Introduction
Materials and Methods
Cell culture
Preparation of Alnus japonica and Portulaca oleracea extract complex
MTT assay
DPPH
Catalase activity
CYP2E1 activity
Experiment animal
Liver Injury Mouse Model
Blood analysis
Statistical analysis
Results
Screening of natural products for improving alcoholic liver damage
Anti-oxidation activity
Evaluation of animal efficacy in improving against acute alcoholic liver damage
Animal efficacy evaluation for improving against chronic alcoholic liver damage
Discussion
Introduction
The liver is the largest organ of the human body. It plays an essential role in carbohydrate metabolism, amino acid and protein metabolism, fat metabolism, bile acid and bilirubin metabolism, vitamin and mineral metabolism, hormone metabolism, detoxification, and anti-microbial activity. Liver disease occurs when hepatocytes are damaged by various causes, such as continuous consumption of large amounts of alcohol, viruses, and drug abuse (Albano, 2002; Lee and Friedman, 2011; Lim et al., 1999). If it is not cured in the early stage, it progresses chronically and eventually goes to cirrhosis or liver cancer through a fibrosis process. Alcoholic liver disease is a diverse group of liver diseases caused by alcohol consumption. According to the World Health Organization (WHO), alcohol deaths are about 1.8 million people yearly (WHO, 2014; 2011). In Korea, liver disease is the leading cause of adult death, following cancer, heart disease, cerebrovascular disease, pneumonia, and diabetes. In particular, in the 40s, who are leading the industry, liver disease caused by alcohol is ranked third. The alcohol-related mortality rate was 6.8 times higher in males (16.3) than in females (2.4), but the gap between men and women was decreasing (11 times in 2005). Alcohol-related deaths surged from the age of 30, indicating that the 50s peaked (Kim, 2009).
Efforts by academia and the pharmaceutical industry to discover liver-protective active substances applicable in the early stages of liver disease are continuing, and silymarin isolated from the Thistle family is known as an active substance that protects the liver and is used for treatment with antiviral drugs (Lieber, 2004). Recently, as interest in alternative medicine that compromised the side effects of pharmaceuticals and food safety at home and abroad has been amplified, research to find a solution from the natural product is increasing (Chayanupatkul and Liangpunsakul, 2014).
Functional raw materials extracted from natural plants have the advantage of having little cost and time in the development process, as their safety and effectiveness have been proven by long-time experience (Kim et al., 2020; Lee et al., 2000b; Lu and Mato, 2008). Portulaca oleracea (commonly purslane) is a perennial plant belonging to the purslane family and has strong vitality (Uddin et al., 2014). It contains many inorganic salts, such as calcium oxalate and dopamine. It is used as a detoxification and diuretic drug in Korea and China and for dry stomach, asthma, and cystitis in the West (Qiao et al., 2019). Alnus japonica bark and heartwood extract are anti-inflammatory, and pharmacology such as analgesic action, gastritis, gastric ulcer treatment effect, antioxidant action, etc., and enemy effect is known (An et al., 1999; Kim et al., 2004; Kim, 2003; Lee et al., 2000a; Farkhondeh and Samarghandian, 2019; Tung et al., 2010). However, the improvement effect of the Portulaca oleracea and Alnus japonica extract composites on the protection of alcoholic liver damage has not been reported.
In this study, we verified the efficacy of acute and chronic alcoholic liver damage-improving animal efficacy assessments with P. oleracea and A. japonica extracts that help to recover liver cells damaged by alcohol consumption. In addition, by measuring the antioxidant activity of natural products, we intend to verify the effect of improving the liver damage of natural products composites.
Materials and Methods
Cell culture
HepG2 and Chang liver cells confirmed the protective effect against liver damage. The cell was cultured in Dulbecco's modified Eagle's medium supplemented with l0% heat-inactivated fetal bovine serum (FBS) containing 100 U/mL penicillin and 100 ug/mL streptomycin. Cells were incubated at 37℃ in a humidified atmosphere with 5% CO2. The cell lines used for in vitro assay were purchased from American Type Culture Collection (ATCC; Manassas, VA, USA).
Preparation of Alnus japonica and Portulaca oleracea extract complex
Dried Portulaca oleracea and Alnus japonica were purchased from human herbs (Daegu, Korea). The APEC were extracted with 50% ethanol for 18 h at 50℃. After filtration, the extracts were concentrated under reduced pressure in a rotary evaporator and dried. The extracts were kept at a temperature of -20℃ until they were needed.
MTT assay
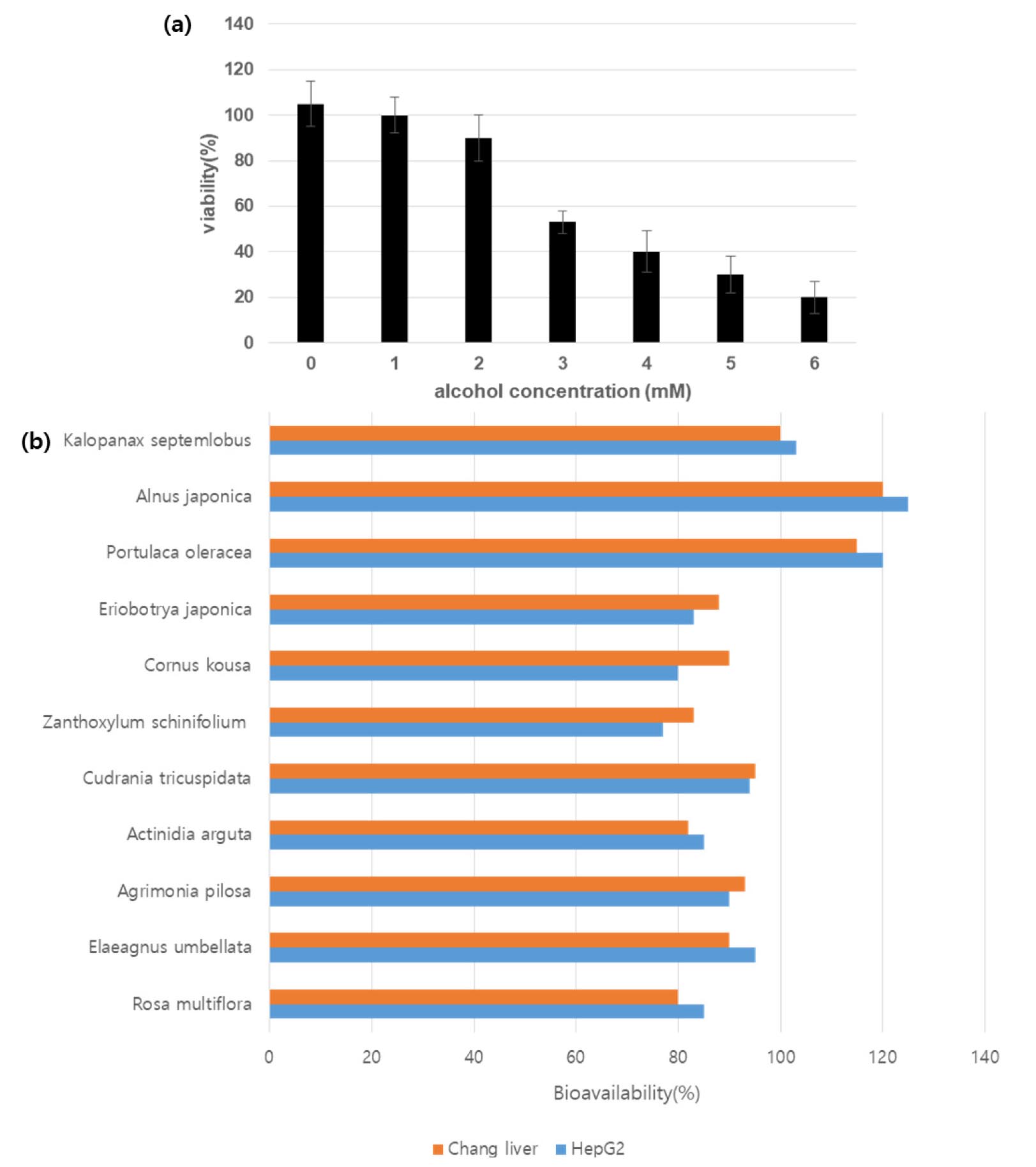
To confirm the cell viability using the MTT method. Briefly, the HepG2 and Chang liver cells were seeded into a 96-well plate at 1.0 x 104 cells/well density and incubated at 37℃ in a humidified atmosphere with 5% CO2 for 24 h. The sample was treated with various concentrations for 48 h. After incubation, the medium was removed and MTT solution was added (500 ㎍/mL of MTT in culture medium) for 4 h. The MTT solution was replaced with 200 μL of DMSO solution and measured absorbance at 570 ㎚ by a microplate reader (Biotek, Winooski, VT, USA).
DPPH
To evaluate the antioxidant activities of Portulaca oleracea and Alnus japonica extracts were obtained by the DPPH assay. The samples were diluted with ethanol (0.5 mL), and the model was mixed with 100 mM sodium acetate (0.25 mL, pH 5.5). An ethanol solution (0.25 mL) of 2,2-diphenyl-1-picrylhydrazyl (DPPH, 200 ㎍/mL) was added to the mixture. The reaction mixture was measured at 517 ㎚ using a microplate reader (Biotek, Winooski, VT, USA). The following formula calculated the DPPH scavenging activity:
where Ac0 equals the absorbance of the control with DPPH only; Ac1, the absorbance of the control without DPPH; As0 the absorbance in the presence of sample with DPPH; and As1, the absorbance in the presence of sample without DPPH.
Catalase activity
As for the catalase activity (CAT), since hydrogen peroxide shows maximum absorbance at 240 ㎚, the amount of hydrogen peroxide consumed by CAT was measured using an absorbance meter. After putting 1.3 mL of substrate (25 mM H2O2 in 50 mM phosphate buffer, pH 7.0) into a cuvette, 20 μL of the sample was added and the absorbance decrease rate was immediately measured at 240 ㎚ to calculate activity. The molar extinction coefficient of hydrogen peroxide at this time was calculated by converting it to 40 μM-1cm-1.
CYP2E1 activity
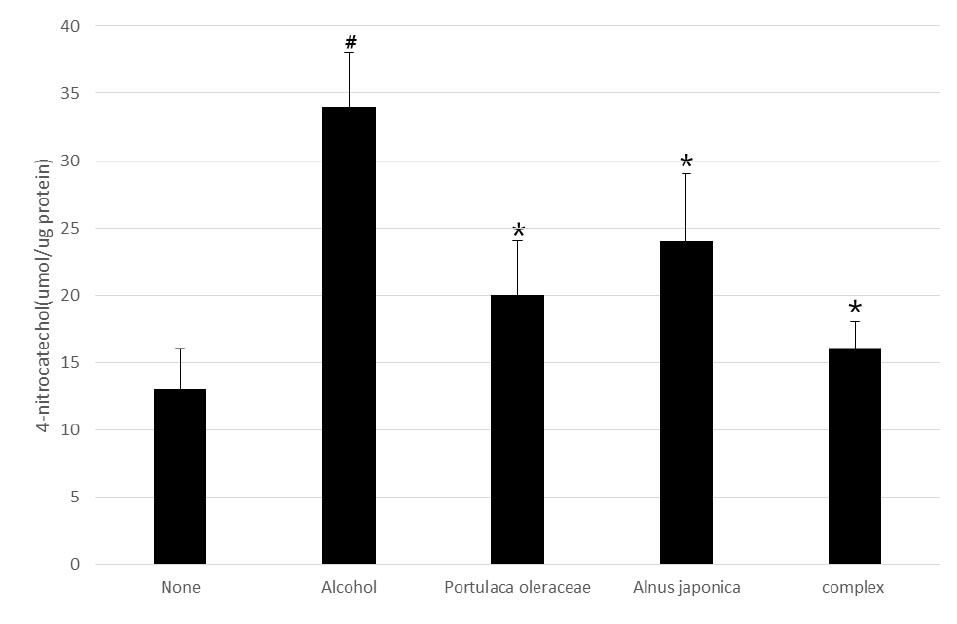
The liver tissue was homogenized by adding 4-fold potassium phosphate buffer (KPB, pH 7.4) and centrifuged at 3,000 rpm for 10 minutes (Labogene, Seoul, Korea) to obtain a supernatant, where measured the CYP2E1 enzyme activity. CYP2E1 enzyme activity was measured using a specific substrate, p-nitrophenol (PNP). The reaction was initiated by adding 0.1 M KPB (pH 7.4), 1 mM PNP, and 0.2 ㎎ liver homogenate to a final concentration of 0.5 mM NADPH. After reacting at 37℃ for 30 minutes, it was terminated by adding 0.2 mL of 20% trichloroacetic acid (TCA). Added 0.5 mL of 2 M NaOH to the supernatant obtained by centrifugation at 10,000 rpm for 5 minutes to develop color, and measured the shade at 535 ㎚ (BioTek Instruments, Inc.), and the standard curve used 4-nitrocatechol, the product of this reaction.
Experiment animal
For the development of acute liver injury and chronic liver injury mouse models, 7-week-old C57BL/6 male mice were purchased from Dooyul Biotech (Seoul, Korea). All experimental animals were maintained under the following conditions: 24 ± 2℃; 50 ± 10% relative humidity; 12-h light and dark cycle. After treatment, the mice were sacrificed under anesthesia. Serum and liver were collected and stored at -80℃ before use. The approved experimental procedures by the Ethics Review Committee of the hy Company Limited R&D Center, Korea (AEC-2018-00054-Y).
Liver Injury Mouse Model
The liver injury mouse model was divided into acute and chronic. The 7-week-old mice were divided into eight animals per group. The nine groups used in the experiment were divided according to body weight. Normal group; Ethanol treated group; Positive control group; 25 ㎎/㎏ of Alnus japonica extract treated ethanol group; 50 ㎎/㎏ of Alnus japonica extract treated ethanol group; 25 ㎎/㎏ of Portulaca oleracea extract treated ethanol group; 50 ㎎/㎏ of Portulaca oleracea extract treated ethanol group; Combination of 25 ㎎/㎏ of Alnus japonica extract and 25 ㎎/㎏ of Portulaca oleracea extract treated ethanol group; Combination of 50 ㎎/㎏ of Alnus japonica extract and 50 ㎎/㎏ of Portulaca oleracea extract treated ethanol group. Alnus japonica and Portulaca oleracea extracts were mixed with ethanol and orally administered. Three ㎎/㎏ of ethanol induces chronic liver injury for four weeks. Treat 5 ㎎/㎏ of ethanol for only one week to cause acute liver injury. Hovenia dulcis extract was used for positive control.
Blood analysis
To confirm catalase (CAT) activity, serum was analyzed using OxiSelect Catalase Activity Assay Kit (Cell Bio, San Diego, CA, USA). The markers of liver injury, such as aspartate aminotransferase (AST), alanine aminotransferase, lactate dehydrogenase (LDH), and albumin (ALB), were performed using TBA-40FR (Toshiba, Tokyo). The results are shown in U/mL.
Statistical analysis
All results are expressed as three independent experiments' mean ±standard deviation (SD). The ANOVA test evaluates the significant difference between means using the statistical analysis software package version 9.2 (SAS Institute, Cary, NC, USA).
Results
Screening of natural products for improving alcoholic liver damage
To screen a functional material that can effectively respond to alcohol-induced liver damage, IC50 was confirmed using HepG2 cells (Fig. 1). The hepatocellular protective effect against alcohol was confirmed under the IC50 condition for 11 natural product extracts selected using the natural product library of hy Co. Ltd. (Fig. 1). Among them, it was confirmed that K. septemlobus, A. japonica, and purslane extracts exhibited the highly cytoprotective effect against alcohol.
Anti-oxidation activity
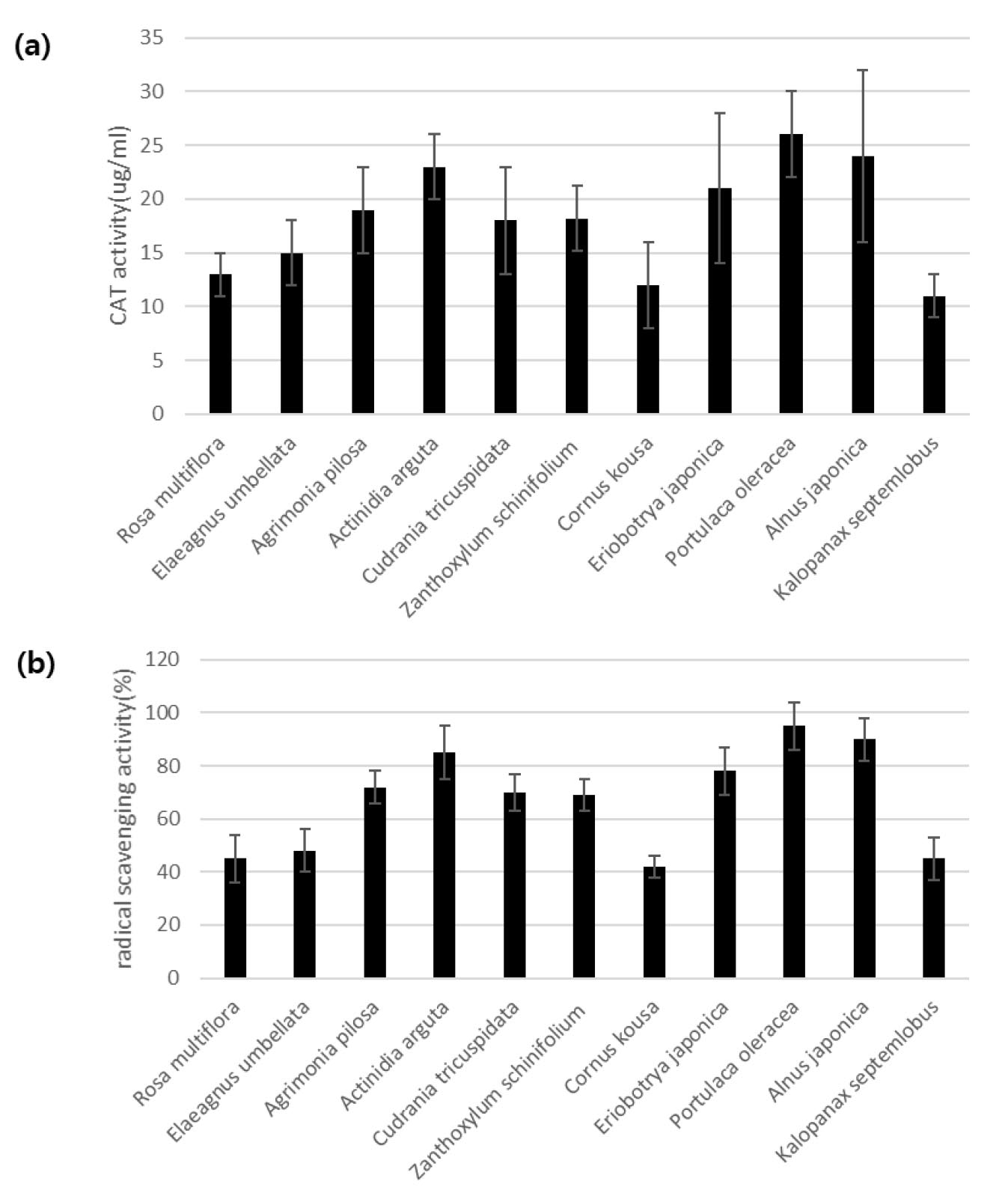
DPPH radical scavenging ability and catalase (CAT) analysis of selected A. japonica, Portulaca extracts, and complexes were performed. In the case of DPPH activity, Alnus showed 1.9 times more antioxidant activity than Portulaca, and in the complex, 2.2 folds more than Portulaca and 20% more activity than Alnus (Fig. 2A). In addition, CAT activity was measured, and it was confirmed that the CAT activity of A. japonica and P. oleracea extracts showed better cat activity when mixed than when the two materials were contained separately (Fig. 2B). In addition, the analysis of total polyphenols and fatty acids widely distributed in plant-based natural products and showing various physiologically active effects confirmed that A. japonica contains 5.25 ± 1.23 ㎎/ml, P. oleracea 5.47 ± 0.93 ㎎/ml and the A. japonica contains 106.7 ㎎/g, which is about three times more fatty acids than the P. oleracea (Table 1).
Table 1.
Analysis of total polyphenol and fatty acids contents in A. japonica and P. oleracea
| Total polyphenol (㎎/ml) | Fatty acids (㎎/100g) | |
| P. oleracea | 5.47 ± 0.93z | 3.165 ± 0.846 |
| A. japonica | 5.25 ± 1.23 | 10.67 ± 2.84 |
Evaluation of animal efficacy in improving against acute alcoholic liver damage
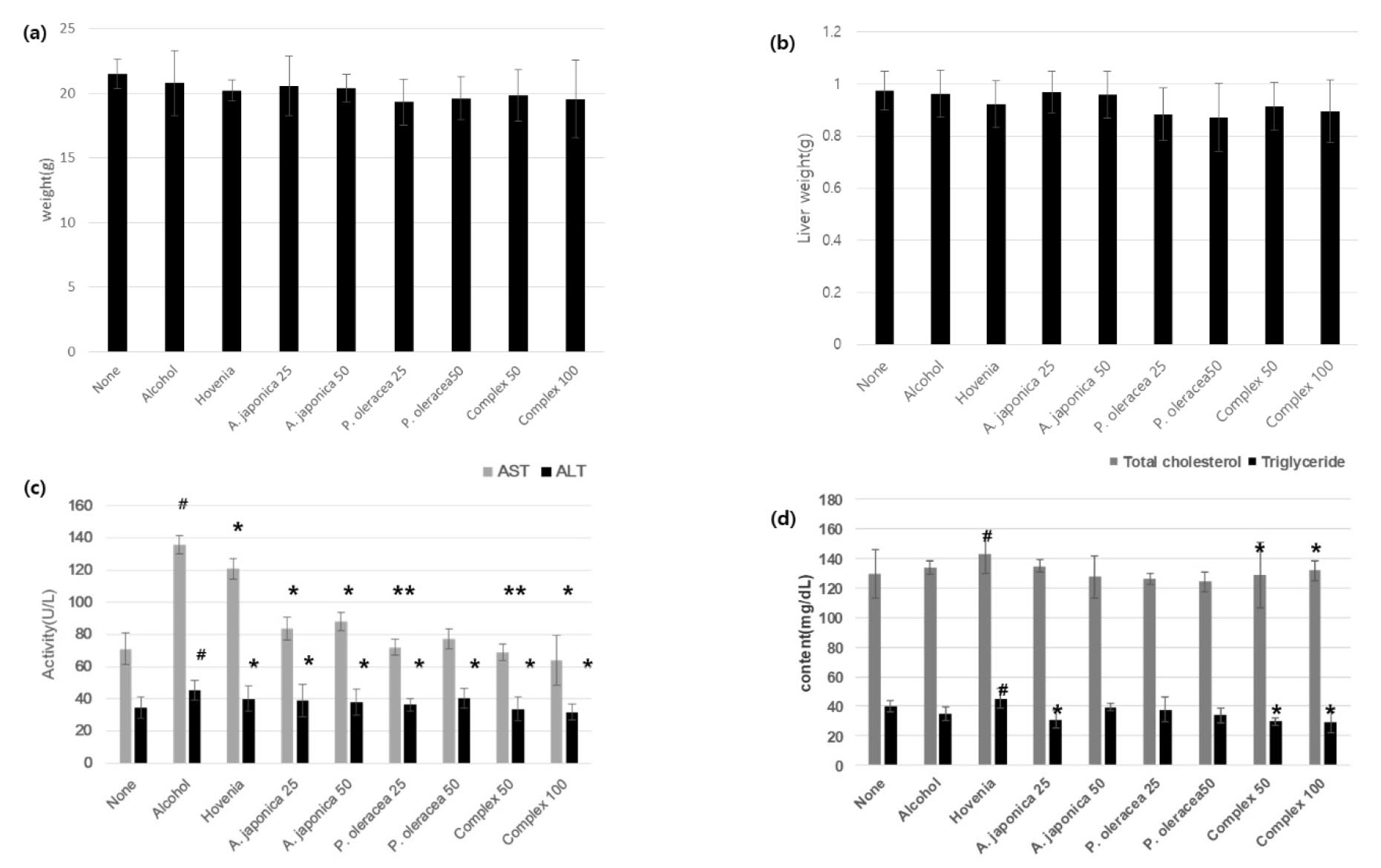
Five ㎎/㎏ of alcohol was administered to male C57BL/6 mice for eight weeks, once daily for a week. As a positive control group, Hovenia dulcis extract powder was used. Weight and liver weight showed a significant decrease in the P. oleracea and complex intake groups.
The result of examining alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), total cholesterol (TC), and triglyceride (TG), which are essential for the diagnosis of liver disease, compared to the group administered only alcohol orally, complex 50 and A significant improvement effect was confirmed in complex 100 (Fig. 3). In particular, AST showed a substantial improvement in the group ingesting A. japonica 25, and P. oleracea 25, and it was confirmed that the complex also showed a significant improvement effect.
Animal efficacy evaluation for improving against chronic alcoholic liver damage
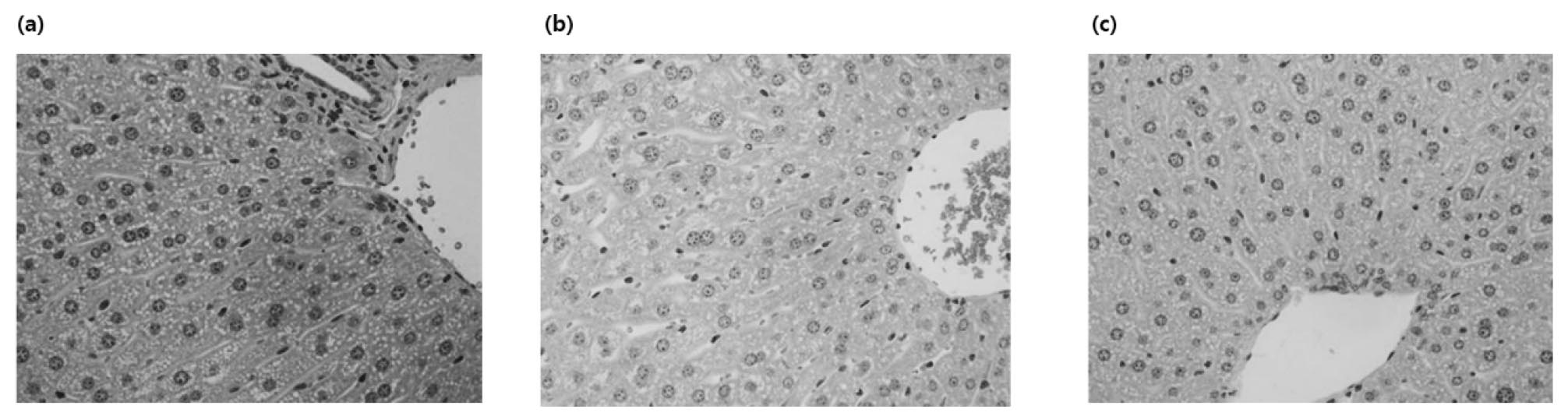
Eight-week-old male C57BL/6 mice were orally administered once daily for four weeks with 3 ㎎/㎏ body weight of alcohol. When hepatocytes are damaged, enzymes present in hepatocytes are released into the blood, and blood levels increase (Fig. 4, Fig. 5). These enzymes and indicators serve as indicators representing the degree of liver damage. As a result of measuring AST and ALT activities in plasma, ALT and AST activities were significantly increased in the alcohol- administered group compared to the normal diet group, confirming that liver tissue was effectively damaged by alcohol. To verify the effects of Alnus 25, Purslane 25, and Complex 50 on alcohol metabolism, which were confirmed by the improvement of acute alcoholic liver damage, the improvement effect on chronic alcoholic liver damage was confirmed (Table 2). In AST and ALT, liver function indices, the Alnus complex showed better physiological activity than Alnus and purslane alone, and A. japonica extract showed a superior LDH reduction effect. CAT activity measured in liver tissue was confirmed to be more effective in the purslane extract than in the A. japonica extract, and in particular, it was confirmed that the ALPOC exhibited the most excellent antioxidant effect (Fig. 4). The state of the liver tissue was established through H&E staining of the liver tissue (Fig. 6). Cell nuclei in the liver tissue were observed in the normal diet group, but many necrosis and hemorrhage were observed in the alcohol-treated group. On the other hand, it was confirmed that necrosis and hemorrhage were reduced in the complex administration group.
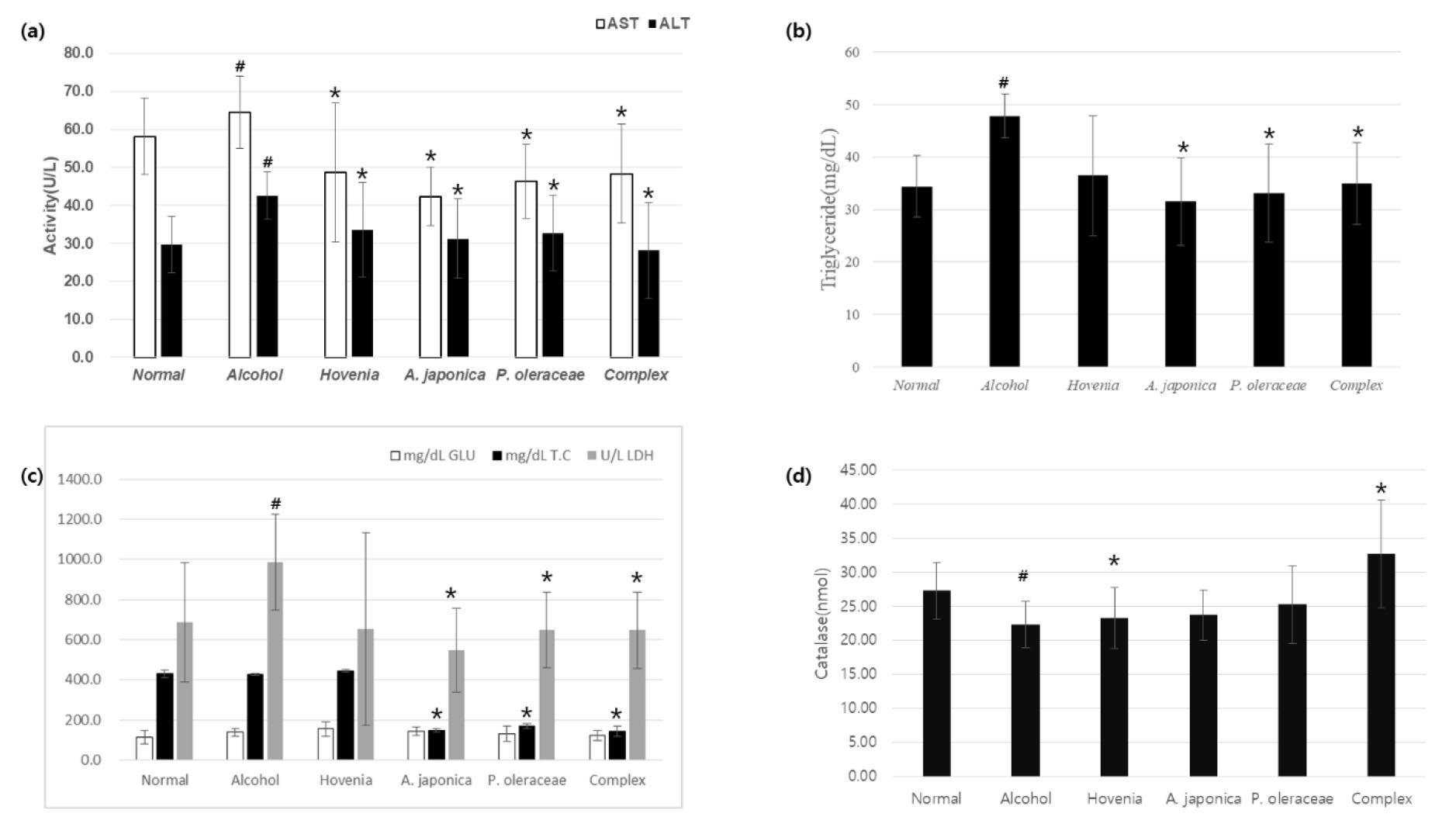
Fig. 4.
Effects of ALPOC on serum biochemical indicator in chronic alcoholic liver damage mouse. a) AST and ALT, b) triglyceride, c) glucose, total cholesterol and Lactate dehydrogenase (LDH), d) catalase. Data are presented as mean ± SEM, n=8. # p < 0.05 compared with the Normal group. * p < 0.05 compared with the alcohol group.
Table 2.
Effects of ALPOC on inflammatory cytokines in alcoholic liver injury mouse. Values are presented as mean ± SEM.
| Dose (㎎/㎏) | IL-6 | IL-8 | TNF-α | |
| None | - | 0 | 0.98 ± 0.3 | 0.4 ± 0.2 |
| Alcohol | - | 2.5 ± 1# | 9.8 ± 0.9# | 2.8 ± 0.5# |
| P. oleracea | 25 | 0.87 ± 0.4 | 2.28 ± 0.2** | 1.2 ± 0.3* |
| A. japonica | 25 | 0.98 ± 0.2* | 1.1 ± 0.3* | 0.98 ± 0.45* |
| ALPOC | 50 | 0.6 ± 0.2* | 1.08 ± 0.2* | 0.8 ± 0.3* |
Discussion
Alcohol is absorbed by the simple diffusion method in the stomach and small intestine, and 50-80% of the alcohol is interested in the small intestine because the alcohol absorption rate is slower in the stomach than in the small intestine. Absorbed alcohol is distributed in various tissues of our body, and the amount of alcohol distributed in the tissues depends on the blood mass of the tissues and the amount of water in the tissues (Lieber, 2004). Very little of the alcohol in the blood is excreted in urine and breathing. Still, more than 90% of the liver undergoes an oxidation process in the liver to decompose into water and carbon dioxide.
Although various efforts have been made to treat liver damage caused by alcohol, materials with curable effects are still needed. In the liver, alcohol is metabolized by alcohol dehydrogenase in the cytoplasm to produce acetaldehyde and ketone. When alcohol is consumed chronically in excess, the microsomal ethanol-oxidizing system in the microsome is involved, and alcohol oxidation is promoted by CYP2E1 (Cytochrome P450 2E1). The third alcohol oxidation pathway is due to catalase present in peroxisome in most tissues, and the effect on alcohol metabolism is very low (Holmström and Finkel, 2014; Wheeler et al., 2001; Yin et al., 2014). Through this study, it was confirmed that the A. japonica – P. oleracea complex identified has high antioxidant activity and effectively removes active oxygen induced by alcohol metabolism through CYP2E1 and catalase. This indicates that the A. japonica – P. oleracea complex can effectively treat liver damage through its antioxidant effects on alcohol.
When hepatocytes are damaged, enzymes present in the hepatocytes are released into the blood, increasing blood levels, and these enzymes and indicators are indicators of liver damage levels (Rasineni and Casey, 2012). Important factors for identifying alcoholic liver are ALT, AST, ALP, gamma-glutamyl transpeptidase (γ-GT), bilirubin, and albumin. In particular, AST and ALT, used most clinically, are widely distributed in the body as catalyzing the amino group transfer reaction between amino acid and a-keto acid. When hepatocytes are damaged, the supply of energy within the cell is reduced, so that + ions flow out of the cell, and Na+, Ca2+, and water flow into the cell (Lee and Friedman, 2011). As a result, the cells expand, the cell membrane is stretched, and AST and ALT in the cytoplasm are released. Therefore, the increase of enzyme activity such as AST and ALT in serum is widely used as an indicator of liver damage research (Plaa and Charbonneau, 1994). In addition, total bilirubin (T-bil) is an index related to acute hepatitis and chronic hepatitis, and triglyceride (TG) is an index that increases due to an increase in the fatty acid synthesis that occurs during chronic irregular A. japonica and P. oleracea complex showed no significant change in liver or body weight as a result of feeding at a dose of 50 ㎎/㎏. In contrast, plasma ALT activity (28.12 U) was 34.9 % lower than that of the alcohol intake group (42.5 U). It showed an improvement effect, and in the case of AST activity, it was confirmed that it showed a very effective hepatoprotective activity by showing an improvement effect of 12.6 %.
In chronic alcoholic liver damage, it was confirmed that ALT showed an improvement effect of 25% or more when a high dose of the complex was ingested. AST showed an improvement effect similar to that of the positive control, Hovenia 100 extract. In addition, through H&E staining analysis, it was observed that necrosis and hemorrhage caused by liver damage were improved by the ALPOC. In terms of the change in catalase, an antioxidant index, the low-dose group of ALPOC showed vigorous antioxidant activity of 127 % or more compared to the alcohol intake group, which was confirmed to offer a very high activity exceeding 20% or more than that of Hovenia extract. On the other hand, there was no significant difference in LDH activity in plasma between the Hovenia extract and the groups.
Studies to improve liver damage caused by alcohol using natural products have been conducted for a very long time. Nevertheless, research results on effective natural materials have not yet appeared. This study confirmed that the ALPOC exhibits an effective preventive effect against liver cell damage caused by pervasive alcohol intake, and it is necessary to verify the effectiveness and safety through human application tests in the future.