Introduction
Materials and Methods
Experimental material
Experimental material extraction
Fraction preparation
Isolation of complex saponin
Cell culture and calcium mobilization control
Isolation of the basilar artery in the rabbit brain
Measurement of blood vessel tension
Statistical analysis
Results
Discussion
Introduction
In contemporary society, hypertension constitutes a significant contributor to mortality rates, with cardiovascular impairment representing a critical risk factor. Elevated blood pressure induces alterations in vascular structures, which can be categorized as structural modifications, which affect the architecture of the blood vessels and their luminal dimensions, and functional alterations, which lead to cardiovascular impairment. For instance, the onset of hypertension precipitates changes in the structural integrity of blood vessels and their luminal capacity, resulting in pathological consequences such as left ventricular hypertrophy. This phenomenon can culminate in heart failure attributable to augmented collagen deposition within the myocardium, thereby facilitating the development of atherosclerosis. This process further exacerbates the inflammatory response, which ultimately compromises the barrier function of endothelial cells, leading to functional disturbances within the cardiovascular system (Jang et al., 2022; Nam, 2022).
Hypertension, classified as a chronic degenerative pathology, constitutes a significant risk factor for cardiovascular diseases, and approximately 20% of the global adult population is affected by this condition. In the Republic of Korea, the number of individuals diagnosed with hypertension has surpassed 7.46 million, representing 14% of the total population. Consequently, the annual healthcare expenditure associated with hypertension is 1.3127 trillion won. The precise etiology of hypertension remains undetermined; however, it is recognized to arise from intricate factors, including genetic predisposition, dietary sodium intake, alcohol consumption, tobacco use, and aging. The renin–angiotensin system (RAS) plays a pivotal role in the mechanisms underlying the elevation of blood pressure (Nam, 2020). RAS activation is triggered by diminished renal blood flow and sodium concentrations, alongside heightened sympathetic nervous system activity, as part of a general physiological response. Renin catalyzes the conversion of angiotensinogen into angiotensin I, which is subsequently transformed into angiotensin II by angiotensin I-converting enzyme (ACE). Angiotensin II exerts a vasoconstrictive effect while simultaneously enhancing sodium reabsorption and promoting potassium excretion, thereby stimulating aldosterone production, which leads to increased blood volume, vasoconstriction, and elevated blood pressure. Currently recognized ACE inhibitors include ramipril, captopril, lisinopril, and spirapril, which are widely administered as anti-hypertensive agents (Ryu et al., 2020). Nonetheless, these drugs undergo rapid degradation post- administration, resulting in their instability and potential off-target effects, which might manifest as adverse reactions such as emesis, generalized weakness, cephalalgia, cough, and anorexia. Thus, ACE inhibitors with superior inhibitory efficacy and stability are warranted, in addition to anti- hypertensive agents that function through novel pharmacological mechanisms (Busch et al., 2000; Nam, 2022).
Kalopanax pictus is a deciduous broad-leaved tree in the Araliaceae family that is found in regions such as Korea, Japan, Manchuria, and China. The outer bark of this plant is utilized as a medicinal herb in traditional oriental medicine. The stem bark of K. pictus has been documented to exert anti-inflammatory, analgesic, anti-bacterial, and anti-arthritis effects; however, its efficacy in the treatment and prevention of hypertension remains undocumented. In recent times, the Korean population has become increasingly susceptible to several adult-onset diseases as a consequence of dietary shifts toward high-fat, high-calorie foods, subsequently leading to a heightened demand for health-promoting functional foods. Research is being undertaken to explore the potential of phytochemicals derived from diverse plant species as functional materials, thereby necessitating a comprehensive understanding of the physiological activities of plants that have primarily been utilized for aesthetic purposes. Prior research regarding K. pictus stem bark primarily focused on the isolation of specific compounds and general studies of physiological activities; nevertheless, preclinical investigations targeting major ailments, including hypertension, are markedly limited. This study investigated the potential of crude saponin isolated from K. pictus stem bark as a functional health material characterized by high safety profiles and minimal adverse effects, as demonstrated through animal model experiments assessing vasorelaxant properties (Hwang et al., 2013; Nam, 2022).
Materials and Methods
Experimental material
The stem bark of K. pictus was procured from Gyeong-dong Market (Jegi-dong, Dongdaemun-gu, Seoul, Korea) in February 2017, and its identity was confirmed by Dr. Soo Jeong Kim. The sample intended for extraction was dried in a shaded environment and subsequently pulverized. A voucher specimen (NIHA P-29-9) has been archived in the herbarium of the Highland Agriculture Research Institute (Pyeongchang, Korea).
Experimental material extraction
The stem bark of K. pictus was pulverized into a powder (9 ㎏) and then chilled with ethanol at room temperature for 6 months. After filtration of the ethanol extract through qualitative filter paper, the resulting filtrate was concentrated using a rotary vacuum evaporator (Tokyo Rikakikai Co., Tokyo, Japan) to yield an ethanol extract (Nam, 2022; Park et al., 2022), as detailed in Table 1.
Table 1.
The yield of the stem bark of K. pictus ethanol extract.
| Sample | Ethanol extractz | |
| Weight (g) | Yield (%) | |
| Stem bark of K. pictus | 374.1 | 4.2 |
Fraction preparation
After suspending 374.1 g of the stem bark of K. pictus ethanol extract in 1 L of distilled water, the extract was eluted in n-hexane (1 L × 3), chloroform (1 L × 3), ethyl acetate (1 L × 3), and butyl alcohol (BuOH, 1 L × 3) in the order of polar to nonpolar solvents (Nam, 2022; Park et al., 2022). The yields after elution and vacuum concentration are presented in Table 2.
Table 2.
The yield of the organic solvent fractions from the stem bark of K. pictus.
| Solvent fractionz | Stem bark of K. pictus | |
| Weight (g) | Yield (%) | |
| n-Hexane | 45.4 | 12.1 |
| Chloroform | 35.6 | 9.5 |
| Ethyl acetate | 15.4 | 4.1 |
| Butyl alcohol | 49.9 | 13.3 |
Isolation of complex saponin
The stem bark of K. pictus was subjected to three rounds of cutting and extraction in ethanol under reflux conditions, followed by evaporation to yield a viscous substance. The ethanol extract was dried under vacuum. The mixed ethanol extracts derived from the stem bark of K. pictus were fractionated utilizing BuOH. Complex saponin was isolated from the BuOH fraction using the Diaion HP-20 resin adsorption technique (Nam, 2024; Park et al., 2022). The resultant isolated complex saponin (7.2 g) served as the sample for the vasodilation assay (Fig. 1).
Cell culture and calcium mobilization control
A7r5 vascular smooth muscle cells were cultivated at 37°C in a 5% CO2/95% air incubator in Dulbecco’s Modified Eagle Medium supplemented with 10% fetal bovine serum, 10 units/mL penicillin, 100 μg/mL streptomycin, and 2 mM l-glutamine. The monolayer-cultured cells residing in the culture flask were washed twice with basal medium and then detached with a 2.5% trypsin solution. The resulting suspended cells were subsequently neutralized using serum medium and centrifuged. Then, the cell pellet was rinsed three times with the cell culture medium. Moreover, a hemocytometer was employed to quantify the cell population, which was calibrated to achieve a density of 5 × 104 cells/mL. And 200 μL of each cell suspension was dispensed into a 96-well plate (dark-clear bottom, Corning, Corning, NY, USA) and incubated for 24 h. Cells maintained within the 96-well plate were treated with 5 μM Fura-2/AM (molecular probe) for 1 h in a CO2 incubator at 37°C and subsequently washed three times with Ca2+-free PSS. After adding 190 μL of PSS containing 1.5 mM Ca2+ to each well, complex saponin isolated from the stem bark of K. pictus, intended for testing, was administered at a concentration of 10 μg/mL, and the assay plate was positioned into the Flexstation 3 reader. A 5-fold concentrated K+ solution was incorporated into the compound plate to ensure a final concentration of 50 mM K+ upon addition. Following the automatic addition of K+ by Soft Max Pro 5.1, the fluorescence intensity was measured (Cauvin et al., 1983; Huang, 1996; Jang et al., 2022; Karaki et al., 1997; Li et al., 1993; Nam, 2022; Park et al., 2008; Velasco et al., 1997).
Isolation of the basilar artery in the rabbit brain
The basilar artery used in this study was isolated using surgery. Briefly, white rabbits weighing 2~2.5 ㎏ were anesthetized via enflurane inhalation, and then the basilar artery at the bottom of the brain was excised while the skull was cut. The excised basilar artery was immersed in Ca2+-free Tyrode’s solution (137 μM NaCl, 5.4 μM KCl, 1 μM MgCl2, 23.8 μM NaHCO3, 5.5 μM glucose, and sucrose) saturated with 95% O2 and 5% CO2 to remove the surrounding connective tissue and fat. The Ca2+-free Tyrode’s solution was used to adjust the osmotic pressure to 300 mOsm/㎏ H2O (Cauvin et al., 1983; Huang, 1996; Jang et al., 2022; Karaki et al., 1997; Li et al., 1993; Nam, 2022; Park et al., 2008; Velasco et al., 1997).
Measurement of blood vessel tension
The separated blood vessels were tested by storing them in Ca2+-free Tyrode’s solution saturated with 95% O2 and 5% CO2. First, the basilar artery was cut 5 ㎜ and fixed to the end of the force transducer using a chromium ring and to the clasp ring at the bottom of the organ bath, maintained at 37°C. Isometric contraction was measured by measuring the contractile force when both ends were fixed. During this measurement, the organ bath was continuously saturated with 95% O2 and 5% CO2. By replacing normal Tyrode’s solution with Tyrode’s solution containing a high K+ concentration (50 mM), the membrane potential was depolarized to −20 mV according to the Nernst equation, and the passage through which the ions traversed was closed. By activating the inactivated L-type calcium channel, the calcium concentration in the vascular smooth muscle cells was increased to induce contraction, and contraction and relaxation were repeated three times. The reactivity of the endothelial cells of the basilar arteries used in this experiment was confirmed by checking whether the contractile force of blood vessels decreased when Tyrode’s solution containing a high K+ concentration was used to induce contraction and then treated with 1 μM acetylcholine. After stabilizing the basilar artery for 10 min with Tyrode’s solution containing a high K+ concentration, complex saponin isolated from the stem bark of K. pictus was applied at different concentrations for approximately 10 min to confirm its vasodilatory effect. Endothelin-1–induced vasoconstriction was reduced by treatment with 3 nM endothelin-1. Tyrode’s solution used in the vasoconstriction and relaxation experiment comprised (in μM) 137 NaCl, 5.4 KCl, 1.5 CaCl2, 1 MgCl2, 23.8 NaHCO3, and 5.5 glucose, and Tyrode’s solution containing a high K+ concentration comprised (in μM) 92.4 NaCl, 50 KCl, 1.5 CaCl2, 1 MgCl2, 23.8 NaHCO3, and 5.5 glucose. The osmotic pressure was maintained at 300 mOsm/㎏ H2O, and the pH was adjusted to 7.4 via saturation with 95% O2 and 5% CO2 (Cauvin et al., 1983; Huang, 1996; Jang et al., 2022; Karaki et al., 1997; Li et al., 1993; Nam, 2022; Park et al., 2008; Velasco et al., 1997).
Statistical analysis
The experimental data were presented as the mean ± standard error (SE), and the alterations in vasoconstriction were delineated in terms of the actual magnitude of contraction as well as the percentage (mean ± SE) of the peak contraction elicited by the contracting agents (K+ and endothelin-1). The statistical significance of each experimental outcome was assessed utilizing Student’s t-test (Nam, 2022; Park et al., 2008).
Results
To assess whether the calcium mobilization force in vascular smooth muscle cells, a major factor in vasoconstriction, can be controlled, the Ca2+-specific fluorescent dye Fura-2 was loaded onto A7r5 cells and then excited at 340/380 ㎚. As presented in Table 3, the emission wavelength intensity was 1.82 ± 0.15 μM when a high K+ concentration was used as a control group, and treatment with complex saponin isolated from the stem bark of K. pictus (10 μg/mL) yielded an intensity of 1.67 ± 0.14 μM, denoting an inhibitory effect. Thus, inhibitory effects on vasoconstriction could be induced by a high K+ concentration.
Table 3.
Tension regulation in vascular smooth muscle cells.
| Sample |
Regulation of K+-induced calcium mobilizing powerz (F340/380 ratio, μM) |
|
Complex saponin isolated from the stem bark of K. pictus (10 µg/mL) |
1.67 ± 0.14 |
| Negative control (untreated cells) | 1.82 ± 0.15 |
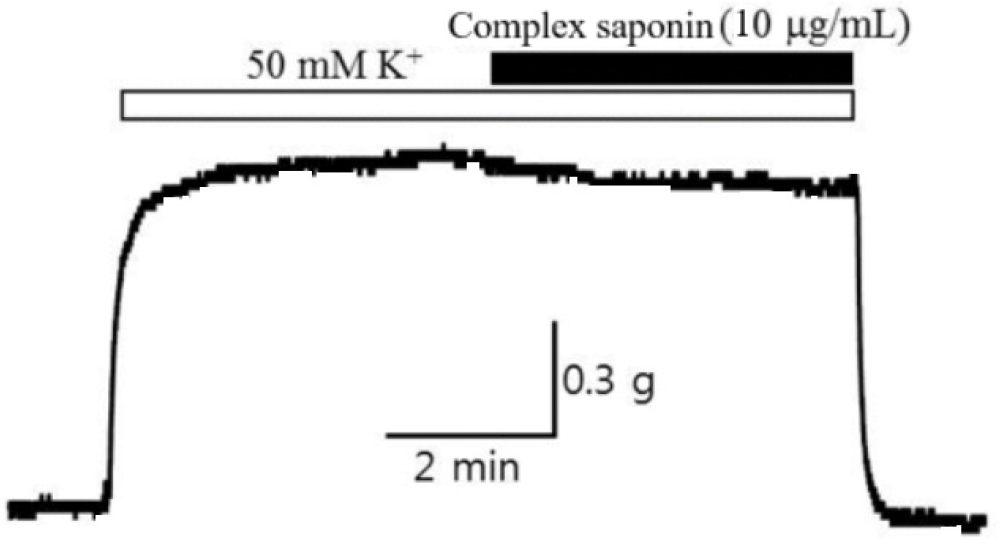
Basic blood vessel tension or blood vessel tension altered by certain conditions manifests as vascular smooth muscle contraction and relaxation. In particular, blood vessel contraction involves an increase in cytoplasmic Ca2+ concentrations via ion inflow through voltage-dependent Ca2+ channels. Upon stimulation with electrical stimulus (e.g., excitation wave transmission from the dominant nerve in the blood vessel), vascular smooth muscle is electrically depolarized, and L-type calcium channels, opened through membrane potential, remain open. This permits Ca2+ ions to flow from the outside to the inside depending on the concentration gradient, resulting in vascular smooth muscle contraction and increased tension in blood vessels (Jagadesh and Subhash, 1998; Tytgat and Daenens, 1997). Therefore, in this study, the relaxation effect of complex saponin isolated from the stem bark of K. pictus (10 µg/mL) was investigated after artificially inducing depolarization using a high K+ concentration. First, Tyrode’s solution containing a high K+ concentration was applied to rabbit basilar arteries, and when vasoconstriction was stabilized after approximately 10 min, the artery was treated with complex saponin, and the degree of relaxation of constricted blood vessels was measured for 10–15 min. Treatment with complex saponin resulted in improved vasodilatation efficacy (Table 4, Fig. 2).
Table 4.
Vasodilatation potency of complex saponin isolated from the stem bark of K. pictus in rabbit basilar arteries after K+-induced vasoconstriction.
| Sample | Vasodilatation efficacyz (EC50)y |
| Basilar arteryx (%) | |
|
Complex saponin isolated from the stem bark of K. pictus (10 µg/mL) | 8.7 ± 4.5 |
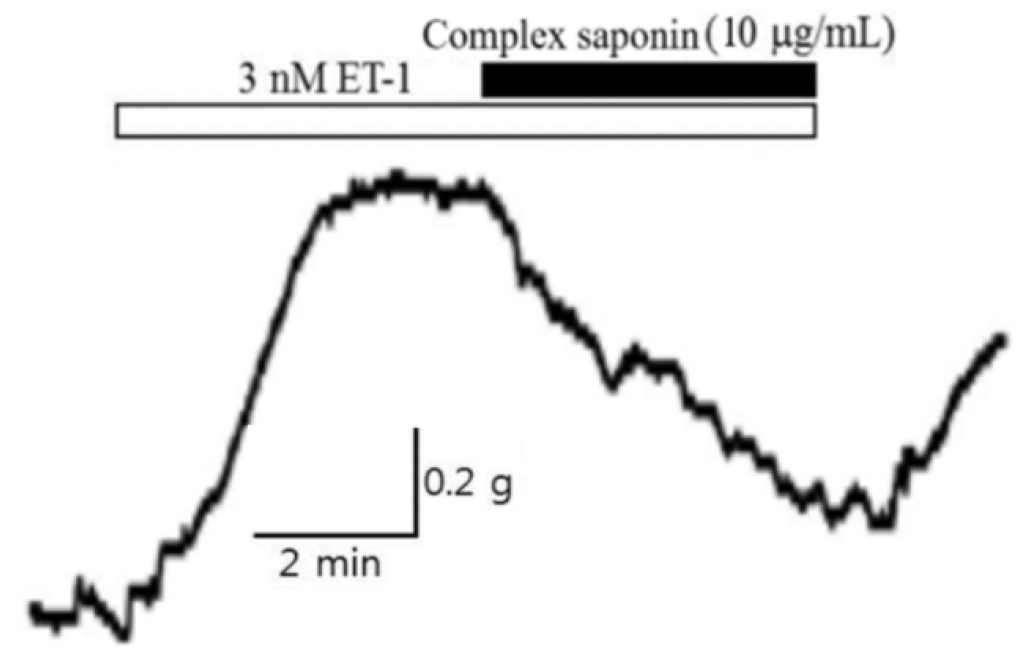
The blood concentration of endothelin-1 increases by more than 100-fold in patients with diabetes, leading to hypertension, renal failure, foot necrosis, and peripheral circulation disorders. Accordingly, the vasodilatory effect of complex saponin on vasoconstriction induced by endothelin-1 was validated. After constricting basilar arteries with 3 nM endothelin-1, vasodilation was observed after treatment with 10 µg/mL complex saponin. As presented in Table 5 and Fig. 3, greater than 72% vasodilatation was achieved. Thus, complex saponin isolated from the stem bark of K. pictus could be utilized as a candidate endothelin receptor inhibitor.
Discussion
Hypertension in adults is clinically characterized by systolic blood pressure of ≥140 ㎜Hg or diastolic blood pressure of ≥90 ㎜Hg (Kim and Kim, 2018). Nonetheless, individuals with elevated blood pressure often exhibit no discernible symptoms, rendering diagnosis contingent upon the measurement of blood pressure. In traditional oriental medicine, the stem bark of K. pictus has been predominantly employed as a therapeutic agent to manage hypertension; thus, prior research investigated the vasorelaxant properties of the stem bark of K. pictus in rabbit basilar arteries subjected to contraction via endothelin-1 (Jang et al., 2022). In this investigation, the vasodilatory effect was assessed following the isolation of complex saponin from the stem bark of K. pictus. Endothelin-1, recognized as the most potent vasoconstrictor within the human circulatory system, exerts its effects on endothelin receptor subtype A located on the membrane of vascular smooth muscle cells and receptor subtype B2 found on the membrane of vascular endothelial cells, thereby increasing vascular tone. Endothelin-1 functions as a modulating agent to diminish vascular tension (Lucchelli et al., 1999). In general, the blood concentrations of endothelin peptides increase in response to inflammatory responses, diabetes, and cardiovascular pathologies, whereas aberrant hyperactivity of the endothelin system is implicated in renal disorders, including chronic renal failure and glomerulosclerosis, and respiratory conditions, such as idiopathic pulmonary fibrosis and chronic obstructive pulmonary disease. Endothelin signaling is associated with various diseases, including diabetic neuropathy, retinopathy, limb necrosis, and malignancies such as prostate and colorectal cancer, which represent significant areas of focus within the realms of mechanical and metabolic disorders (Pancrazio et al., 1998). In the present investigation, the inhibitory effects of complex saponin extracted from the stem bark of K. pictus on endothelin-1–induced constriction of cerebral basilar arteries were meticulously examined. Consequently, studies focused on the structural and functional derivation of complex saponin isolated from the stem bark of K. pictus should be conducted to assess its potential to modulate endothelin activity with the goal of developing health-functional foods or pharmaceutical products.