Introduction
Materials and Methods
Materials
HSL Preparation
Cell Culture and Treatment
Western Blot Analysis
Statistical Analysis
Results and Discussion
HSL increases the level of p62/SQSTM1 protein in RAW264.7 cells
HSL-mediated increase of p62/SQSTM1 is dependent on TLR4
HSL-mediated Increase of p62/SQSTM1 is Dependent on p38, JNK, and NF-κB
HSL-mediated Activations of p38, JNK, and NF-κB are Dependent on TLR4
HSL-mediated Increase of Nrf2 Protein Level Results from TLR4-dependent Activation of p38, JNK, and NF-κB Signaling
Introduction
Humans live in coexistence with various microorganisms existing inside and outside. Nevertheless, humans avoid various diseases caused by pathogenic microorganisms because of their innate and adaptive immune systems (Hirayama et al., 2018). Antigens are recognized and eliminated by an innate immune response in the early stages of infection, and the adaptive immune response removes the antigen by secreting antigen-specific antibodies (Hirayama et al., 2018). Activation of these immune responses is necessary for maintaining homeostasis in the body. Research has shown that the consumption of specific functional foods such as ginseng, mushrooms, chlorella, and probiotics (Lactobacillus plantarum) strengthening the activity of immune cells can stimulate the activity of immune cells, providing protection against cancer, viruses, and bacteria (Kim et al., 2022).
Macrophages, which are innate immune cells, recognize foreign pathogens through various membrane receptors and eliminate them through phagocytosis (Schenten and Medzhitov, 2011). In addition, T- and B-cells (adaptive immune cells) are activated by pro-inflammatory cytokines produced by activated macrophages (Duque and Descoteaux, 2014). And macrophages have an antigen presentation function that provides antigen information to adaptive immune cells (Barker et al., 2002). Therefore, activation of macrophages is a good strategy to simultaneously increase innate immunity and adaptive immune response.
Hibiscus syriacus (HS), the national flower of Korea, is a traditional medicinal plant that has been used for detoxification, tonicity, and antipyretic purposes (Karunarathne et al., 2019). In Korea, Hibiscus syriacus is currently registered as a food raw material, and interest in developing functional foods using Hibiscus syriacus is growing. Recently, we confirmed that Hibiscus syriacus leaves extract increased the secretion of inflammatory cytokines and phagocytosis in macrophages, which indicates that Hibiscus syriacus leaves may exert immunostimulatory activity (Geum et al., 2022).
Autophagy is a recycling process through bulk degradation, which is essential for maintaining normal biological functions (Wu and Lu, 2020). Autophagy of macrophages is involved in phagocytosis, pro-inflammatory cytokine release, inflammatory response, and antigen presentation function, which are major functions of macrophages (Wu and Lu, 2020). Therefore, autophagy is essential for normal macrophage function. So, we investigated the effect of Hibiscus syriacus leaves on the autophagy of macrophages.
Materials and Methods
Materials
Chemical inhibitors (TAK-242, C29, PD98059, SB203580, SP600125, BAY 11-7082, and LY294002) were purchased from Sigma-Aldrich (St. Louis, MO, USA). The primary antibodies (p-p38, p-JNK, p-IKK, p-IκBα, p62/SQSTM1, Nrf2, or β-actin) and the secondary antibodies (anti-rabbit or anti-mouse) were purchased from Cell Signaling (Beverly, MA, USA).
HSL Preparation
Dried Hibiscus syriacus leaves (10 g) were immersed in 200 mL of DH2O at 60℃ for 6 h. Then the extraction supernatant was collected after the centrifugation at 15,000 rpm. The final water extracts from Hibiscus syriacus leaves (HSL) were obtained by freeze-drying the extraction supernatant.
Cell Culture and Treatment
The cell line used in this study is mouse macrophages, RAW264.7 cells (American Type Culture Collection, Manassas, VA, USA). The environmental condition for cell maintenance is 5% CO2, 37℃, humidified atmosphere. DMEM/F-12 medium containing 10% fetal bovine serum, penicillin (100 U/ mL), and streptomycin (100 ㎍/mL) was used for cell maintenance. HSL was dissolved in sterile distilled water, and chemical inhibitors were dissolved in DMSO. When cells were treated with HSL and chemical inhibitors, sterile distilled water and DMSO were used as controls, respectively.
Western Blot Analysis
After the quantitative analysis of cellular proteins with a BCA kit (Thermo Fisher Scientific, Waltham, MA USA), sodium dodecyl-sulfate polyacrylamide gel electrophoresis was carried out to separate the protein. Then, the separated protein was transferred to a nitrocellulose membrane (Thermo Fisher Scientific). After blocking the membranes for 1 h, the primary antibodies (p-p38, p-JNK, p-IKK, p-IκBα, p62/SQSTM1, Nrf2, or β-actin) were treated to the membranes at 4℃ overnight. After that, secondary antibodies were treated to membranes for 1 h. The protein band on the membranes was detected using LI-COR C-DiGit Blot Scanner.
Statistical Analysis
All the data are shown as mean ± SD (standard deviation). Statistical significance was determined by Student’s t-test. Differences with *P <0.05 was considered statistically significant.
Results and Discussion
HSL increases the level of p62/SQSTM1 protein in RAW264.7 cells
Viral pandemics such as COVID-19 are raising the need for vaccine development. An adjuvant essential in vaccine development is used to boost antigen-specific immune responses by T cells and B cells (Chou et al., 2020). The enhancement of the antigen-specific immune responses by the adjuvant is achieved by activating the antigen-presenting function of macrophages (Coffman et al., 2010). Autophagy enhances the phagocytosis and antigen-presenting ability of macrophages (Wu and Lu, 2020). p62/SQSTM1 (p62/sequestosome 1) is an important cargo receptor in the autophagy process (Lamark et al., 2009). p62/SQSTM1 is responsible for converting cargo into ubiquitinated aggregates and sequestering them in autophagosomes (Komatsu and Ichimura, 2010). Since p62/SQSTM1 targets invading pathogens, p62/ SQSTM1 is important for the autophagy of these pathogens (Zheng et al., 2009). So, to investigate the effect of HSL on macrophage autophagy, we measured the level of p62/SQSTM1 protein in HSL-treated RAW264.7 cells. As shown in Fig. 1A, HSL increased p62/SQSTM1 protein level at 3 h of treatment and continued to increase p62/SQSTM1 protein level until 24 h. HSL dose-dependently increased p62/SQSTM1 protein level (Fig. 1B). These results show that HSL can have a positive effect on the induction of autophagy in macrophages.
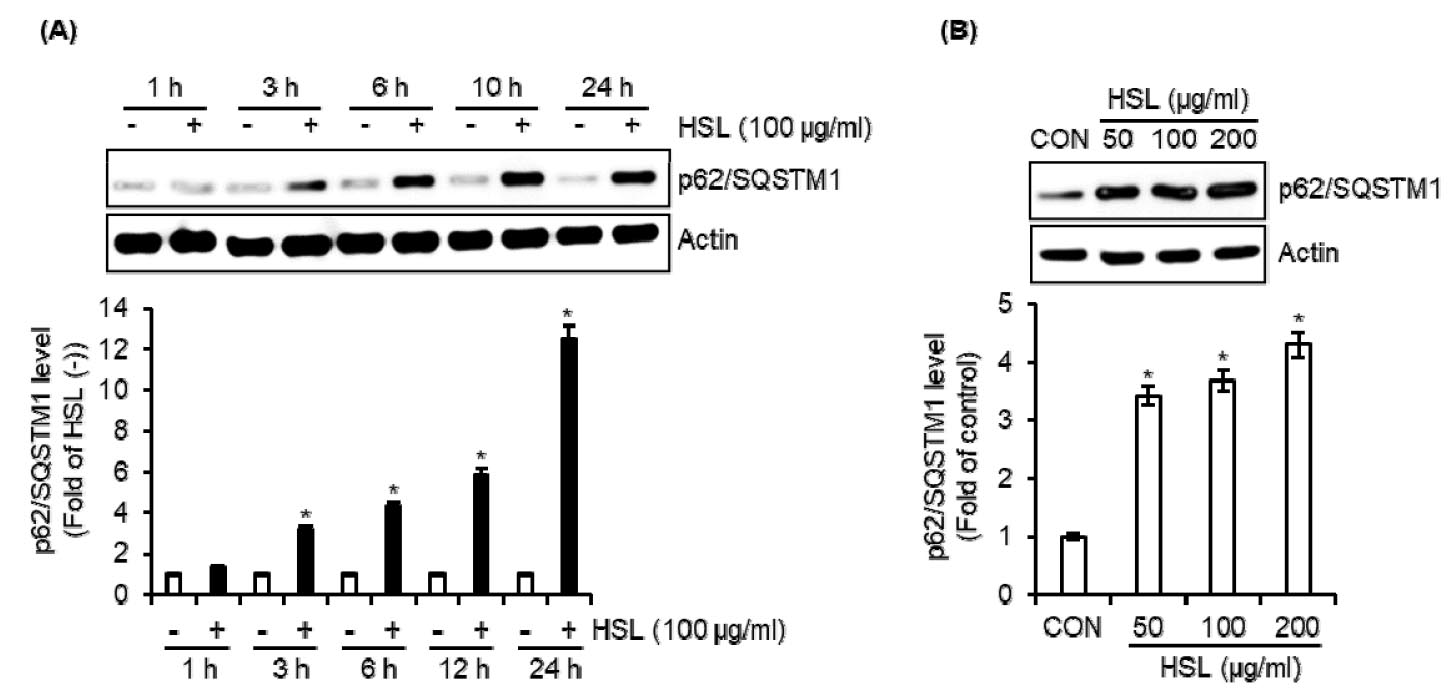
Fig. 1.
The effect of HSL on the expression of p62/SQSTM1 in RAW264.7 cells. (A) RAW264.7 cells were treated with HSL (100 ㎍/mL) for the indicated times. (B) RAW264.7 cells were treated with HSL (100 ㎍/mL) at the indicated concentrations for 6 h. The level of p62/SQSTM1 was analyzed by Western blot analysis.
HSL-mediated increase of p62/SQSTM1 is dependent on TLR4
Macrophages initiate the body's immune response by recognizing the foreign pathogen with TLRs (Toll-like receptors), which is an important process in the body's immune defense (Chou et al., 2020). Recently, it has been found that TLRs enhance antigen-processing and antigen-presenting functions of macrophages by inducing autophagy (Chou et al., 2020). Therefore, we investigated the involvement of TLR2 and TLR4 in the HSL-mediated increase of p62/SQSTM1 protein level in RAW264.7 cells. The increase of p62/SQSTM1 protein level by HSL was induced in TLR2-suppressed RAW264.7 cells by C29 (Fig. 2). However, TAK-242-mediated inhibition of TLR4 resulted in the dramatic downregulation of HSL-mediated increase of p62/SQSTM1 protein level (Fig. 2). These findings suggest that the HSL-mediated increase of p62/SQSTM1 may be dependent on TLR4. Indeed, TLR4-dependent autophagy is reported to promote the clearance of foreign pathogens via autophagosomes (Xu et al., 2007). Also, TLR4-mediated autophagy induction is an effective target for adjuvant development, as TLR4 agonists are clinically used as adjuvants with autophagy function (Steinhagen et al., 2011; Toussi and Massari, 2014). Therefore, TLR4-mediated autophagy of HSL provides the basis for HSL to be considered as an agent for adjuvant development.
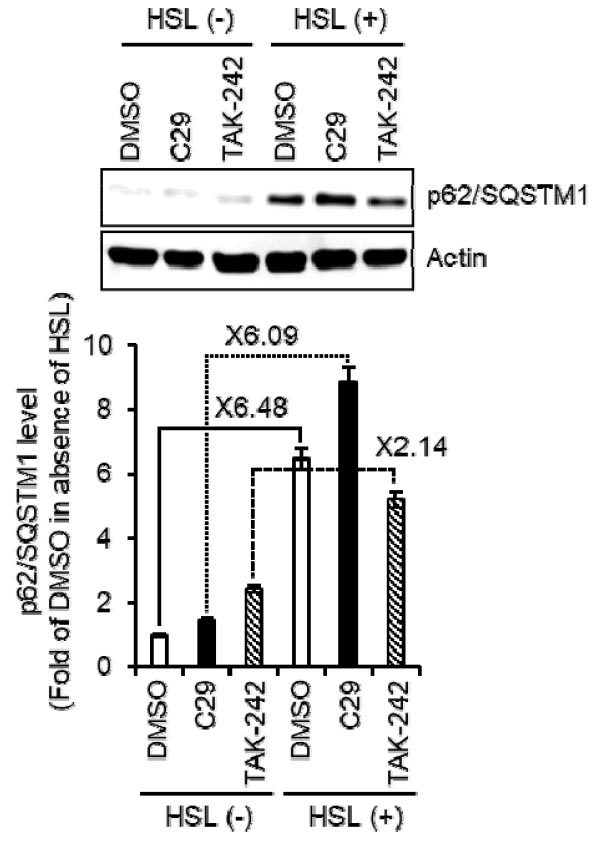
Fig. 2.
The effect of TLR2/4 on the HSL-mediated expression of p62/SQSTM1 in RAW264.7 cells. RAW264.7 cells were treated with HSL (100 ㎍/mL) in the absence or presence of C29 (TLR2 inhibitor, 100 nM) or TAK-242 (TLR4 inhibitor, 5 ㎍/mL) for 6 h. The level of p62/SQSTM1 was analyzed by Western blot analysis.
HSL-mediated Increase of p62/SQSTM1 is Dependent on p38, JNK, and NF-κB
Activation of MAPKs (Mitogen-activated protein kinases), NF-κB (Nuclear factor kappa B), or PI3K/AKT (phosphoinositide 3-kinase/protein kinase B) is an essential requirement for TLR4-mediated autophagy (Puissant et al., 2012; van der Varrt et al., 2014; Yazid and Hung-Chih, 2021). So, we investigated which signaling is involved in the induction of TLR4-mediated autophagy by HSL in RAW264.7 cells. PD98059- and LY294002-mediated suppression of ERK1/2 (Extracellular signal-regulated protein kinase 1/2) and PI3K, respectively, did not reverse the HSL-mediated increase of p62/SQSTM1 protein level (Fig. 3A and 3B). However, the inhibition of p38 by SB203580, JNK (c-Jun N-terminal Kinase) by SP600125, and NF-κB by BAY 11-7082 dramatically blocked the increase of p62/SQSTM1 protein level by HSL (Fig. 3A). These findings suggest that activations of p38, JNK, and NF-κB may be required for the HSL-mediated increase of p62/SQSTM1 protein level.
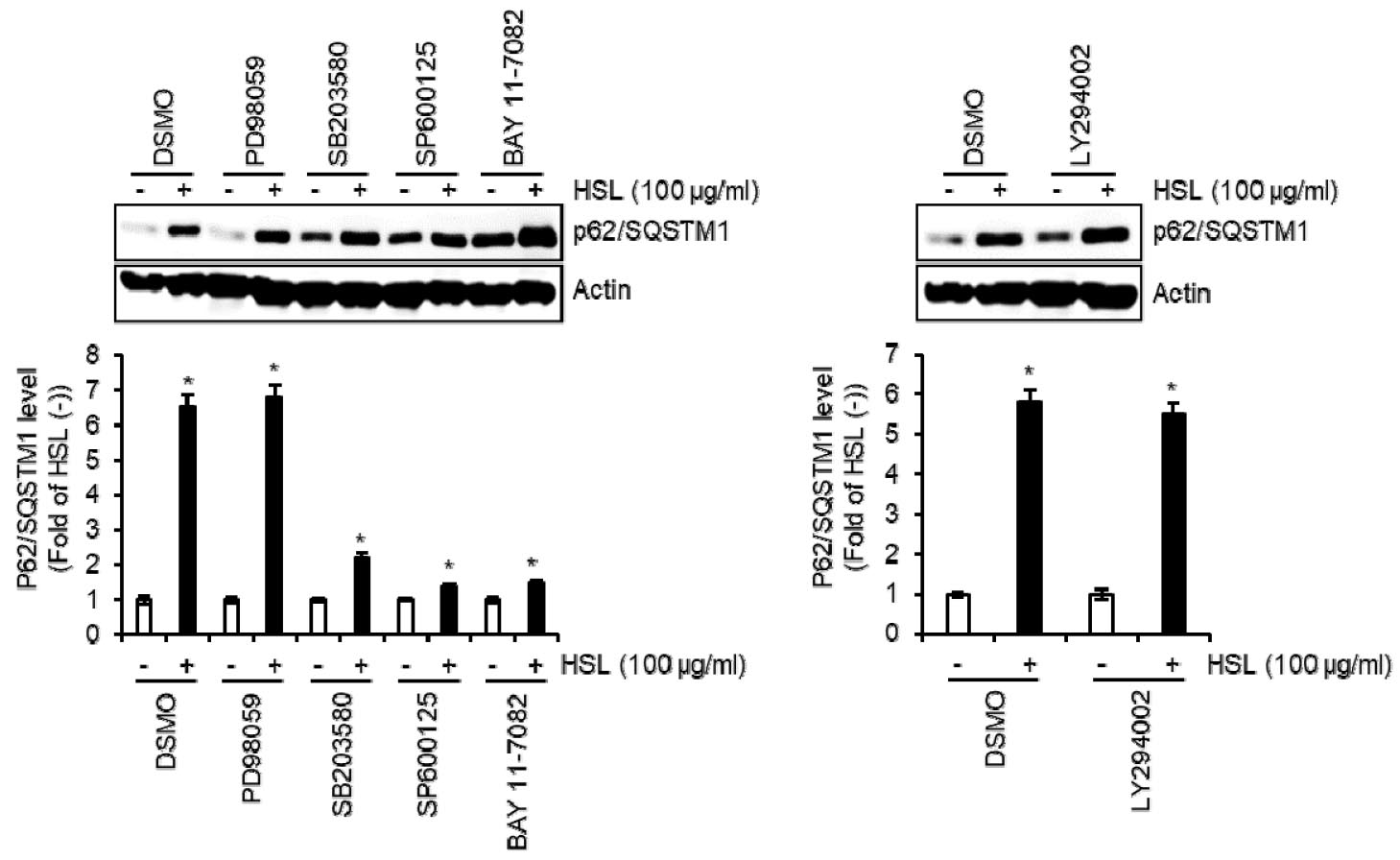
Fig. 3.
The effect of MAPK, NF-κB, and PI3K/AKT signaling on the HSL-mediated expression of p62/SQSTM1 in RAW264.7 cells. RAW264.7 cells were treated with HSL (100 ㎍/mL) in the absence or presence of PD98059 (ERK1/2 inhibitor, 40 μM), SB203580 (p38 inhibitor, 40 μM), SP600125 (JNK inhibitor, 40 μM), BAY 11-7082 (NF-κB inhibitor, 20 μM), or LY294002 (PI3K inhibitor, 40 μM) for 6 h. The level of p62/SQSTM1 was analyzed by Western blot analysis.
HSL-mediated Activations of p38, JNK, and NF-κB are Dependent on TLR4
Since TLR4, p38, JNK, and NF-κB contributed to the increase of p62/SQSTM1 protein level by HSL, we investigated that HSL activates p38, JNK, and NF-κB signaling. First, since the p38 phosphorylation and JNK phosphorylation reflect their activation, the p38 phosphorylation and JNK phosphorylation was investigated in HSL-treated RAW264.7 cells. HSL was shown to rapidly induce phosphorylation of p38 and JNK (Fig. 4A). NF-κB is activated by phosphorylation-mediated degradation of IκB-α by activated IKK (Liu et al., 2017). IκB-α degradation transfers p66 from the cytoplasm to the nucleus, which in turn induced the transcription of various factors (Liu et al., 2017). As shown in Fig. 4A, increased phosphorylation of IKK and IκB-α and decreased level of IκB-α were observed in HSL-treated RAW264.7 cells. These findings indicate that HSL may activate p38, JNK, and NF-κB signaling. We also found that TAK-242-mediated inhibition of TLR4 resulted in blocking the phosphorylation of p38, JNK, IKK and IκB-α, and IκB-α degradation by HSL (Fig. 4B). These results suggest that HSL-mediated activation of p38, JNK, and NF-κB signaling may be dependent on TLR4.
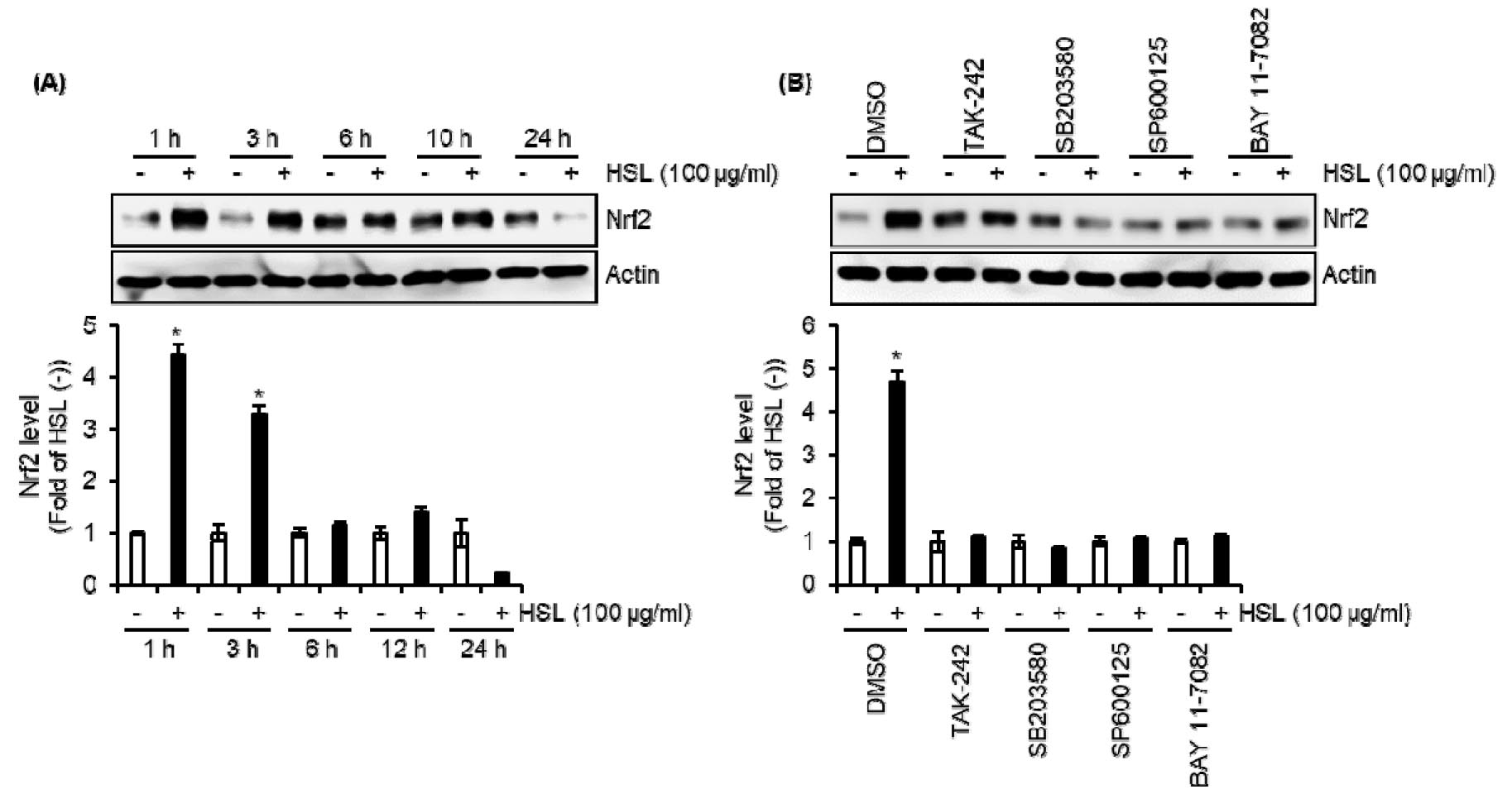
HSL-mediated Increase of Nrf2 Protein Level Results from TLR4-dependent Activation of p38, JNK, and NF-κB Signaling
It has been elucidated that Nrf2 (Nuclear factor erythroid 2-related factor 2) participates in the TLR4-mediated innate immune response (Thimmulappa et al., 2006). In addition, Nrf2 is involved in the autophagy process by increasing the expression of p62/SQSTM1 (Fujita et al., 2011; Fujita and Srinivasula, 2011). Thus, we investigated the contribution of Nrf2 to the expression of TLR4-dependent p62/SQSTM1 by HSL. As shown in Fig. 5A and 5B, the Nrf2 protein level was elevated in HSL-treated RAW264.7 cells, but the inhibition of TLR4 by TAK-242, p38 by SB203580, JNK by SP600125, and NF-κB by BAY 11-7082 attenuated HSL-mediated increase of Nrf2 protein level. These findings suggest that the HSL-mediated increase of Nrf2 protein level is dependent on TLR4-dependent activation of p38, JNK, and NF-κB signaling.
In this study, we observed that HSL increases p62/SQSTM1 expression through activating Nrf2 expression via TLR4-dependent activation of p38, JNK, and NF-κB in RAW264.7 cells. These findings may be evidence to show that HSL can induce autophagy in macrophages. Because macrophage autophagy is one of the main strategies that can enhance innate and adaptive immune responses, it is thought that HSL can be used as a healthy functional food agent or adjuvant that can increase the immune response of the human body.
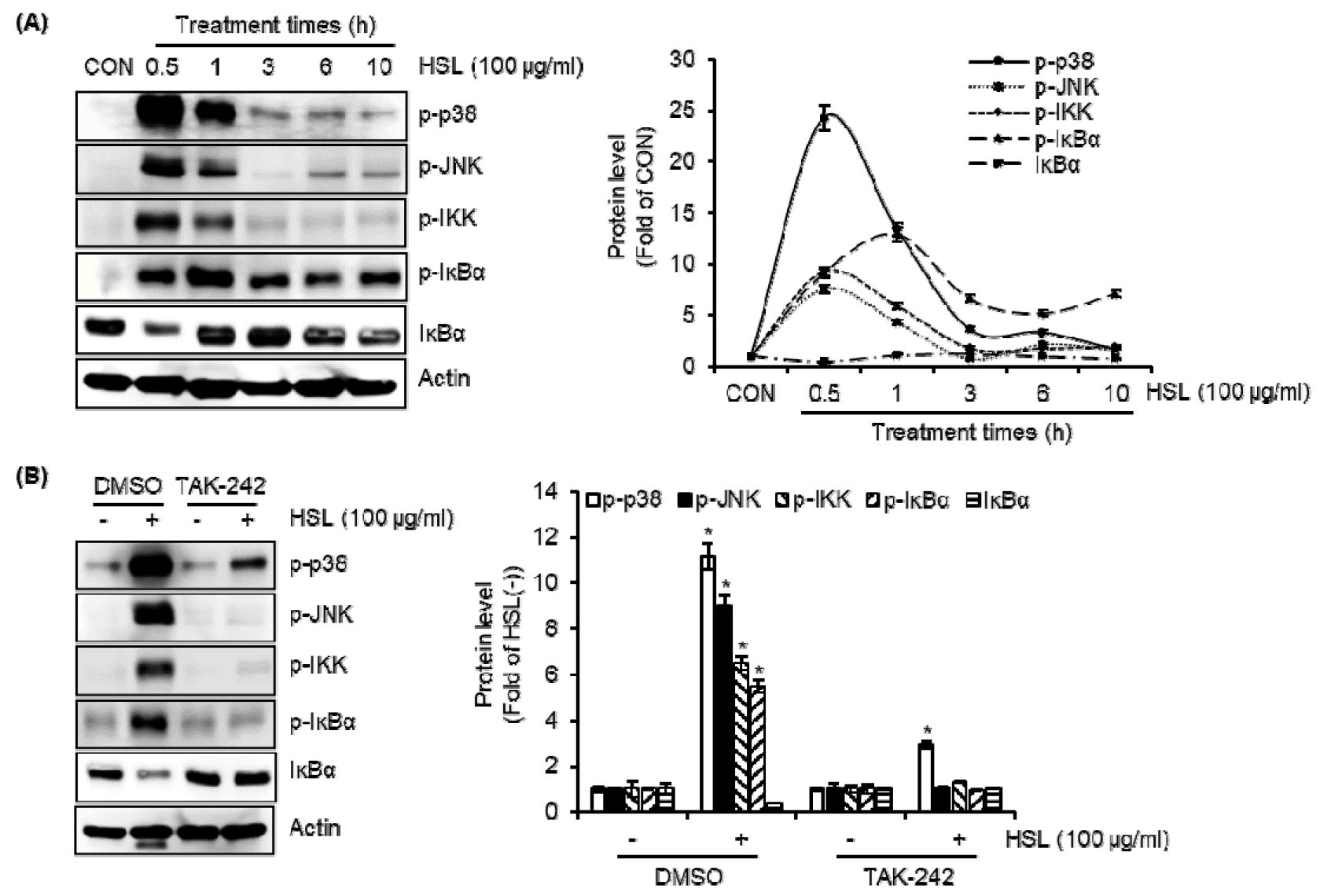
Fig. 4.
The effect of HSL on the activation of p38, JNK, and NF-κB in RAW264.7 cells. (A) RAW264.7 cells were treated with HSL (100 ㎍/mL) for the indicated times. (B) RAW264.7 cells were treated with HSL (100 ㎍/mL) in the absence or presence of TAK-242 (TLR4 inhibitor, 5 ㎍/mL) for 30 min. The level of p-p38, p-JNK, p-IKK, p-IκBα, or IκBα was analyzed by Western blot analysis.