Introduction
Materials and Methods
Plant material
Test Microorganisms
Antibiotics
Minimum Inhibitory Concentration (MIC)
Checkerboard dilution test
Colorimetric assay using MTT test
Time-kill curve assay
Statistical analysis
Results
Discussion
Introduction
Methicillin-resistant Staphylococcus aureus (MRSA) has been reported since 1961 and is one of the most significant pathogens causing infections today. Due to its resistance to almost all antibiotics, treatment options are limited (Cha et al., 2015; Ji et al., 2007). MRSA is characterized by being resistant to antibiotics and is not easily treated, mechanisms of resistance to antibiotics have been reported according to various antibiotics (Lee et al., 2017). Bacteria quickly acquire resistance to them and neutralize antibiotics (Eum and Park, 2007). Therefore, the antibacterial activity of Chamaecyparis obtusa (Siebold & Zucc.) Endl. was studied in order to find natural products with antibacterial activity. The main purpose of this study is to investigate the in vitro effect on MRSA. Chamaecyparis obtusa (Siebold & Zucc.) Endl. of Cupressaceae is native to Taiwan, Japan, and is native to Jeju Island and southern regions (Jung et al., 2012; Yang et al., 2017). The strong scent of Chamaecyparis obtusa (Siebold & Zucc.) Endl. is known to be effective for sterilization, deodorization, blood circulation for skin beauty, colds, etc., and the major components are found to be monoterpene and sesquiterpene (Jung et al., 2012). In addition, Chamaecyparis obtusa (Siebold & Zucc.) Endl. leaf essential oil has various physiological activities such as anti-inflammatory, antibacterial, antioxidant, deodorizing, insecticidal and neurostable (Yang et al., 2017). However, while numerous studies have investigated the biological activities of C. obtusa leaves, little attention has been paid to the antibacterial potential of its wood. The heartwood of Chamaecyparis obtusa contains a large amount of hinokinin, a dibenzylbutyrolactone lignan (Ohashi et al., 1987; Takaku et al., 2001). A study by Bajpai et al. (2014) showed that essential oil extracts from Chamaecyparis obtusa sawdust exhibit antibacterial activity against food-borne bacteria and can be utilized as a natural antibacterial agent due to their cell membrane-destructive activity. Based on these findings, it can be hypothesized that the wood of Chamaecyparis obtusa may also possess antibacterial activity. This study presents a study demonstrating the antimicrobial activity and synergistic effect of Chamaecyparis obtusa (Siebold & Zucc.) Endl. on MRSA and methicillin-sensitive (MSSA) strains.
Materials and Methods
Plant material
In 2019, the wood of Chamaecyparis obtusa (Siebold & Zucc.) was purchased from a cypress woodworking shop in Jangseong-gun, Korea, and taxonomic identification was performed by Jeong Dae-gi and Shin Dong-won. Chamaecyparis obtusa (Siebold & Zucc.) Endl. xylem was cut to an appropriate size and dried. 208.010 g of xylem was extracted with 2 L of ethanol for 3 h. The extract was filtered (Adbantec No. 1, Kashiwa-si, Japan) and concentrated under reduced pressure to obtain an extract. The extract was dissolved in dimethyl sulfoxide (DMSO, Sigma Chemical Company, St. Louis, Missouri) and used in the experiments.
Test Microorganisms
Five Clinical isolates (MRSA) were obtained from five different patients at Wonkwang University Hospital (Iksan, South Korea). The other 2 strains were S. aureus ATCC 33591 and S. aureus ATCC 25923. Before use, all of the bacteria were cultured in Mueller-Hinton Broth (MBH) and Mueller-Hinton Agar (MHA) (Difco Laboratories, Baltimore, MD, USA). The bacteria were suspended in Mueller-Hinton Broth and the incubated at 36.5℃ for 24 hr.
Antibiotics
Ampicillin (AM), Oxacillin (OX) and Vancomycin (VC) (Sigma Chemical Co. St. Louis, M0, USA) were used.
Minimum Inhibitory Concentration (MIC)
The minimum Inhibitory Concentration (MIC) was determined using the broth microdilution method according to the clinical and Laboratory standards Institute guideline (CLSI., 2000). Briefly, a preparation of the microorganisms inoculated were done on 24 hr Broth cultures, and the suspensions were adjusted to a 0.5 McFarland standard turbidity (approximately 1.5× 10⁸ CFU/mL). Final inoculums were adjusted to the 1.5×10⁶ CFU/mL. These serially diluted cultures were then incubated at 36.5℃ for 24 hr. MIC was defined at the lowest concentration of AM, OX, VC, Chamaecyparis obtusa (Siebold & Zucc.) Endl. extracts. At the end of the incubation period, the well plates were visually examined for turbidity. Cloudiness indicates that bacterial growth has not been inhibited by the concentration of antimicrobial agents contained in the medium. A colorimetric assay for rapid detection of the presence of bacteria was also performed (see below, Colorimetric assay using 3-4,5-dimethylthiazol-2- yl)-2,5-diphenyl tetrazolium bromide [MTT] test).
Checkerboard dilution test
The synergistic combinations were investigated in the preliminary checkerboard method performed using the MRSA, MSSA and the five isolate strains came from fourteen patients via MIC determination, according to the CLSI guidelines (Mazumdar et al., 2005). The MIC was defined as the lowest concentration of drug alone or in combination that inhibited the visible growth. The in vitro interaction was quantified by determining the fractional inhibitory concentration (FIC). The FIC index was calculated as follows: FIC = (MIC of drug A in combination/MIC of drug A alone) + (MIC of drug B in combination/MIC of drug B alone). FIC indices (FICI) were interpreted as follows: <0.5, synergy; 0.5-0.75, partial synergy; 0.76-1.0, additive effect; >1.0-4.0, indifference; and >4.0, antagonism. All experiments were independently repeated three times.
Colorimetric assay using MTT test
A colorimetric assay based on MTT for rapid detection of the presence of bacteria was performed as previously described (Joung et al., 2015; Luis et al., 2014; Shi et al., 2008). Briefly, a stock solution of 5 ㎎/mL MTT (Sigma) was prepared in phosphate-buffered saline and kept at -70℃. A final concentration of 1 ㎎/mL of MTT was used in the assay. After 24hrs of incubation a 36.5℃, 20 μL of the yellow MTT was added to the 96-well microtiter plate and incubated for an additional 20 min. The presence of a blue color indicates the presence of bacteria. The color change was evaluated visually, and no absorbance-based quantification at OD570 ㎚ was performed.
Time-kill curve assay
A time-kill curve assay was performed as previously described (Chang et al., 1995) to study the effects of antimicrobial agent concentration on bacterial growth over time. To explain whether the antibacterial activity of EEC was associated with altered membrane permeability or the action of MDR (multidrug resistant) pumps, we examined the antibacterial activity of EEC in the presence or absence of ampicillin (AM) or oxacillin (OX) or vancomycin (VC), respectively. A standard inoculum of 1.5×10⁶ CFU/mL was used. Chamaecyparis obtusa (Siebold & Zucc.) Endl. was used with antibiotics. A test plate containing only MHB plus inoculum served as the control. The viable strain was performed at 36.5°C. for 48 hrs at OD 600 ㎚. Checked by time at OD 600 ㎚. All experiments were conducted in triplicate.
Statistical analysis
All experiments were performed in triplicate. The data obtained from the experiments were expressed as the mean ± SEM. Dunnett’s t-test was used for multiple comparisons. P-values <0.01 were considered to indicate a statistically significant difference.
Results
The ethanol extract had a MIC of 250 ㎍/mL for S. aureus ATCC 33591 and a 250 ㎍/mL MIC for S. aureus ATCC 25923 (Table 1 and 2). Chamaecyparis obtusa (Siebold & Zucc.) Endl. xylem extract showed antibacterial activity against MRSA and MSSA strains. The FICI values of EEC+AM and EEC+OX were 0.02-0.09 and 0.01-0.09, respectively, indicating synergistic effect (Table 2 and 3). In addition, the FICI value of EEC+VC ranged from 0.07 to 0.25, also indicating a synergistic effect (Table 4).
Table 1.
The S. aureus strains used in the experiments.
| S. aureus strains | Class | MecA gene | β-Lactamas acativity | Antibiotic resistance pattern |
| ATCC 33591 | MRSA | +y | + | AMx, OXx |
| ATCC 25923 | MSSA | -y | - | - |
| Clinical isolates | ||||
| DPS -1z | MRSA | + | + | AM, OX |
| DPS -2 | MRSA | + | - | AM, OX |
| DPS -3 | MRSA | + | + | AM, OX |
| DPS -4 | MRSA | + | - | AM, OX |
| DPS -5 | MRSA | + | - | AM, OX |
Table 2.
Result of the combined effect of ethanol extract of Chamaecyparis obtusa (Siebold & Zucc.) Endl. Xylem and AM agains S. aureus.
| MICs (㎍/mL) | |||||
| S. aureus strain | yEEC Alone | With AMx | AM Alone | With EEC | wFICI |
| ATCC 33591 | 250 | 3.9 | 500 | 3.9 | 0.02 |
| ATCC 25923 | 250 | 7.8 | 7.8 | 0.48 | 0.09 |
| DPS -1z | 250 | 7.8 | 1000 | 31.25 | 0.06 |
| DPS -2 | 250 | 3.9 | 500 | 3.9 | 0.02 |
| DPS -3 | 250 | 3.9 | 125 | 1.9 | 0.03 |
| DPS -4 | 250 | 3.9 | 125 | 1.9 | 0.03 |
| DPS -5 | 125 | 3.9 | 62.5 | 1.9 | 0.06 |
Table 3.
Result of the combined effect of ethanol extract of Chamaecyparis obtusa (Siebold & Zucc.) Endl. Xylem and OX agains S. aureus.
| MICs (㎍/mL) | |||||
| S. aureus strain | yEEC Alone | With OXx | OX Alone | With EEC | wFICI |
| ATCC 33591 | 250 | 1.9 | 500 | 3.9 | 0.01 |
| ATCC 25923 | 250 | 3.9 | 15.62 | 0.48 | 0.04 |
| DPS -1z | 250 | 7.8 | 62.5 | 3.9 | 0.09 |
| DPS -2 | 250 | 3.9 | 250 | 3.9 | 0.03 |
| DPS -3 | 250 | 1.9 | 500 | 7.8 | 0.02 |
| DPS -4 | 250 | 3.9 | 500 | 3.9 | 0.03 |
| DPS -5 | 125 | 1.9 | 62.5 | 0.97 | 0.03 |
Table 4.
Result of the combined effect of ethanol extract of Chamaecyparis obtusa (Siebold & Zucc.) Endl. Xylem and VC agains S. aureus.
| MICs (㎍/mL) | |||||
| S. aureus strain | yEEC Alone | With VCx | VC Alone | With EEC | wFICI |
| ATCC 33591 | 250 | 1.9 | 3.9 | 0.48 | 0.13 |
| ATCC 25923 | 250 | 1.9 | 7.8 | 0.48 | 0.07 |
| DPS -1z | 250 | 1.9 | 3.9 | 0.48 | 0.13 |
| DPS -2 | 250 | 1.9 | 7.8 | 0.48 | 0.13 |
| DPS -3 | 250 | 7.8 | 7.8 | 0.97 | 0.15 |
| DPS -4 | 250 | 1.95 | 7.8 | 0.97 | 0.13 |
Time-kill assays were conducted to evaluate the time- dependent synergistic effects of Chamaecyparis obtusa (Siebold & Zucc.) Endl. extract and antibiotics. The control group did not show any reduction in OD at 600 ㎚. In contrast, the combined treatment with EEC and antibiotics resulted in a reduction of bacterial counts (Fig. 1 and 2). In MRSA strains, the combinations of 1/4 EEC with AM, OX, and VC exhibited bacterial reduction rates of 93.29%, 106.76%, and 104.03%, respectively, which were more than twofold higher than those observed with the 1/2 EEC combinations. In MSSA strains, the combinations of 1/2 EEC with AM, OX, and VC showed bacterial reduction rates of 49.01%, 51.61%, and 54.60%, respectively, whereas the combinations of 1/4 EEC with these antibiotics showed a decreasing trend up to 36 h but an increase at 48 h.
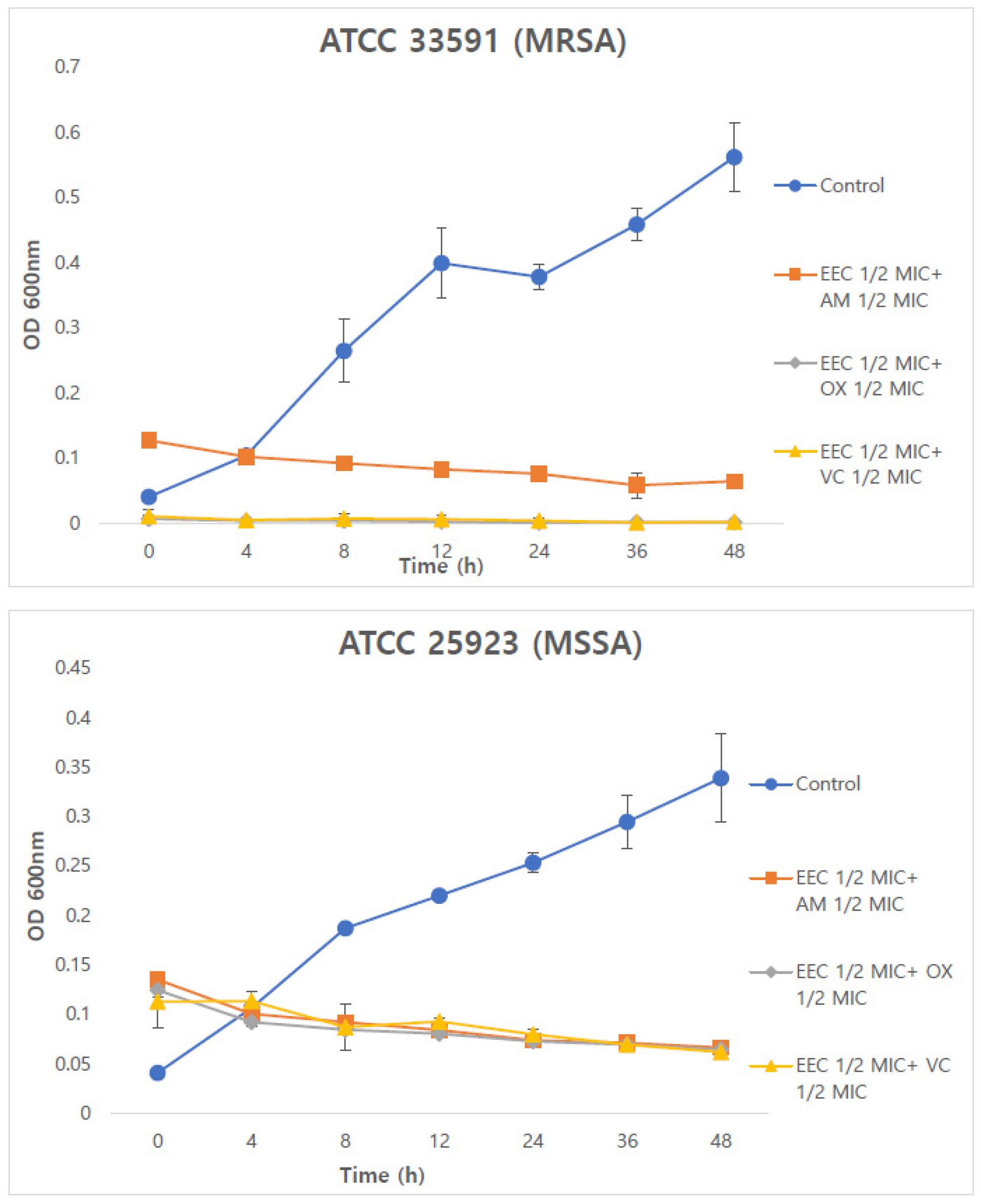
Fig. 1.
Time-kill curve of combined 1/2 EEC and 1/2 AM, OX, VC against MRSA and MSSA strains. Data represent the OD values from three independent experiments (n=3). EEC; ethanol extract of Chamaecyparis obtusa (Siebold & Zucc.) Endl. xylem; AM. Ampicillin; OX, oxacillin; VC, vancomycin; MRSA, methicillin-resistant Staphylococcus aureus; MSSA, methicillin-sensitive Staphylococcus aureus; MIC, minimum inhibitory concentration. * p<0.01 compared with AM, OX or VC alone (Dunnett’s t-test).
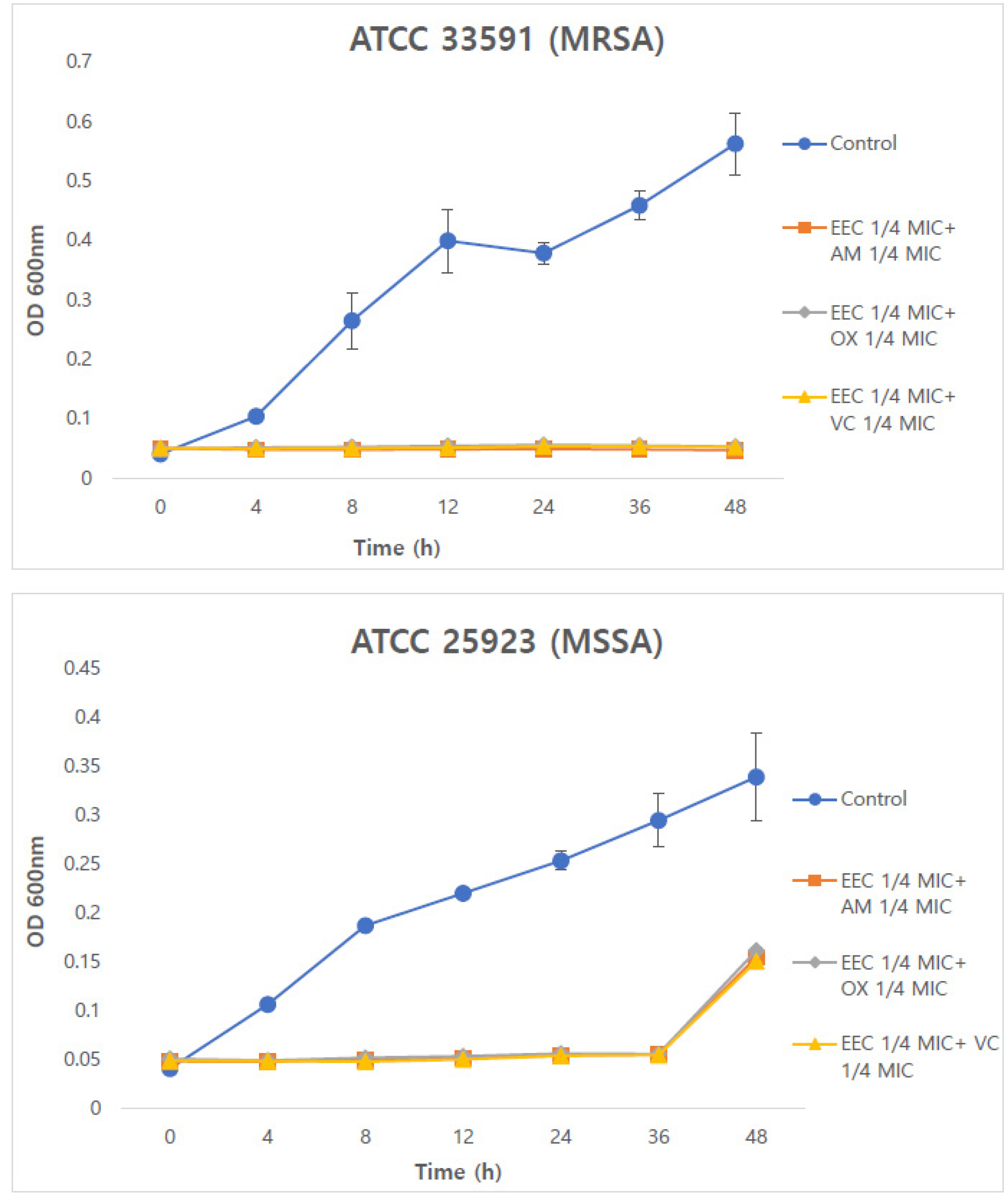
Fig. 2.
Time-kill curve of combined 1/4 COX and 1/4 AM, OX, VC against MRSA and MSSA strains. Data represent the OD values from three independent experiments (n=3). EEC; ethanol extract of Chamaecyparis obtusa (Siebold & Zucc.) Endl. xylem; AM, ampicillin; OX, oxacillin; VC, vancomycin; MRSA, methicillin-resistant Staphylococcus aureus; MSSA, methicillin-sensitive Staphylococcus aureus; MIC, minimum inhibitory concentration. * p<0.01 compared with AM, OX or VC alone (Dunnett’s t-test).
Discussion
Methicillin-resistant Staphylococcus aureus (MRSA) is a bacterium that causes high mortality by causing pneumonia, skin infection, soft tissue infection and sepsis, and is one of the representative pathogen-causing bacteria (Cha et al., 2015; Lee et al., 2007). Although vancomycin has been developed and used in the treatment of MRSA, vancomycin- resistant S. aureus (VRSA) bacteria were first found in Japan in 1996, and then in many countries around the world, vancomycin-resistant S. aureus bacteria are reported (Eum and Park, 2007; Shin and Seong, 2006). Recently, the emergence of super bacteria such as vancomycin-resistant Staphylococcus aureus (VRSA) has seriously raised the problem of antibiotic resistance (Cha et al., 2015). Combination therapy is the most commonly recommended empirical treatment for bacterial infections in intensive care units (Joung et al., 2018). The purpose of this study is to investigate combination therapy for MRSA strains and MSSA strains to find solutions due to antibiotic resistance problems. The Chamaecyparis obtusa (Siebold & Zucc.) Endl. xylem ethanol extract had MICs ranging from 125 ㎍/mL to 250 ㎍/mL against MRSA and MSSA strains, and the checkerboard dilution test was conducted to confirm synergies with AM, OX and VC for both variants as well as EEC alone action. When tested against ATCC 33591 and ATCC 25923, this data showed that EEC alone had a moderate inhibitory effect on the growth of MRSA strains. But with AM, OX and VC, EEC showed very effective antibacterial activity in FICI. Similar effects were also observed in MSSA strain. These results showed that EEC in combination with antibiotics could effectively inhibit MRSA growth. The time-kill curve experiments performed at OD 600 ㎚ for 0–48 h demonstrated that EEC enhanced the antibacterial effects of antibiotics, contributing to the reduction of MRSA and MSSA strain counts. In particular, the combination of 1/4 EEC with 1/4 OX and VC showed the most pronounced reduction effect in MRSA strains. These findings suggest that EEC has potential as an adjunctive therapeutic agent against MRSA and MSSA strains and indicate the possibility of reducing the required dosage of conventional antibiotics. Therefore, EEC provides meaningful evidence for its application as a natural antimicrobial candidate that may reduce antibiotic-associated side effects while complementing or partially substituting existing antibiotics.




