Introduction
Materials and methods
Plant material: Jeju native plants collection and in vitro culture
General Culture Conditions and Media Preparation
Optimization of in vitro conditions for AR cultures
Proliferation and bioreactor-based scale-up of AR cultures
Data collection and Calculation of Growth Parameters
Statistical analysis
Results
Screening and responsiveness grading of collected explants
Optimization of in vitro conditions for Grade I-ranked AR cultures
Biomass productivity performance and growth kinetics in bioreactor cultures of Grade I-ranked AR
Discussion
Appendix
Introduction
Jeju Island, designated as a UNESCO World Natural Heritage Site, harbors exceptional plant biodiversity shaped by its volcanic origin, diverse microclimates, and ecological isolation (Woo et al., 2013). This unique biogeographical context has fostered the evolution of numerous endemic and native plant species exhibiting distinct physiological and phytochemical traits (Kim, 2023; Son et al., 2025). Despite their ecological and biotechnological significance, particularly as reservoirs of specialized metabolites with cosmeceutical and pharmaceutical applications, their practical utilization remains limited owing to their ecological fragility, poor propagation efficiency, and lack of scalable cultivation protocols (Go, 2010; Kang et al., 2010).
The conservation and utilization of region-specific plant resources are emphasized under international frameworks such as the CBD and the Nagoya Protocol on ABS, both of which advocate the equitable use of genetic resources and the development of sustainable bioutilization pathways (Pörtner et al., 2023). However, traditional propagation methods, such as seed germination and vegetative cutting, are often ineffective for endemic, rare, or endangered species because of challenges such as seed dormancy, low germination rates, and environmental dependence (Ali, 2024; Kang et al., 2010). These limitations necessitate the establishment of ex situ propagation platforms that are capable of preserving genetic fidelity while enabling scalable biomass production.
In this context, in vitro plant culture systems have emerged as powerful tools for clonal propagation, biodiversity preservation, and bioactive compound production (Halder et al., 2021; Seo et al., 2025). These systems offer year-round aseptic production under precisely controlled conditions, thus ensuring reproducibility, disease-free biomass, and phytochemical consistency (Anuruddi et al., 2023). Moreover, by optimizing the basal medium composition, carbon source, and plant growth regulator concentrations, species- specific responses such as callus induction, direct shoot organogenesis, and adventitious root (AR) formation (Adem et al., 2024). Such responses are often governed by intrinsic physiological traits and the regulation of plant growth regulator-responsive transcription factors (Monfort et al., 2018).
To achieve industrial scalability, bioreactor technologies have been integrated with in vitro culture, enabling the mass production of high-value plant biomass (Bapat et al., 2023). Airlift and stirred-tank bioreactors, in particular, facilitate efficient gas exchange and nutrient delivery, which enhance cell proliferation and biomass yield (Sharma and Shahzad, 2013). These systems not only support high-throughput propagation but also serve as standardized platforms for downstream applications, including metabolite extraction, bioactivity validation, and production of plant-derived raw materials for the cosmetic and pharmaceutical industries (Murthy et al., 2023; Valdiani et al., 2019).
Accordingly, the present study has aimed to develop a technically optimized and scalable in vitro propagation platform for ecologically significant Jeju native plant species. An initial collection of 27 native taxa was screened for their response to culture initiation. Based on AR induction efficiency and morphological stability, four species, Hibiscus hamabo (HIH), Calystegia soldanella (CAS), Tetragonia tetragonoides (TET), and Lycoris chejuensis (LYC), were selected for further study. Aseptic cultures were established from various explants, followed by the systematic optimization of culture conditions, including basal medium type, carbon sources, and plant growth regulator combinations. Subculturing efficiency and biomass performance were evaluated over four propagation cycles, and the best-performing lines were scaled up using 5 L airlift bioreactors.
Materials and methods
Plant material: Jeju native plants collection and in vitro culture
The four Jeju native plants used in this study, CAS, HIH, LYC, and TET were collected from their natural habitats on Jeju Island between February 2020 and December 2022. Each of which was georeferenced in situ and complementary ecological metadata based on published literature and expert consultations (Chung et al., 2017; Chung et al., 2023; Go, 2010). Specific collection details and the explants used for in vitro culture are summarized in Appendix 1.
All sampling activities were conducted under permits issued by relevant local authorities. Notably, the collection of HIH, a designated endangered species at the time of study, was performed under special authorization from the Yeongsan River Basin Environmental Office (Permit Nos. 2021-8, 2022-5) (NIBR, 2021). Pressed voucher specimens for all collected taxa were deposited in the Jeju Biodiversity Database (JBRI-P09-003, JBRI-P15-006, JBRI-P14-008, JBRI-P16-045), in accordance with the integration standards of the National Institute of Biological Resources, Republic of Korea.
For in vitro culture initiation, freshly collected material, comprising seeds and vegetative tissues such as leaves, nodal segments, roots, and stems, was selected based on species- specific regenerative potential and tissue type. Explant sterilization followed a validated three-step surface disinfection protocol: (i) 15 min rinse under running tap water, (ii) immersion in 70% ethanol for 60 s, and (iii) treatment with 2.0% (v/v) sodium hypochlorite solution supplemented with Tween- 20 (1 drop per 100 mL) for 15 min. Residual disinfectant was removed by quadruple rinsing with sterile distilled water (Gantait and Kundu, 2017).
All handling procedures were performed aseptically under a Class II laminar airflow cabinet. Explant dissection, inoculation, and culture transfer were conducted using autoclaved instruments and flame-sterilized forceps and scalpels to prevent microbial contamination.
General Culture Conditions and Media Preparation
Unless otherwise specified, all media used in this study were supplemented with a vitamin complex (Duchefa Biochemie, Haarlem, Netherlands) and solidified with 2.3 g·L-1 gelrite. The pH of the media was adjusted to 5.8 using 1N NaOH or HCl prior to autoclaving at 121°C for 20 min. All cultures were maintained in a growth chamber at 25±1°C, dark condition and approximately 50% relative humidity.
Optimization of in vitro conditions for AR cultures
To establish optimized protocols for the induction of ARs in the four Grade I-ranked Jeju native plant species (HIH, CAS, TET, and LYC), a series of factorial experiments were conducted under controlled in vitro conditions. The experimental framework aimed to systematically assess the effects of four critical variables: (A) basal media composition, (B) sucrose concentration, (C) MS salt strength, and (D) type and concentration of auxins.
Five basal media formulations were tested: Murashige and Skoog (MS), McCown’s Woody Plant Medium (WPM), Gamborg’s B5, Schenk and Hildebrandt (SH), and Chu’s N6 (Chu, 1981; Gamborg et al., 1968; Lloyd and McCown, 1980; Murashige and Skoog, 1962; Schenk and Hildebrandt, 1972). These media were tested with sucrose at five concentrations (10, 20, 30, 40, and 50 g·L-1).
To assess the effects of mineral ion concentration on morphogenic response, MS medium was prepared at four different salt strengths: 1/8X, 1/2X, 1X (standard), and 2X. These were cross-assessed with species-specific auxin treatments. The auxins tested included indole-3-butyric acid and 1-naphthaleneacetic acid, applied at concentrations of 0.1, 1.0 and 2.0 ㎎·L-1. The cytokinins 6-benzylaminopurine, kinetin, and thidiazuron were also tested during preliminary trials but were excluded from final optimization because of limited root-promoting efficacy. Plant growth regulator regimes were selected based on prior empirical evidence and literature precedents (Bhatla and Lal, 2023).
Morphogenic responses were analyzed weekly for up to four weeks. Key metrics included: (i) Contamination rate (%): proportion of explants exhibiting microbial infection, (ii) Callus induction frequency (%): Proportion of explants forming unorganized cell masses, (iii) AR formation frequency (%): Number of explants forming distinct root structures, and (iv) Survival rate (%): Explants remaining viable without necrosis or discoloration. Photographic records and quantitative data were collected and statistically analyzed to identify optimal medium compositions and hormone regimes for each species.
Proliferation and bioreactor-based scale-up of AR cultures
Grade I-ranked AR explants of the four species (HIH, CAS, TET, and LYC) were selected for subculture-based multiplication and bioreactor scale-up (Lee et al., 2022a).
Initial AR cultures were maintained on their respective optimized solid media, with subculturing performed at four- or eight-week intervals over four cycles. Each batch consisted of a minimum of three biological replicates, and growth parameters were recorded at each cycle as described in next section.
For scale-up, liquid culture systems were established using 5 L airlift bioreactors (custom-fabricated, autoclavable borosilicate glass vessels fitted with silicone aeration tubing; modified from Duran® wide-neck flasks) with a 4 L working volume. Liquid media were prepared according to species- specific formulations (Table 1). Inoculum densities ranged from 1.0 to 5.0 g·L-1. Bioreactors were maintained under the general culture conditions, with continuous aeration using sterile filtered air and ambient light exposure. Culture periods were 4 or 8 weeks.
Table 1.
Optimized culture medium composition for adventitious root induction in Grade I-ranked Jeju native plant explants.
| Sample codez | Scientific name (Korean name) | Location site | Medium composition |
| JBRI-S20-003 | Hibiscus hamabo (황근) | Pyeongdae, Gujwa, Jeju | 1/2 MS, 40 g·L-1 sucrose, 2 ㎎·L-1 IBA |
| JBRI-S20-026 | Calystegia soldanella (갯메꽃) | Taeheung, Namwon, Seogwipo | MS, 30 g·L-1 sucrose, 0.1 ㎎·L-1 IBA |
| JBRI-S21-003 | Tetragonia tetragonoides (번행초) | Taeheung, Namwon, Seogwipo | MS, 30 g·L-1 sucrose, 0.1 ㎎·L-1 NAA |
| JBRI-S20-012 | Lycoris chejuensis (제주상사화) | Hannam, Namwon, Seogwipo | 1/2 MS, 30 g·L-1 sucrose, 2 ㎎·L-1 IBA |
Throughout the culture duration, morphological parameters such as root elongation, branching pattern, pigment production, and culture homogeneity were monitored and photographed.
Data collection and Calculation of Growth Parameters
To assess culture growth, fresh weight (FW) and dry weight (DW) were recorded. Dry weight was determined by drying samples in a forced-air drying oven at 40°C for 72 h.
Growth parameters were calculated using the following formulas:
Where W1 and W2 are the inoculation density (g·L-1) multiple by working volume (L) and FW (g FW·batch-1), respectively, and CP denotes the culture period (weeks).
Statistical analysis
All experimental data were analyzed using a completely randomized design, comprising at least three biological replicates per treatment, unless otherwise specified. Analysis of variance (ANOVA) was performed to determine the significance of differences among treatment means. Where significant differences were observed, mean comparisons were conducted using Duncan’s Multiple Range Test (DMRT) at a 5% probability level (p &5). All statistical procedures were carried out using the SAS studio software package (SAS Institute Inc., Cary, NC, USA).
Results
Screening and responsiveness grading of collected explants
A total of 41 explants from 27 native species were collected (Appendix 1) and evaluated for their in vitro morphogenic responsiveness. This responsiveness was categorized based on a grading system defined in the Materials and Methods.
The screening revealed that only four samples (9.75%) were graded as Grade I, demonstrating consistent and reproducible AR formation under the initial in vitro conditions. The high-performance explants included Hibiscus hamabo (JBRI-S20-003), Lycoris chejuensis (JBRI-S20-012), Calystegia soldanella (JBRI-S20-026), and Tetragonia tetragonoides (JBRI-S21-003).
Most of the remaining explants were categorized as either Grade II (68.30%, 28 samples), indicating moderate responsiveness, or Grade III (21.95%, 9 samples), reflecting recalcitrant or unresponsive behavior under the tested conditions.
Optimization of in vitro conditions for Grade I-ranked AR cultures
Among the five basal media (B5, MS, N6, SH, and WPM) tested, MS medium consistently yielded the highest AR induction rates across all four species (p &5, DMRT; Fig. 1A). Notably, HIH and CAS exhibited AR induction frequencies exceeding 90% on the MS medium, whereas SH and N6 showed markedly lower responses (<60%).
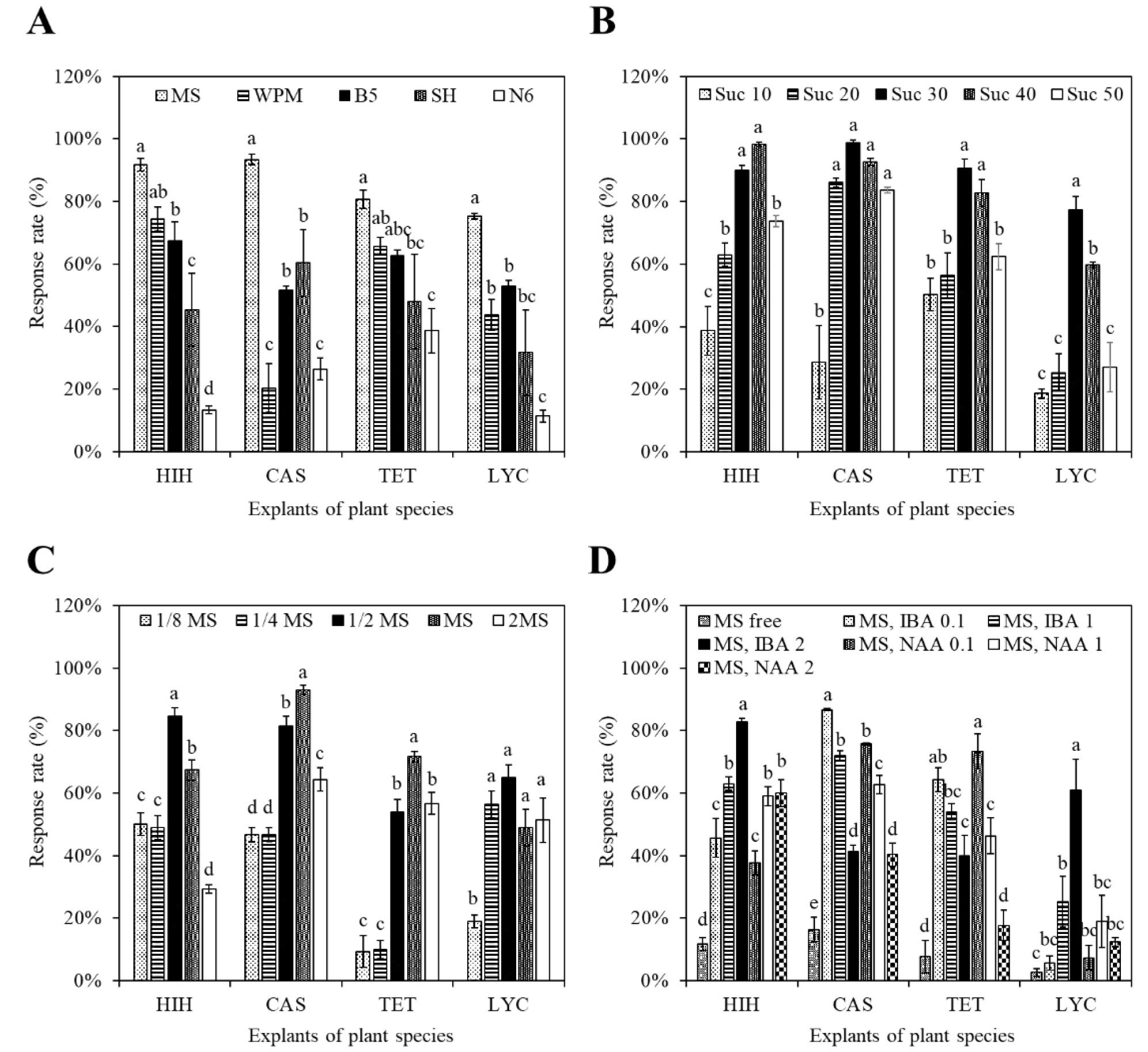
Fig. 1.
Effects of culture conditions on adventitious root induction in Grade I-ranked Jeju native plant explants. (A) Basal medium (MS, WPM, B5, SH, N6), (B) Sucrose concentration (10-50 g·L⁻¹), (C) MS salt strength (1/8X to 2X), and (D) Auxin type and concentration (IBA, NAA at 0.1, 1.0, 2.0 ㎎·L⁻¹). Data represent means ± standard error (n = 3). Different letters denote statistically significant differences (p &5, DMRT). Jeju native plant explants are Hibiscus hamabo (HIH), Calystegia soldanella (CAS), Tetragonia tetragonoides (TET), and Lycoris chejuensis (LYC).
Sucrose concentration was a critical determinant of AR initiation (Fig. 1B). A concentration of 30 g·L-1 yielded the highest rooting frequencies across all species, while suboptimal (10 g·L-1) or supra-optimal (≥40 g·L-1) concentrations resulted in significant reductions in AR formation.
Salt concentration significantly influenced the rooting outcomes (Fig. 1C). 1/2 MS facilitated the highest AR induction in HIH and LYC. Conversely, CAS and TET responded best to full-strength MS. In all of the cases, AR induction was severely inhibited at both extreme dilutions (1/8 MS) and concentrations (2 MS).
The application of exogenous auxins is essential for AR induction (Fig. 1D). IBA at 2.0 ㎎·L-1 promoted the most robust AR formation and elongation, particularly in HIH and LYC. In contrast, higher concentrations of NAA (≥2.0 ㎎·L-1) led to excessive callus proliferation without root formation, especially in CAS and TET. Auxin-free media failed to induce AR development across all species.
Two-way ANOVA revealed a significant main effect (p &5) of basal medium, sucrose concentration, and auxin concentration on the AR induction frequency across all species. Additionally, significant interaction effects were observed, indicating species-specific synergies between the culture parameters.
The optimal AR induction conditions for each species are presented (Table 1): (i) HIH: 1/2 MS, 40 g·L-1 sucrose, and 2 ㎎·L-1 IBA, (ii) CAS: MS, 30 g·L-1 sucrose, and 0.1 ㎎·L-1 IBA, (iii) TET: MS, 30 g·L-1 sucrose, and 0.1 ㎎·L-1 NAA, and (iv) LYC: 1/2 MS, 30 g·L-1 sucrose, and 2 ㎎·L-1 IBA.
Biomass productivity performance and growth kinetics in bioreactor cultures of Grade I-ranked AR
HIH demonstrated a progressive increase in FW and DW across cycles, reaching a maximum DW of 30.80 g and RGR of 3.19 in the fourth cycle (Table 2). Its DM decreased from 10.34% to 7.96%.
Table 2.
Biomass productivity and growth kinetics from adventitious root cultures of Hibiscus hamabo cultured in 5 L airlift bioreactors of four subculture cycles.
| Subculture cycles | Inoculation density (g·L-1) |
Fresh weight (g FW·batch-1) |
Dry weight (g DW·batch-1) |
Dry matterz (%) |
Growth indexy | Relative growth ratex |
| 1st | 4.0 | 206.31cw | 21.33c | 10.34a | 11.89c | 2.56d |
| 2nd | 228.12c | 20.72c | 9.08b | 13.26c | 2.66c | |
| 3rd | 306.62b | 25.43b | 8.30c | 18.16b | 2.95b | |
| 4th | 389.21a | 30.80a | 7.96c | 23.33a | 3.19a |
yGrowth index = [Fresh weight (g FW·batch-1) – {Inoculation density (g·L-1) × working volume (L)}] / [Inoculation density (g·L-1) × working volume (L)].
CAS achieved the best proliferation metrics among all tested species (Table 3). Despite a minimal inoculum (1.0 g·L-1), FW exceeded 400 g FW·batch-1 from the second cycle, and DW peaked 31.74 g DW·batch-1 in the fourth cycle. Notably, GI and RGR reached 104.65 and 4.66, respectively.
Table 3.
Biomass productivity and growth kinetics from adventitious root cultures of Calystegia soldanella cultured in 5 L airlift bioreactors of four subculture cycles.
| Subculture cycles |
Inoculation density (g·L-1) |
Fresh weight (g FW·batch-1) |
Dry weight (g DW·batch-1) | Dry matterz (%) |
Growth indexy | Relative growth ratex |
| 1st | 1.0 | 364.67bw | 26.88c | 7.38ab | 90.17b | 4.51b |
| 2nd | 409.72a | 28.41b | 6.93b | 101.43a | 4.63a | |
| 3rd | 412.06a | 29.17b | 7.08ab | 102.01a | 4.63a | |
| 4th | 422.61a | 31.74a | 7.52a | 104.65a | 4.66a |
yGrowth index = [Fresh weight (g FW·batch-1) – {Inoculation density (g·L-1) × working volume (L)}] / [Inoculation density (g·L-1) × working volume (L)].
TET exhibited a steady biomass increase in each cycle, achieving 304.88 g FW·batch-1 and 21.79 g DW·batch-1 by the fourth cycle (Table 4). The DM remained stable (~6.6%), indicating balanced water content and tissue integrity.
Table 4.
Biomass productivity and growth kinetics from adventitious root cultures of Tetragonia tetragonoides cultured in 5 L airlift bioreactors of four subculture cycles.
| Subculture cycles |
Inoculation density (g·L-1) |
Fresh weight (g FW·batch-1) |
Dry weight (g DW·batch-1) |
Dry matterz (%) |
Growth indexy | Relative growth ratex |
| 1st | 5.0 | 135.91dw | 8.56d | 6.29c | 5.80d | 1.92d |
| 2nd | 163.23c | 10.07c | 6.17c | 7.16c | 2.10c | |
| 3rd | 256.42b | 17.49b | 6.82b | 11.82b | 2.55b | |
| 4th | 304.08a | 21.79a | 7.18a | 14.20a | 2.72a |
yGrowth index = [Fresh weight (g FW·batch-1) – {Inoculation density (g·L-1) × working volume (L)}] / [Inoculation density (g·L-1) × working volume (L)].
LYC showed the slowest proliferation kinetics, with FW increasing modestly to 104.99 g FW·batch-1 in the fourth cycle (Table 5). However, it consistently maintained a high DM (>10%) and a relatively low mean RGR (1.79).
Table 5.
Biomass productivity and growth kinetics from adventitious root cultures of Lycoris chejuensis cultured in 5 L airlift bioreactors of four subculture cycles.
| Subculture cycles |
Inoculation density (g·L-1) |
Fresh weight (g FW·batch-1) |
Dry weight (g DW·batch-1) |
Dry matterz (%) |
Growth indexy | Relative growth ratex |
| 1st | 4.0 | 92.52bw | 9.37b | 10.13b | 4.78b | 1.75b |
| 2nd | 91.89b | 9.39b | 10.22ab | 4.74b | 1.75b | |
| 3rd | 94.71b | 9.54b | 10.07b | 4.92b | 1.78b | |
| 4th | 104.99a | 11.50a | 10.96a | 5.56a | 1.88a |
yGrowth index = [Fresh weight (g FW·batch-1) – {Inoculation density (g·L-1) × working volume (L)}] / [Inoculation density (g·L-1) × working volume (L)].
Quantitative comparisons established a species-specific hierarchy of proliferation efficiency: CAS > HIH > TET > LYC. The visual assessment of the bioreactor-grown roots (Fig. 2) supported these findings. CAS and HIH produced fine, fibrous, and highly branched ARs, which are favorable for liquid-phase nutrient absorption. In contrast, TET were moderately thickened with fewer lateral branches, whereas LYC formed compact, nodular structures with low branching density, potentially limiting gas and nutrient exchange.

Fig. 2.
Morphological comparison of adventitious root cultures in plate (Top row) and 5 L airlift bioreactor (Bottom row). (A, E) Hibiscus hamabo, (B, F) Calystegia soldanella, (C, G) Tetragonia tetragonoides, and (D, H) Lycoris chejuensis. The dark, opaque appearance of the medium in (G) is not contamination but is due to the significance exudation of phenolic compounds from the T. tetragonoides roots after 4 weeks of culture.
Discussion
The successful in vitro morphogenesis of native plants is often limited by their recalcitrant nature. In our initial screening, clear trends emerged where AR formation was predominantly observed in rhizomatous or herbaceous species (CAS, TET), which are known for their high regenerative capacity and rapid cellular differentiation (Ko et al., 2008; Lee et al., 2021; Lee et al., 2024). Conversely, woody species such as Ficus erecta and Sageretia theezans predominantly produce calli or exhibit limited shoot or root organogenesis (Hesami et al., 2018; Kim et al., 2023). These trends are likely attributable to their lower cellular plasticity, persistent dormant structures such as bulbs, and elevated levels of phenolic compounds, which are known to interfere with in vitro morphogenesis (Bach et al., 2015). The activation grading system introduced in this study thus proved effective for rapidly identifying high-potential species, underscoring the need for species-specific protocols.
The subsequent optimization phase confirmed the critical role of media components. The superior performance of MS medium is likely attributable to its higher total nitrogen and macronutrient composition, which support enhanced cellular proliferation in herbaceous species (Monfort et al., 2018; Phillips and Garda, 2019). The responsiveness of TET and LYC to both MS and WPM indicated their adaptability. Sucrose at 30 g·L-1 was optimal, aligning with previous studies that identify sucrose as both a key carbon source and an osmotic agent to support energy metabolism and early organogenic signaling (Saddhe et al., 2021; Yaseen et al., 2013). Declines at ≥40 g·L-1 are likely due to osmotic stress. Similarly, salt concentration was critical; the success of 1/2 MS for HIH and LYC (halophytic or geophytic species) suggests that reduced ionic strength may mitigate osmotic pressure or ion toxicity (Kaleem et al., 2022; Lee et al., 2022b). In all of the cases, AR induction was severely inhibited at both extreme dilutions (1/8 MS) and concentrations (2 MS), confirming that deviations from optimal ionic balance disrupted morphogenic competence (Lee et al., 2021; Pasternak and Steinmacher, 2024). The indispensable role of auxins was also confirmed, with species-specific preference for IBA (CAS, HIH, LYC) versus NAA (TET) highlighting differential auxin signaling responses (da Costa et al., 2018; Pasternak and Steinmacher, 2024). These findings emphasize that the refined, species-tailored protocols developed here are a robust basis for scalable propagation.
In the bioreactor scale-up, the proliferation hierarchy (CAS > HIH > TET > LYC) likely reflects fundamental physiological differences associated with their life forms (Druege et al., 2019). The species tested are categorized as herbaceous plants (CAS, TET), a woody shrub (HIH), and a geophyte (LYC). Herbaceous species like CAS generally exhibit rapid in vitro cell proliferation (Baque et al., 2012), while woody plants (HIH) can be recalcitrant due to phenolic compound secretion (McCown, 2000), and geophytes (LYC) may have slower kinetics related to dormancy metabolism. It was precisely to address these known differences that various media, including WPM, were evaluated during the initial optimization (Li et al., 2023; McCown and Sellmer, 1987). The observed root morphologies (Fig. 2), from the fine, fibrous of CAS and HIH to the compact, nodular structures of LYC, directly support these growth differences, as morphology impacts nutrient and gas exchange efficiency.
The progressive increase in biomass across four cycles in HIH and TET suggests successful adaptation. This trend can be mechanistically attributed to the physiological adaptation of the root tissues to the high-aeration, liquid-phase environment and a potential selection process favoring cell lines with higher proliferation and shear stress tolerance (Murthy et al., 2024). If these cultures were extended beyond four cycles, growth would likely reach a plateau due to nutrient depletion, metabolite accumulation, or physical space constraints (Isah et al., 2018). Furthermore, long-term subculturing poses risks of somaclonal variation or a decline in vigor due to senescence (Majumder et al., 2025).
These findings have confirmed the feasibility of using airlift bioreactor systems for mass production of AR biomass from Jeju native plants (Hussain et al., 2022; Pasternak and Steinmacher, 2024). Moreover, the observed root morphologies have critical downstream implications for drying kinetics, pulverization efficiency, and compound extraction yield (Bapat et al., 2023; Valdiani et al., 2019).
In conclusion, this study successfully established and validated a bioreactor-based AR culture platform for Jeju native plants. The identification of high-performance candidates like CAS and HIH, with robust biomass productivity, confirms the industrial feasibility of this system. This platform provides a scalable template that serves a dual purpose: (i) the ex situ conservation of valuable plant genetic resources and (ii) a sustainable, industrial-scale production system for phytochemicals without ecological pressure on wild populations. This approach, which aligns with global biodiversity frameworks (CBD, ABS) and ESG principles, lays the groundwork for subsequent studies on elicitor-mediated metabolite enrichment, phytochemical profiling, and cosmetic validation.




