Introduction
Materials and Methods
Reagents
Animals
Preparation of Pu-erh tea extract (PTE)
Induction of AD Skin lesions
Measurement of dermatitis severity
Experiment of Scratching behavioral
Cell culture
Cell viability assay
Cytokine assay
Histamine assay
Western blot analysis
Statistical analysis
Results
Effect of PET on DNCB-induced AD symptoms in mice
Effect of PET on the DNCB-induced histamine and IgE serum levels in mice
Effect of PET on compound 48/80-induced scratching behavior in mice
Effect of PET on the secretion of inflammatory cytokines in activated-HaCaT cells
Effect of PET on NF-kB activation in activated-HaCaT cells
Discussion
Introduction
Atopic dermatitis (AD) is a chronic inflammatory skin disease that mainly occurs in infancy or childhood (Berke et al., 2012). Recently, the incidence of AD has been increasing due to various reasons such as increased environmental pollution, western-style diet and increased stress (Buske-Kirschbaum et al., 2001). The main symptoms of AD are dry skin, itching, erythema, and eczematous lesions of the skin (Allam and Novak, 2006). AD is typically treated with steroids, antihistamines, and antibiotics, but long-term treatment can cause incidence of serious side effect such as immunosuppression, vasoconstriction, epidermal barrier dysfunction and skin thinning (Shiohara et al., 2004). Therefore, alternative treatments derived from natural products with high effectiveness and lower toxicity are being investigated as potential anti-AD agents.
Keratinocyte is main epidermal cell and play critical role in AD development (Humeau et al., 2022). In AD progression, keratinocyte induces various inflammatory mediators, including interleukin (IL)-1 and IL-6 and other chemokines, leading to tissue damage. Recent studies have reported that inflammatory cytokines are highly expressed in keratinocytes of AD patients (Zlotnik and Yoshie, 2000). Nuclear factor-kappa B (NF-kB) exerts a crucial function by affecting the expression of inflammatory-related genes (Lappas et al., 2002). NF-kB activation is a key step in the progression of AD. Increased NF-kB activity associated with the secretion of high levels of interleukin (IL)-6 and tumor necrosis factor (TNF)-a was shown to be involved in skin inflammation (Bunikowski et al., 2001). Thus, suppression of inflammatory cytokine and NF-kB activation may contribute to the development of a useful therapeutic strategy for treatment of AD.
Pu-erh tea, one of popular teas, has various health benefits, including prevention of type II diabetes and inhibition of tumor cell progression (Du et al., 2012; Lin and Lin-Shiau, 2006). It was reported that anti-inflammatory mechanism of Pu-erh tea via suppression the activation of HIF- 1a in LPS-stimulated RAW264.7 cells (Kim, 2023). Pu-erh tea promotes skeletal muscle glucose transport through activating Akt in rat (Ma et al., 2013). Pu-erh tea contains various type of effective ingredients, such as theanine, gallic acid, catechin, epicatecin, caffein, epigallocatechin (Du et al., 2012). However, research on the precise mechanism of Pu-erh tea on AD is not completely understood. This study aimed to investigate whether Pu-erh tea alleviate AD in vivo and in vitro. We determined the beneficial effect of Pu-erh tea on AD clinical symptoms induced by 2,4-dinitrochlrobenzene (DNCB) in mice. In addition, we investigated the effect of Pu-erh tea on the levels of inflammatory mediators and NF-kB activation in HaCaT cells.
Materials and Methods
Reagents
Fetal bovine serum (FBS) and Roswell Park Memorial Institute (RPMI)-1640 were purchased from JR Scientific, Inc. (Woodland, CA, USA). 2,2′-azino-bis (3-ethylbenzthiazoline-6-sufonic acid (ABTS), compound 48/80, DNCB, MTS (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxy phenyl)-2-(4-sulfophenyl)-2H-tetrazolium), phosphate-buffered saline (PBS), avidin peroxidase (AP), tween 80 and other reagents were purchased from Sigma (St. Louis, MO, USA). The mouse IgE and human IL-6/ IL-8 assay kits were purchased from BD Bioscience (San Diego, USA). Nuclear extraction kit, bicinchoninic acid (BCA) and enhanced chemiluminesence (ECL) were obtained from Pierce Thermo Scientific (Rockford, IL, USA). Specific NF-κB antibodies (Abs) was obtained from Santa Cruz Biotechnology (Dallas, TX, USA).
Animals
Male ICR mice (male, 6-weeks-old) was obtained from Hyochang Science (Daegu, Korea). Animals were kept under a 12 h light/dark cycle (light on 09:00-20:00) at room temperature (23 ± 2°C) and humidity (55 ± 10%). All animal experiments were conducted in accordance with the Animal Experiment Ethics Committee of Daegu Haany university guidelines (DHU2020-074).
Preparation of Pu-erh tea extract (PTE)
Pu-erh tea was obtained from tea market, Chaye (Wonju-si, Gangwon-do, Korea). The dried Pu-erh tea (100 g) was pulverized into a fine powder and decocted with distilled water (1 L) for 3 h. Afterwards, it was concentrated under reduced pressure using a rotary evaporator (Hahnshin S&T Co., Gyeonggi-do, Korea) and lyophilized in a freeze dryer (Eyela, Japan). The yield of the extract was about 9.1%. The resulting powder sample was stored at -20°C. The PTE was diluted in PBS and filtered through a 0.22 ㎛ filter.
Induction of AD Skin lesions
DNCB, diluted in vehicle (3 acetone: 1 olive oil) used as a sensitizer for inducing AD skin lesions in mice (Yang and Myung, 2023). Mice were shaved with a clipper before sensitization. Mice were divided into 4 groups (n= 6): group 1; vehicle, group 2; DNCB, group 3; DNCB plus 10 ㎎/㎏ of PET and group 4; DNCB plus 100 ㎎/㎏ of PET. Exposed skin was treated with 200 𝜇L of a 1% DNCB 3 times per week. After sensitization, the skin was challenged, with 200 𝜇L of a 0.25% DNCB solution every day for 2 weeks. PTE (10 ㎎/㎏ or 100 ㎎/㎏) was oral administrated for 2 weeks. After sacrifice, blood sample was collected and stored at -20°C.
Measurement of dermatitis severity
The dermatitis severity was measured according to the Eczema Area and Severity Index scoring system: 0, no symptom; 1, mild symptom; 2, moderate symptom; 3, severe symptom. The sums of the score were determined as the dermatitis severity for scaling/dryness, excoriation/erosionand, erythema/haemorrhage and oedema (Hanifin et al., 2001).
Experiment of Scratching behavioral
The behavioral experiments were performed according to the method of Sugimoto et al. (1998). The rostral part of mice was clipped and 50 μg/㎏ of compound 48/80 (dissolved in tween 80) was intradermally injected. Control mice received a tween 80 injection in place of the scratching agent. After the intradermal injection, the mice were put back into the same cage for the scratching observation. Scratching of the injected sites by the hind paw were evaluated and compared with those of the other sites. PTE (10 ㎎ /㎏ or 100 ㎎/㎏) was administered before the compound 48/80 injection. Scratching behaviors was counted for 30 min.
Cell culture
Human immortalized keratinocyte (HaCaT cells) was cultured in RPMI containing with 100 IU/mL penicillin, 100 ㎎/mL streptomycin, and 10% FBS at 37°C in 5% CO2 atmosphere at 95% humidity. HaCa T cells were stimulated with TNF-α (10 ng/mL) plus IFN-γ (10 ng/mL).
Cell viability assay
To determine the cell viability at various PTE concentrations, MTS assay was conducted. Cells were treated with various concentr‘ations of PTE (0.01, 0.1 and 1 ㎎/mL) for 12 h. After then, MTS (30 μL) was subsequently added for 2 h and the absorbance at 490 ㎚ was evaluated by a microplate reader.
Cytokine assay
Cytokine amount derived from cells were measured using modified enzyme-linked immunosorbent assay (ELISA). Briefly, micro plates were coated with monoclonal primary Abs against IL-8 and IL-6 Abs and incubated overnight at 4°C. After washes, the sample or standard solution of IL-8 and IL-6 was incubated for 2 h. After additional washing, the plates were incubated with biotinylated secondary Abs for 2 h. After washing, AP and ABTS substrate were sequentially reacted. The absorbance at 405 ㎚ was evaluated.
Histamine assay
Histamine concentration in serum was quantified with histamine assay kit according to the manufacturer’s protocol (Neogen, Lexington, USA).
Western blot analysis
Nuclear lysates derived from cells were separated using nuclear extraction kit. The protein was quantified with BCA assay kit and reacted at 100°C for 10 min with 2x sample lysis buffer. Afterwards, they were electrophoresed with 10% SDS-PAGE (SDS-Polyacrylamide gel) and transferred to nylon membranes. The membranes were blocked in 5% skim milk for 2 h and reacted with NF-kB p65 primary Abs at 4°C overnight. Afterwards, the membrane was incubated with secondary Abs for 2 h. After washing, protein levels were determined with ECL detection system.
Statistical analysis
Results are represented as the mean ± S.D. The statistical analyses were performed using an independent ANOVA with a Tukey post hoc test using SPSS statistics 23.0 (IBM SPSS, USA). P &5 was considered significant.
Results
Effect of PET on DNCB-induced AD symptoms in mice
To measure the improving effects of PET on the clinical symptoms of AD, mice were treated with DNCB. When the mice received PET (10 or 100 ㎎/㎏), AD symptoms including excoriation, skin thickening and erythema were significantly improved in DNCB-induced mice (Fig. 1A). Additionally, we analyzed the clinical severity score, indicator of AD severity. As shown in Fig. 1B, we observed that the skin severity of PET (10 ㎎/㎏ or 100 ㎎/㎏) group were significantly lowered than those of DNCB group.
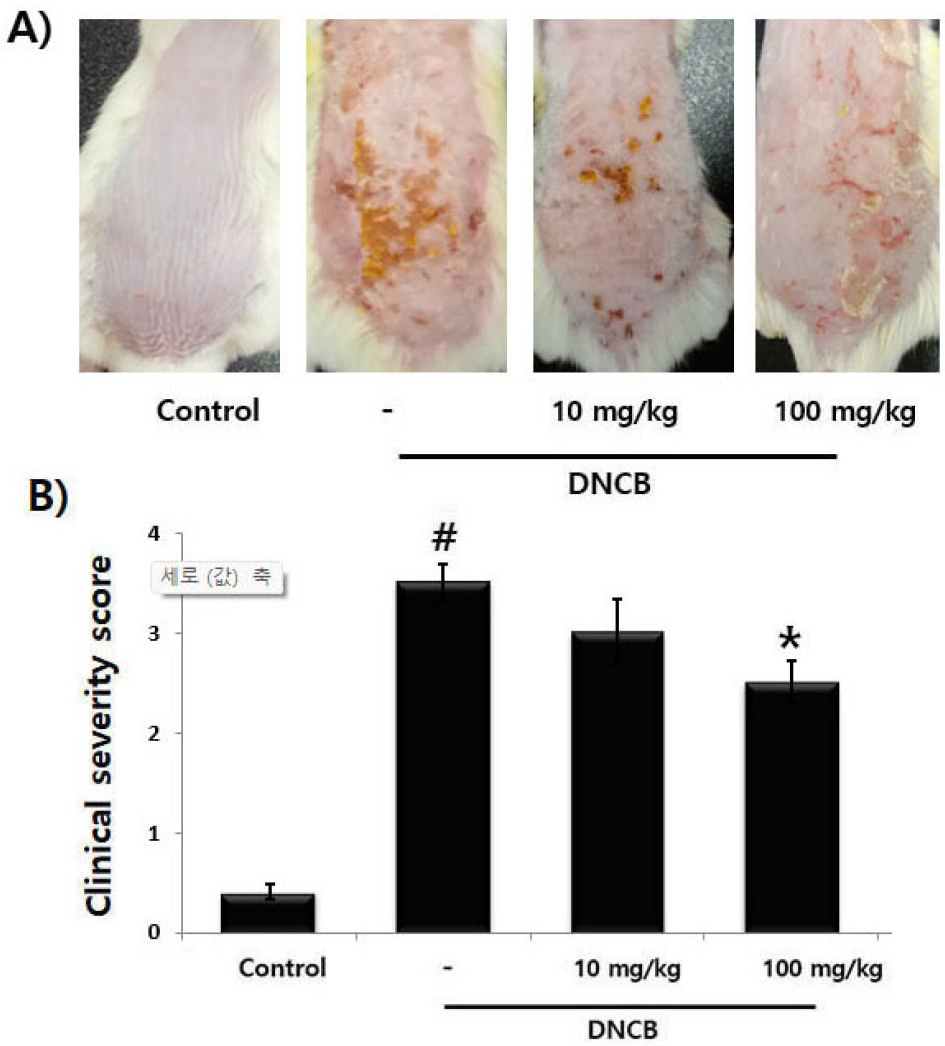
Fig. 1.
The regulatory effect of PET on DNCB-induced AD in mice. (A) The mice (n=6) were sensitized with DNCB applied to skin for 3 weeks. After 1 weeks, PET (10 ㎎/㎏) or (100 ㎎/㎏) was orally administered for 2 weeks. (B) The scores of skin severity are represented. Results are shown the mean ± S.D. of three independent experiments (#P &5 vs. control group, *P &5 vs. DNCB-alone treatment group).
Effect of PET on the DNCB-induced histamine and IgE serum levels in mice
Increase of histamine and IgE is main features of AD pathology. Especially, histamine is the cause of itching and is a major factor in inducing allergic inflammation. Inhibition of histamine levels may help treat AD. In this study, we investigated the effect of PET on the serum histamine and IgE levels. The results showed that PET reduced the serum of the histamine and IgE levels induced by DNCB in a concentration-dependent manner (Fig. 2A and B). The inhibition rate of the histamine and IgE levels by PET (100 ㎎/㎏) was approximately 31.5% (P &5) and 28.8% (P &5), respectively.
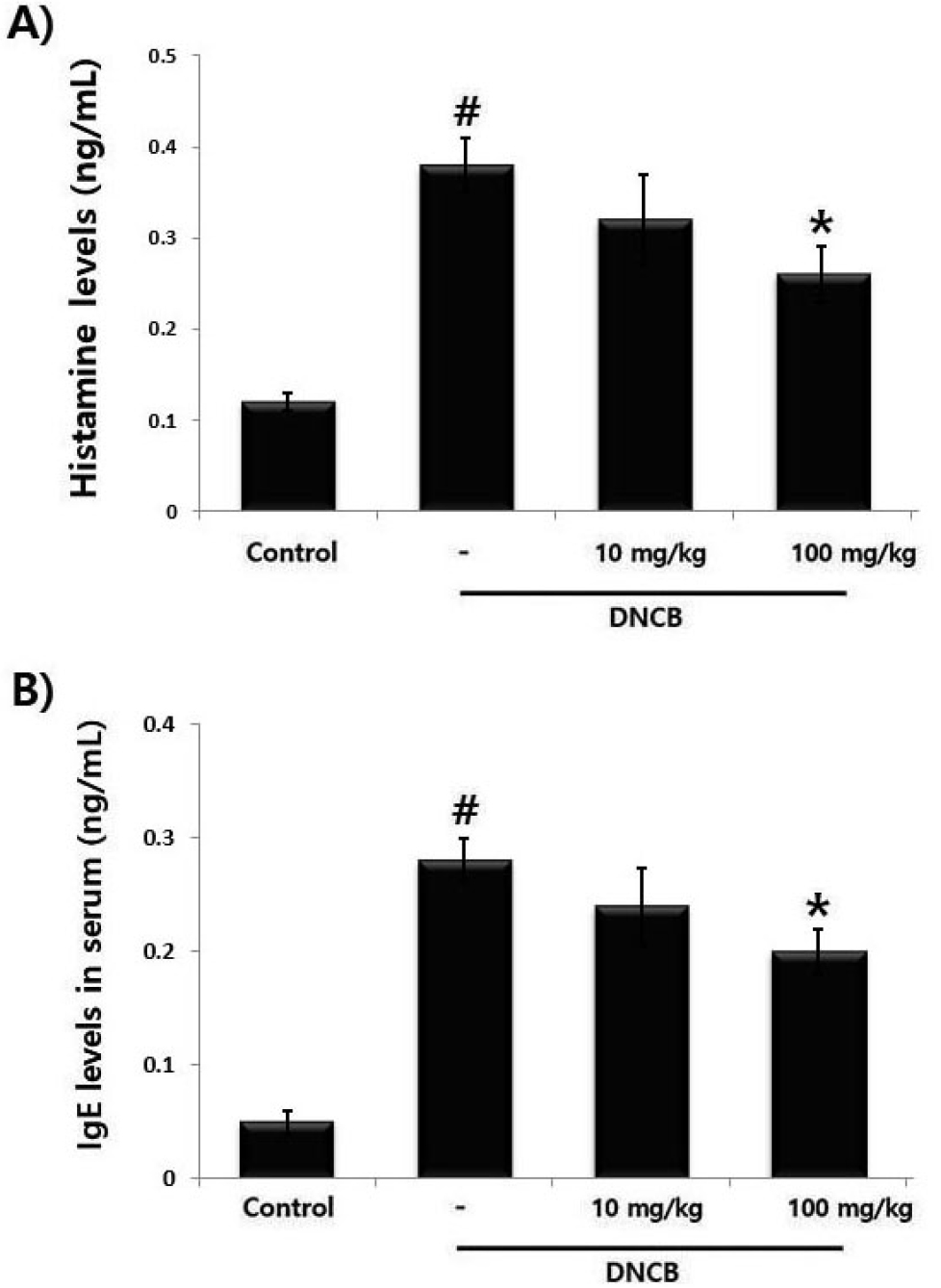
Fig. 2.
The regulatory effect of PET on DNCB-induced histamine and IgE serum levels mice. (A and B) Blood samples were collected and the amount of serum histamine and IgE in the indicated groups is measured using assay kits. Results are represented the mean ± S.D. of three independent experiments (#P &5 vs. control group, *P &5 vs. DNCB-alone treatment group).
Effect of PET on compound 48/80-induced scratching behavior in mice
Pruritus is known to be a representative clinical symptom of AD. The anti-pruritic effect of PET was determined on the compound 48/80-induced scratching behavior in mice. When the PET was administered, the scratching behaviors was decreased (Fig. 3). The inhibition rate of PET (100 ㎎/㎏) was approximately 28.4% (P &5).
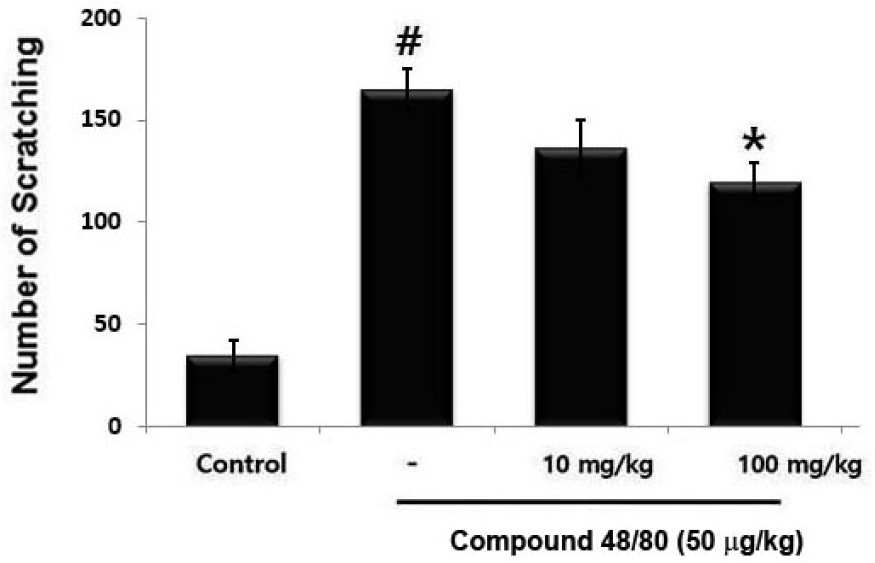
Fig. 3.
The regulatory effect of PET on compound 48/80- induced scratching behavior in mice. PET (10 ㎎/㎏) or (100 ㎎/㎏) was administered before compound 48/80 (50 μg/㎏) intradermal injection. Scratching behavior was counted for 30 min. Results are represents the means ± S.D. of three independent experiments (#P &5 vs. control group, *P &5 vs. compound 38/80-alone treatment group).
Effect of PET on the secretion of inflammatory cytokines in activated-HaCaT cells
Keratinocyte-derived cytokine and chemokine are associated with inflammatory responses of AD (Fedenko et al., 2011). To explore the effect of PET on cell viability, cells were incubated with PET (0.01, 0.1 and 1 ㎎/mL) for 24 h. After then, the cytotoxic effects of PET were measured via MTS assay. We observed that PET did not affect cell viability (Fig. 4A). To evaluate the anti-atopic effect of PET, cells were pretreated with various concentrations of PET (0.01, 0.1 and 1 ㎎/mL) and then incubated with TNF-α plus IFN-γ for 12 h. We measured whether PET regulates the IL-8 and IL-6 levels via ELISA. The results showed that PET significantly attenuated the TNF-α plus IFN-γ-induced the IL-8 and IL-6 secretion in a dose-dependent manner (Fig. 4B). The inhibition rates of IL-8 and IL-6 by PET (1 ㎎/mL) were approximately 46.9%, and 35.8%, respectively.
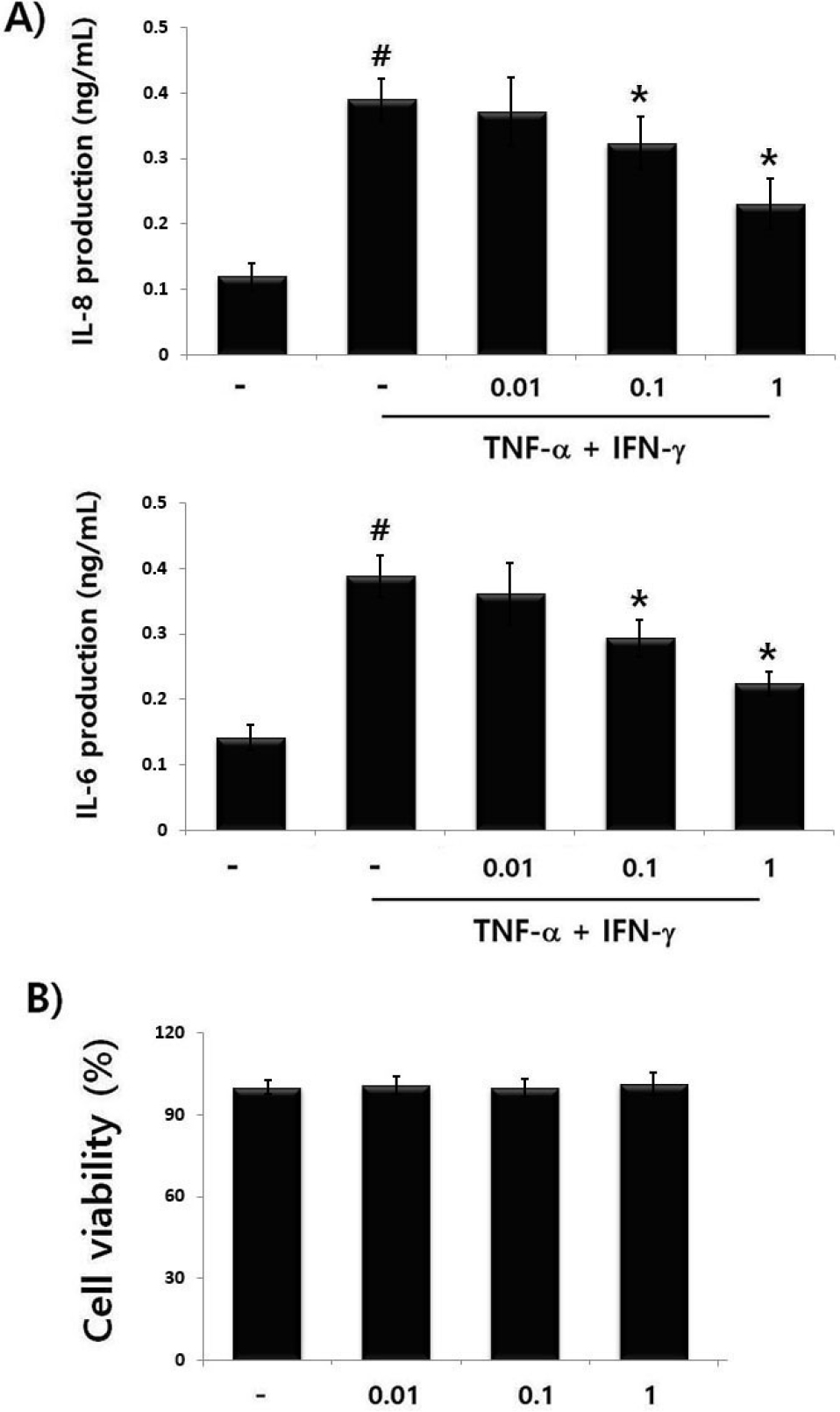
Fig. 4.
The regulatory effect of PET on cell viability and cytokine secretion in activated- HaCa T cells. (A) Cells were treated with PET (0.01, 0.1 and 1 ㎎/ mL) for 12 h and cell viability was evaluated by the MTS assay. (B) Cells were treated with PET (0.01, 0.1 and 1 ㎎/ mL) and then stimulated with TNF-α (10 ng/mL) + IFN-γ (10 ng/mL) for 12 h. The amount of IL-8 and IL-6 levels was measured using ELISA. Results are shown the mean ± S.D. of triplicate determinations from triplicate separate experiments (#P &5 vs. control, *P&5 vs. TNF-α+ IFN-γ stimulation).
Effect of PET on NF-kB activation in activated-HaCaT cells
As the attenuation of NF-kB activation has been linked to anti-inflammatory reaction (Gilmore and Garbati, 2011), we predicted that the anti-atopic mechanism of PET may be mediated through the down-regulation of NF-kB activation. In this study, we measured the regulatory effect of PET on the translocation of NF-kB p65 into the nucleus. Cells were pretreated with various concentrations of PET (0.01, 0.1 and 1 ㎎/mL) and then incubated with TNF-α plus IFN-γ for 2 h. Afterwards, nuclear NF-kB p65 levels were evaluated with the Western blot. The finding showed that TNF-α plus IFN-γ considerably increased the nuclear NF-kB p65 level, indicating the nuclear translocation of NF-kB p65. However, PET alleviated the increase in the nuclear NF-kB p65 levels in HaCaT cells (Fig. 5A). The relative rations of NF-kB p65 are shown in Fig. 5B.
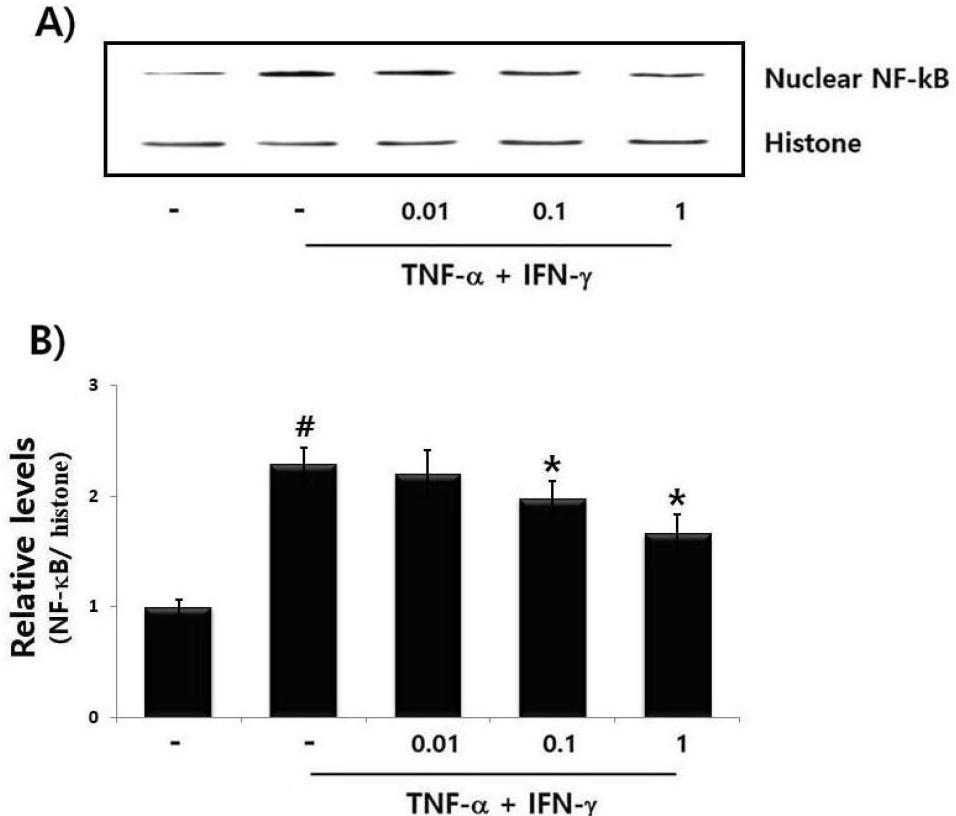
Fig. 5.
The regulatory effect of PET on NF-κB in activated HaCa T cells. Cells (6 × 106 cells/well) were treated with PET (0.01, 0.1 and 1 ㎎/ mL) and then activated with TNF-α (10 ng/mL) + IFN-γ (10 ng/mL) for 2 h. Nuclear extracts were isolated using nuclear extraction kit and measured for NF-κB (RelA/p65) by Western blot analysis. (B) The relative levels of NF-κB were shown. Results are represented in the mean ± S.D. of triplicate determinations from triplicate separate experiments (#P &5 vs. control, *P &5 vs. TNF-α + IFN-γ stimulation).
Discussion
Tea has been a subject of growing interest because of their health benefit properties. Pu-erh tea is known to exhibit beneficial effects on human health, such as anti-oxidative, anti-cancer and anti-obesity properties. However, the regulatory mechanisms of Pu-erh tea in AD-like dermatitis are not completely understood. The findings of this study showed that PET alleviated DNCB-induced AD symptoms and compound 48/80-induced pruritus in mice. Furthermore, we showed that the anti-atopic mechanism of PET was mediated by the suppression of inflammatory cytokine and NF-κB levels in HaCaT cells.
AD is an intractable skin disease whose representative symptoms are eczematous lesions and severe itching, which causes repeated and worsening rashes and inflammation (Gold and Kemp, 2005). Currently, steroids, antihistamines and antibiotics are used as treatments for AD, depending on the symptoms. However, it is known that these drugs cause various side effects when taken for a long period of time (Hengge et al., 2006). Therefore, there is a need to develop natural products-derived treatments with fewer side effects. DNCB is a substance that causes allergic contact dermatitis (Shiohara et al., 2004). It is known that after DNCB sensitization, hyperkeratosis and infiltration of inflammatory cells are observed in skin tissue, and a rapid inflammatory response occurs through the combined action of immune cells (Lee et al., 2010). In this study, we investigated the anti-atopic efficacy of PET in DNCB-induced AD model. The findings revealed that PET significantly relieved the DNCB-induced the symptoms of AD, such as excoriation, erythema and skin thickening in AD-like skin lesion. Additionally, PET attenuated the compound 48/80-induced scratching behavior in mice. Pruritus is a representative symptom of AD and is caused by excessive production of histamine (Schaper-Gerhardt et al., 2020). In this study, we determined the effect of PET on the serum histamine and IgE levels. As a result of the experiment, PET administration significantly reduced the DNCB-induced the histamine and IgE levels in mice. Our finding suggested that PET exerts anti-atopic effects by suppressing the AD symptoms and histamine and IgE levels in mice.
Keratinocyte is main epidermal cells that play a critical role in AD. Keratinocyte-derived cytokine and chemokine induces the infiltration of inflammatory cells into sites of inflammation. IL-6, one of inflammatory cytokines, aggravates AD lesions (Humeau et al., 2022). IL-8 is a chemotactic factor known to increase the infiltration of immune cells into skin inflammation lesions and tissue damage (Mukaida et al., 2000; Wong et al., 2001). It was reported that the levels of the AD-related cytokines were increased in keratinocytes of AD patients (Zlotnik and Yoshie, 2000). In this study, we investigated that PET regulates keratinocyte-derived the IL-8 and IL-6 levels. Our results showed that PET attenuated the IL-8 and IL-6 secretion in a dose-dependent manner in TNF-α plus IFN-γ-activated HaCaT cells. In particular, the inhibition rate of IL-8 and IL-6 release by PET (1 ㎎/mL) was approximately 46.9%, and 35.8%, respectively.
NF-κB is a transcription factor that regulates the expression of various genes. Activation of NF-κB is associated with AD pathogenesis through the overproduction of inflammatory cytokines (Han et al., 2023). In skin inflammatory process, NF-κB is translocated into nucleus and induced the synthesis of inflammatory cytokines and chemokine. Based on this phenomenon, alleviating of NF-kB activation has been identified as an anti-atopic strategy. To identify the anti-atopic mechanism of PET, we measured that PET could regulate the activation of nuclear NF-kB p65. The finding showed that the enhanced nuclear NF-kB p65 level was inhibited by PET in TNF-α plus IFN-γ-activated HaCaT cells. Thus, we suggested that PET exerts its anti-atopic mechanism through the down-regulation of nuclear NF-kB activation. The beneficial effects of Pu-erh tea are attributable to its bioactive components such as polyphenols, caffeine, flavonoids, catechins, thearubigins content (Roda et al., 2019). It was reported that theabrownin isolated from Pu-erh tea attenuated the intestinal inflammation through inhibition of the NF-κB and AKT pathways in mice (Zhao et al., 2024). In present study, the effect of major components of Pu-erh tea on AD progression was not elucidated. Thus, further study is necessary to clarify the role of bioactive component of Pu-erh in AD.
In conclusion, this study indicated that PET regulates AD progression in vivo by suppression DNCB-induced clinical symptoms of AD and compound 48/80-induced pruritus in mice. Furthermore, our results suggested that the anti-atopic mechanism of PET may be attributed to the attenuation of AD-related cytokines and nuclear NF-kB p65 activation in TNF-α plus IFN-γ-activated HaCaT cells. Therefore, our findings demonstrated that PET could potentially be developed as novel therapeutic agents for AD treatment.




