPK, NF-κB and PI3K/AKT signaling pathway in RAW264.7 cells
To demonstrate the possible mechanism by which BCS activates immunomodulators such as NO, iNOS, IL-1β, IL-6, IL-12, TNF-α and MCP-1, we investigated MAPKs, NF-κB and PI3K/ATK signaling pathways. When RAW264.7 cells were treated with 100 ㎍/mL of BCS for the indicated times, ERK and JNK phosphorylation was significantly increased after exposure to BCS for 1, 3, 6, 10 and 24 h, respectively (Fig. 3A). Furthermore, it was demonstrated that p38 phosphorylation was significantly activated after exposure to BCS for 6, 10 and 24 h, respectively in a time-dependent manner (Fig. 3A). NF-κB is an essential transcription factor that regulates genes involved in both the innate and adaptive immune responses. To assess whether BCS regulates NF-κB activation, BCS-mediated phosphorylation levels of IKK and IκB-α, and total IκB-α were measured in RAW264.7 cells. The p-IKKα/β levels were significantly increased after exposure to BCS for 1, 3, 6 and 10 h, respectively, the p-IκBα levels were significantly increased after exposure to BCS for 3, 6, 10 and 24 h, respectively (Fig. 3B). These results indicate that BCS may activate NF-κB signaling pathway. Further investigations were conducted to elucidate the action point of BCS in RAW 264.7 cells. PI3K/AKT is an upstream factor for NF-κB activation (Karin and Ben-Neriah, 2000; Park et al., 2009), we investigated the regulation of AKT phosphorylation level by BCS treatment in RAW 264.7 cells. As demonstrated in Fig. 3C, phosphorylation of AKT was significantly increased after exposure to BCS for 1, 3 and 6 h without changing total AKT protein levels. Therefore, BCS may exert immunostimulatory effects through the activation of the MAPK and AKT/NF-κB signaling pathways in RAW264.7 macrophages.
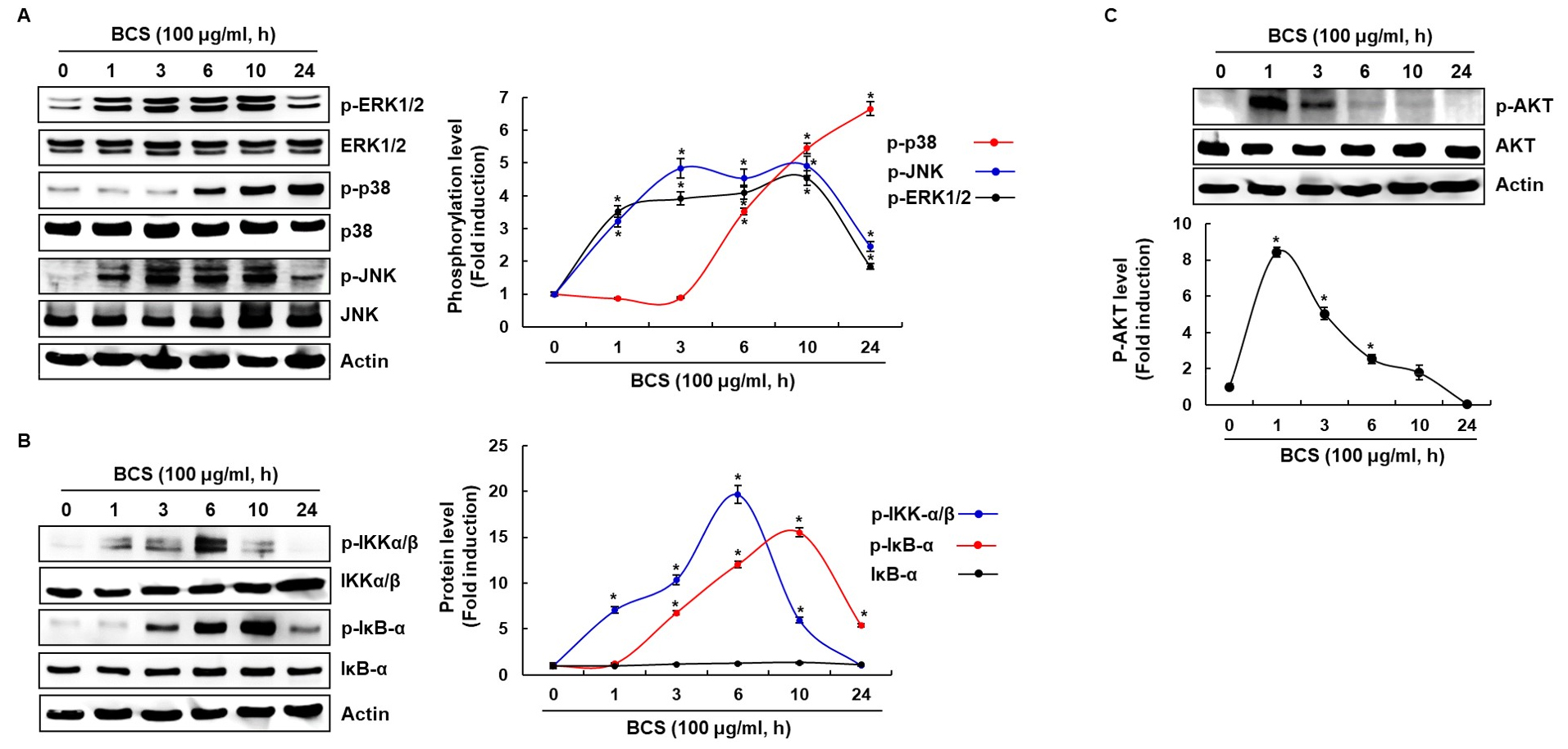
Fig. 3
Effect of BCS on the MAPK, NF-κB and AKT signaling activation in RAW264.7 cells. (A, B, C) RAW264.7 cells were treated with BCS for the indicated times. All cell lysates were subjected to SDS-PAGE and the Western blot was performed using antibodies against p-ERK1/2, p-p38, p-JNK, p-IKKα/β, p-IκB-α, p-AKT, ERK1/2, p38, JNK, IKKα/β, IκB-α, AKT or actin. Actin was used as internal control for Western blot analysis. *P < 0.05 compared to the cells without the treatment.
Contribution of AKT, JNK and NF-κB to BCS-mediated expression of immunomodulators in RAW264.7 cells
To determine the signaling pathways such as MAPK, NF-κB and PI3K/ATK associated with BCS’s macrophage activation, the cells was pretreated with the specific kinase inhibitors such as PD98059 (ERK1/2 inhibitor), SB203580 (p38 inhibitor), SP600125 (JNK inhibitor), BAY 11-7082 (NF-κB inhibitor), LY294002 (PI3K/AKT inhibitor) and NAC (ROS inhibitor), and then co-treated with BCS for 24h. As a result, inhibition of ERK1/2 by PD98059 and inbition of p38 by SB203580 did not affect BCS-induced NO prodution (Fig. 4A). However, JNK by SP600125, NF-κB by BAY 11-7082, AKT by LY294002 and ROS by NAC was found to significantly down regulate the BCS-induced NO, iNOS, IL-1β and IL-6 generation (Fig. 4A and 4B). These results indicate that BCS-mediated expression of immunomodulators may be dependent on AKT, JNK and NF-κB.
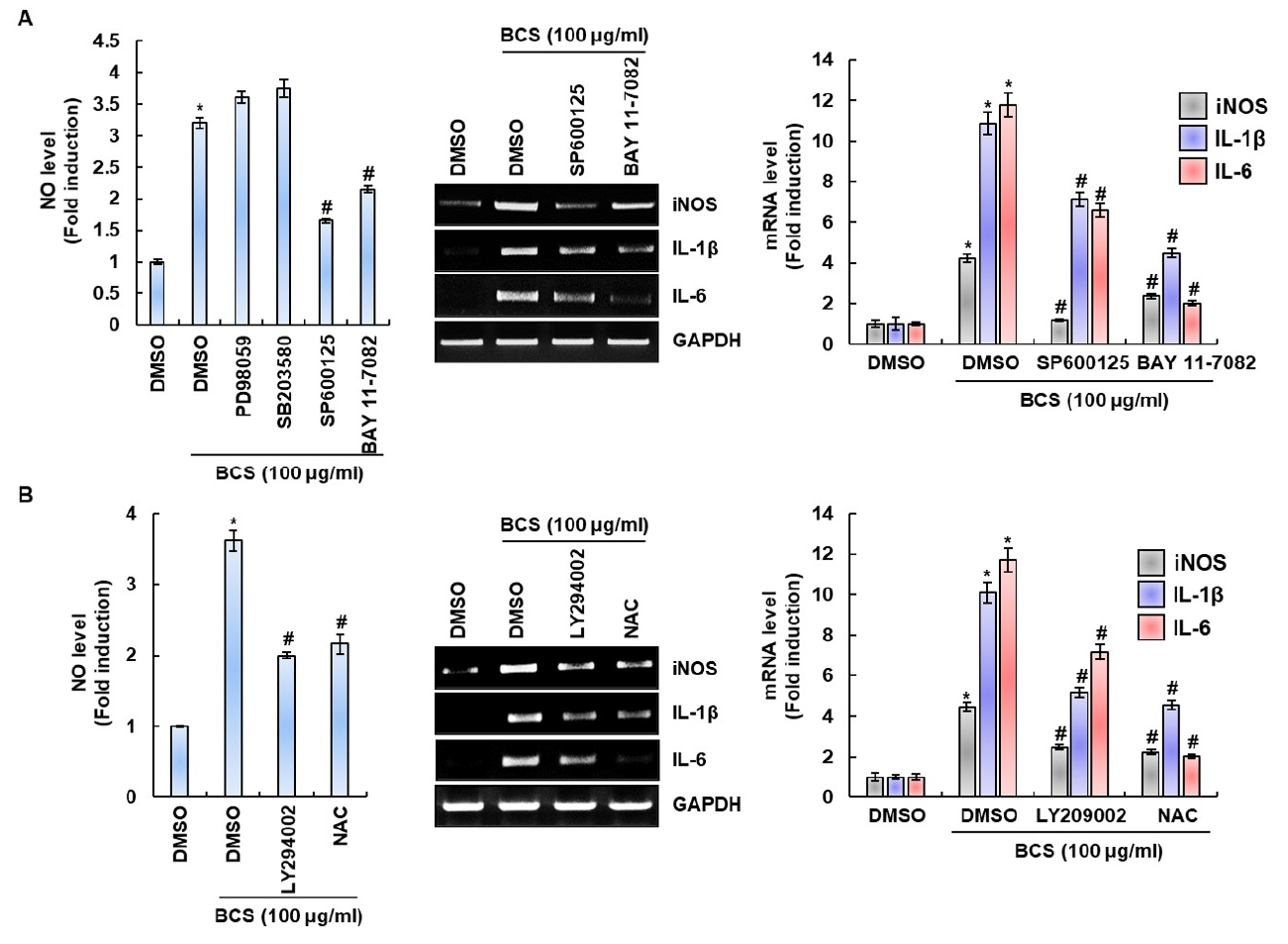
Fig. 4
Effect of MAPK, NF-κB and MAPK signaling on BCS-mediated expression of immunomodulators in RAW264.7 cells. (A, B) The cells were pretreated with PD98059 (20 μM), SB203580 (20 μM), SP600125 (20 μM), BAY 11-7082 (20 μM), LY294002 (20 μM) or NAC (10 mM) for 2 h and then co-treated with BCS for 24 h. For RT-PCR analysis, total RNA was prepared. GAPDH was used as internal control for RT-PCR. *P < 0.05 compared to the cells without the treatment. #P < 0.05 compared to the cells with BCS alone.
Effect of TLR2 on BCS-mediated expression of immunomodulators through the activation of AKT, JNK and NF-κB
The recognition of microbial pathogens and their components by TLRs triggers the activation of intracellular signaling and results in production of immunomediators such as NO, iNOS, IL-1β, IL-6, IL-12 and TNF-α (Chang, 2010; Deng et al., 2016; Kawamoto et al., 2008; Kopf et al., 2010; Liu et al., 2017; Zheng et al., 2017). Thus, we evaluated the effect of TLR2 on BCS-mediated expression of immumodulators. When RAW264.7 cells were treated with BCS (50 and 100 ㎍/mL ml), the mRNA expression of TLR2 were significantly increased (Fig. 5A). As shown in Fig. 5B, TLR2 inhibition by anti-TLR2 dramatically suppressed NO production and the expression of iNOS, IL-1β, IL-6 and TNF-α by BCS in RAW264.7 cells. In addition, we demonstrated that TLR2 inhibition by anti-TLR2 significantly reduced the phosphoylation level of AKT, JNK and IKK (Fig. 5C). These results indicate that TLR2 may be involved in BCS-mediated expression of immunomodulators through the activation of AKT, JNK and NF-κB.
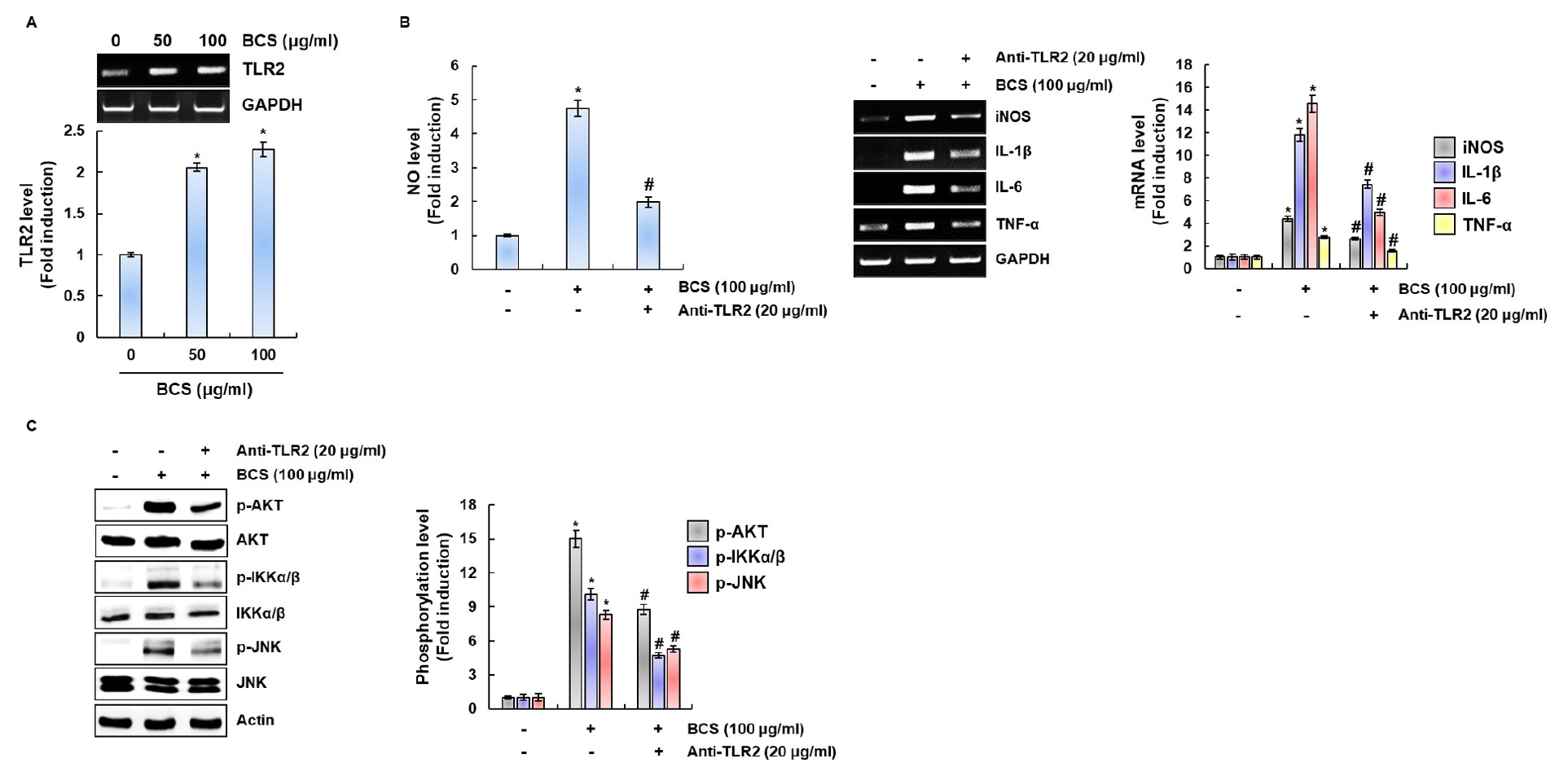
Fig. 5
Effect of TLR2 on BCS-mediated expression of immunomodulators through the activation of AKT, JNK and NF-κB. (A) Gene expression of TLR2 in RAW264.7 cell treated with BCS for 24 h. For RT-PCR analysis, total RNA was prepared. GAPDH was used as internal control for RT-PCR. (B) The cells were pretreated with anti-TLR2 (20 μM) for 2 h and then co-treated with BCS for 24 h. For RT-PCR analysis, total RNA was prepared. GAPDH was used as internal control for RT-PCR. (C) RAW264.7 cells were pretreatment with Anti-TLR2 (20 μM) for 2 h and then co-treated with BCS for 1 h. All cell lysates were subjected to SDS-PAGE and the Western blot was performed using antibodies against AKT, JNK, IKKα/β, p-AKT, p-JNK, p-IKKα/β or actin. Actin was used as internal control for Western blot analysis. *P < 0.05 compared to the cells without the treatment. #P < 0.05 compared to the cells with BCA alone.
Inhibitory effect of BCS on pro-inflammatory mediators and MAPK/NF-κB signaling activation in LPS-stimulated RAW264.7 cells
Excess expression of immunomodulators such as NO, iNOS, IL-1β, IL-6 and TNF-α can damage cells and tissues then resulting in the development of various inflammatory diseases (Billack, 2006; Cho et al., 2014; Jung et al., 2007; Koh and DiPietro, 2011; Lee et al., 2015). As shown in Fig. 6A, excessive NO production was observed in the cell treated with LPS alone. However, NO production by LPS-stimulated RAW264.7cells were markedly inhibited by BCS. In addition, the overexpression of iNOS, IL-1β, IL-6 and TNF-α was observed in the cell treated with LPS alone, while BCS significantly suppressed iNOS, IL-1β, IL-6 and TNF-α expression in LPS-stimulated RAW264.7 cells. Thus, we investigated whether BCS inhibits IκB-α degradation and MAPKs phosphorylation. As shown in Fig. 6B, the treatment of LPS alone markedly increased the phosphorylation of MAPK (ERK1/2, p38 and JNK) and IκB-α degradation. However, the presence of BCS significantly attenuated LPS-mediated phosphorylation of ERK1/2, p38 and JNK. However, BCS similarly induced LPS-mediated IκB-α degradation compared to the cells treated with LPS alone. These data indicate that BCS may inhibit MAPK activation to block the excessive expression of immunomodulators such as NO, iNOS, IL-1β, IL-6 and TNF-α.
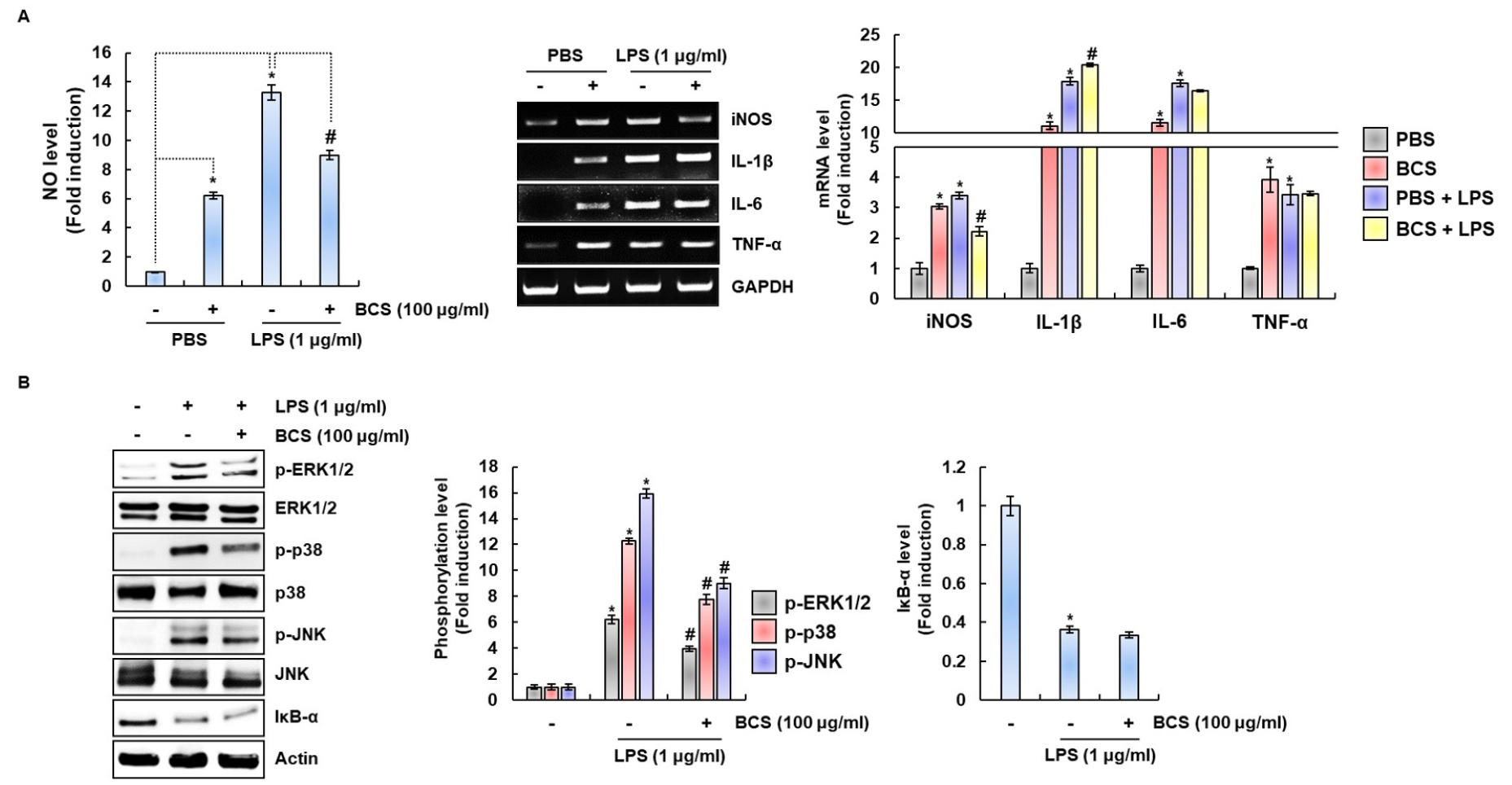
Fig. 6
BCS inhibits the pro-inflammatory mediators in LPS-stimulated RAW264.7 cells. (A) RAW264.7 cells were pretreated with BCS for 6 h and then co-treated with LPS (1 ㎍/mL) for 18h. The determination of NO production was measured by Griess assay. Total RNA was prepared after LPS and BCS treatment. GAPDH was used as internal control for RT-PCR. *P < 0.05 compared to the cells without the treatment, and #P < 0.05 compared to the cells treated with LPS alone. (B) RAW264.7 cells were pretreated with BCS for 6h and then cotreated with LPS (1 ㎍/mL) for 20 min. All cell lysates were subjected to SDS-PAGE and the Western blot was performed using antibodies against p-ERK1/2, p-p38, p-JNK, ERK1/2, p38, JNK, IκB-α or actin. Actin was used as internal control for Western blot analysis.
Discussion
Bok choy is one of the representative crops belonging to Brassicaceae (Kurilich et al., 1999) and is a vegetable that is consumed worldwide but is particularly popular in China and Southeast Asia. Recent reports (Khanam et al., 2012; Neugart et al., 2018) have suggested that brassica vegetables constitute valuable sources of phytochemicals characterized by sulfur-containing glucosinolates and methylcysteinsulfoxide compounds. They contain high levels of vitamins (C, E), minerals (Ca, K), carotenoids, and phenolic compounds (Gioia et al., 2020; Kim and Lee, 2010). Brassica vegetables have been reported to have various physiological activities such as antioxidant, anti-cancer and cardioprotective activity (Kris-Etherton et al., 2002; Maruthanila et al., 2014; Soengas et al., 2011). However, studies on immune-enhancing and anti-inflammatory activitys of bok choy sprout have not been conducted properly. Therefore, in our present study, we investigated immune enhancing activity of BCS in the murine macrophage cell line RAW264.7.
Macrophages have biological function including regulation of inflammation and induction of immunity (Kasimu et al., 2017). Once activated, macrophages secrete a variety of immunomodulators such as NO, iNOS, IL-1β, IL-6, IL-12 and TNF-α (Liu et al., 2017; Shi and Pamer, 2011; Zheng et al., 2017). While proper NO level plays an important role in homeostasis and immunity, excessive NO level is known to cause a variety of human inflammatory diseases (Zou et al., 2018). Thus, NO production have been extensively used both in vitro and in vivo to evaluate immune activation and immunotoxicity (Divate and Chung, 2017). In this study, we found that BCS enhanced the NO production of RAW264.7 macrophages in the absence of LPS. iNOS is the most crucial enzyme to synthesize NO after the activation of macrophages. Our results showed that BCS treatment increased the expression levels of iNOS, which are associated with NO production. LPS is a potent activator of macrophages, which is caused inflammatory response by inducing the release of inflammatory mediators (Gasparrini et al., 2017). We found that BCS was markedly inhibited production of NO and significantly suppressed expression of iNOS in LPS-stimulated RAW264.7 cells. From these results, BCS blocked NO production through downregulation of iNOS expression, which shows that BCS may exert anti-inflammatory activity. Therefore, BCS regulated expression levels of iNOS, which are associated with NO production to coordinate immunomodulatory and anti-inflammatory activity. In addition, we confirmed that the production of immunomodulators by BCS is similar to that by BC. In view of the fact that sprouts have shorter production periods than grown vegetables, and then they can be produced year round, these results show that BCS is highly likely to be used as a agent for boosting immunity.
The MAPK/NF-κB signaling pathway plays a critically important role in the immune system including gene expression, immune response, and cell proliferation (Hayden and Ghosh, 2008; Xi et al., 2011). MAPKs (ERK1/2, p38 and JNK) family is involved in immunomodulatory and inflammatory reactions, which plays an important role in production of IL-1β, IL-6, iNOS, and pro-inflammatory cytokine (Cho et al., 2007; Cho et al., 2017; Fisher et al., 2006; Guo et al., 2015; Hommes et al., 2003; Nakamura et al., 2006; Pearson et al., 2001). Therefore, we explored whether MAPKs (ERK1/2, p38 and JNK) were involved in immunostimulatory and anti-inflammatory activities of BCS in RAW264.7cells. Our results demonstrated that BCS could increase the expression of phosphorylated MAPKs (ERK1/2, p38 and JNK) in normal macrophages. In addition, the enhancement of p-ERK1/2, p-p38 and p-JNK induced by LPS was down-regulated by BCS. These results suggested that BCS may induce the activation of ERK1/2, p38 and JNK.
NF-κB, as an essential transcription factor, has a crucial effect on immune system. IκB-α is phosphorylated by IKK and then dissociated from NF-κB, allowing the NF-κB p65 subunit translocates into the nucleus (Ren et al., 2017). In this study, we found that BCS up-regulated the phosphorylation of IKK as well as the phosphorylation of IκB-α. These results indicate that BCS may activate NF-κB signaling pathway. To elucidate the action point of BCS were investigated AKT, as an upstream factor for NF-κB activation (Karin and Ben-Neriah, 2000; Park et al., 2009). AKT can promote IKK phosphorylation (Ozes et al., 1999) and then result in IκB-α degradation (Park et al., 2009). As demonstrated in result, phosphorylation of AKT was significantly increased after exposure to BCS. Therefore, BCS exerts its immunostimulatory effects through the activation of the MAPK and AKT/NF-κB signaling pathways in RAW264.7 cells. However, BCS similarly induced LPS-mediated IκB-α degradation compared to the cells treated with LPS alone. These data indicate that BCS may inhibit the phosphorylation of MAPK (ERK1/2, p38 and JNK) to block the expression of pro-inflammatory mediators.
The MAPK/NF-κB signaling pathway regulates the production of cytokines related to immunomodulatory (Kopitar-Jerala, 2015). Indeed, many MAPK/NF-κB inhibitors such as PD98059 (ERK1/2 inhibitor), SB203580 (p38 inhibitor), SP600125 (JNK inhibitor) and BAY 11-7082 (NF-κB inhibitor) inhibits the expressions of pro-inflammatory mediators such as iNOS, COX-2, TNF-α and IL-1β (Chen et al., 1999; Nieminen et al., 2006; Seo and Jeong, 2020; Shan et al., 2010). As a result, JNK by SP600125, NF-κB by BAY 11-7082, AKT by LY294002 and ROS by NAC was found to significantly down regulate the BCS-induced NO, iNOS, IL-1β and IL-6 generation. These results indicate that BCS-mediated expression of immunomodulators may be dependent on AKT, JNK and NF-κB.
Toll-like receptors (TLRs) can recognizes distinct pathogen- associated molecular patterns (PAMPs) from bacteria, viruses, fungi and parasites (Kawai and Akira, 2010). TLR2, as the most studied TLRs, that recognizes a variety of lipoproteins, lipopeptides, peptidoglycans and lipoteichoic acid et al (Kang et al., 2009). The recognition of microbial pathogens and their components by TLRs triggers the activation of intracellular signaling and results in production of immunomediators such as NO, IL-1β, IL-6, IL-12 and TNF-α (Chang, 2010; Deng et al., 2016; Kawamoto et al., 2008; Kopf et al., 2010; Liu et al., 2017; Zheng et al., 2017). So we further hypothesized that TLR2 may be involved on BCS-stimulated expression of immumodulators. When RAW264.7 cells were treated BCS, the mRNA expression of TLR2 was significantly increased, TLR2 inhibition by anti-TLR2 dramatically suppressed NO production and the expression iNOS, IL-1β, IL-6 and TNF-α. In addition, we demonstrated that TLR2 inhibition by anti-TLR2 significantly reduced the phosphoylation level of AKT, JNK and NF-κB. AKT, a serine/threonine kinase and an upstream factor for NF-κB activation, can promote IκB kinase phosphorylation (Ozes et al., 1999) and then resultin IκB degradation (Park et al., 2009). On the basis of our results, BCS could induce NF-κB activation via AKT, IKK and JNK phosphorylation. Our results indicate that TLR2 may be involved in BCS-mediated expression of immunomodulators through the activation of AKT, JNK and NF-κB.
In conclusion, summarizing the our results, BCS increased the production of immunomodulators through TLR2-dependent activation of JNK, NF-κB and AKT signaling in RAW264.7 cells, which indicates that BCS may contribute to enhancing the immune function through macrophage activation. Thus, BSC can be used as an important natural product for the development of immune-enhancing agents in the future. However, it is considered that studies on the production conditions of BCS which can increase the immune enhancing activity of BCS.





